Nano emulsion as well as preparation method and application thereof
A nano-emulsion, nano-emulsion technology, applied in general/multifunctional contrast agents, pharmaceutical formulations, photodynamic therapy and other directions, can solve problems such as limited efficacy, achieve good application prospects, and enhance the effect of photodynamic therapy.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
