Application of nitrogen-containing heterocyclic ester compounds in the preparation of drugs against Coxsackie virus type b3
A technology of coxsackie virus and heterocyclic ester, which is applied in the directions of antiviral agents, medical preparations containing active ingredients, and pharmaceutical formulations, can solve problems such as evaluation of biological activity, and achieves economical, rapid, simple synthesis process, The effect of easy mass production and promotion
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
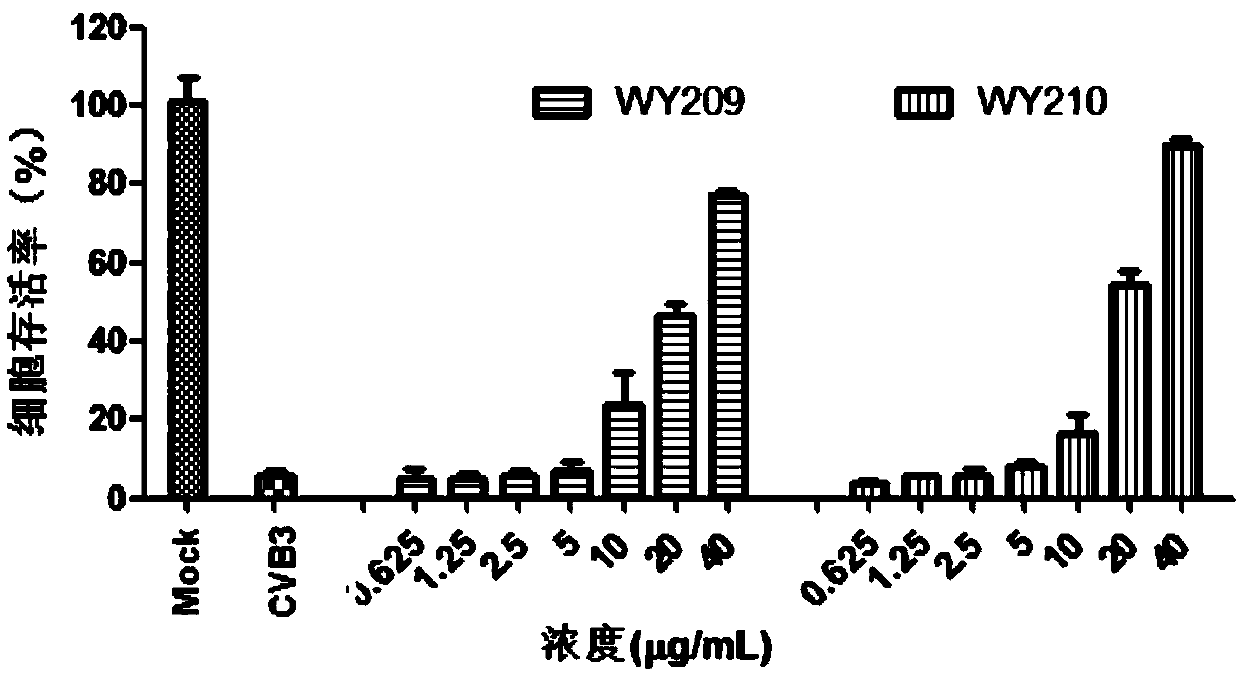
[0020] [Example 1] Evaluating the anti-CVB3 activity of WY209 and WY210
[0021] 1. Test method:
[0022] 1.1 Toxicity of compounds to host Hep-2 cells
[0023] Hep-2 cells were plated in 96-well plates at 37°C, 5% CO 2 After the monolayer was grown in the incubator, the cell culture solution was discarded, and the cell maintenance solution containing different concentrations of the test compound was added to continue the culture. After 48 hours, the cytotoxicity was visually observed under the microscope and recorded respectively, and the cell survival rate was determined by the MTT method. SPSS 11.5 software calculates the median toxic concentration (Median cyctoxic concentration, CC50) of the drug on the cells. Cell viability=(average OD of drug group 492 Value / average OD of cell control group 492 value) × 100%.
[0024] 1.2 Inhibitory activity of compounds against CVB3
[0025] Hep-2 cells were plated in 96-well plates at 37°C, 5% CO 2 After the monolayer was grown ...
Embodiment 2
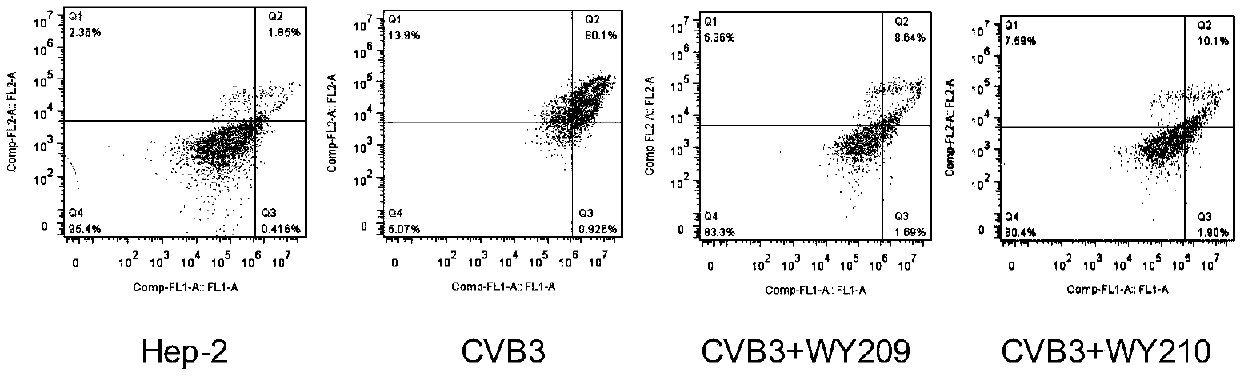
[0034] [Example 2] Inhibitory effect of WY209 and WY210 on Hep-2 cell apoptosis caused by CVB3
[0035] 1. Test method
[0036] Hep-2 cells in the logarithmic growth phase were plated on 24-well plates, and the cells were infected with 100TCID50CVB3 after the monolayer was overgrown. After incubation at 37°C for 1.5h, the virus solution was removed, and the cell maintenance solution containing 40μg / mL WY209 and WY210 was added. After about 48 hours, the cells were collected, and the apoptosis was detected on a flow cytometer using the Annexin V-FITC / PI apoptosis detection kit.
[0037] 2. Test results
[0038] Experimental results such as image 3 As shown, 40μg / mL WY209 and WY210 can effectively inhibit the apoptosis caused by CVB3. When the apoptosis rate of the virus control group was 81.02%, and the apoptosis rate of the normal untreated cells was 2.26%, the apoptosis rates of the cells treated with 40 μg / mL WY209 and WY210 were 10.33% and 12%, respectively. It can be ...
Embodiment 3
[0039] [Example 3] The inhibitory effect of WY210 on the production of CVB3 progeny virus
[0040] 1. Test method
[0041] Hep-2 cells in the logarithmic growth phase were plated in 24-well plates, and 100TCID after the monolayer was overgrown 50 CVB3 infected cells, incubated at 37°C for 1.5h, removed the virus solution, washed three times with PBS, and added cell maintenance solution containing 40μg / mL WY210. After 48 hours, the cells and supernatant were collected and lysed by freezing and thawing three times at -20°C and 37°C. TCID 50 Methods To determine the titer of CVB3 virus.
[0042]2. Test results
[0043] like Figure 4 As shown, the virus titer of Hep-2 cells treated with 40 μg / mL WY210 was significantly decreased compared with the virus control group, indicating that the compound had a strong inhibitory effect on the production of CVB3 progeny virus.
[0044] In summary, the ester compounds WY209 and WY210 with novel structures have strong inhibitory activity...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More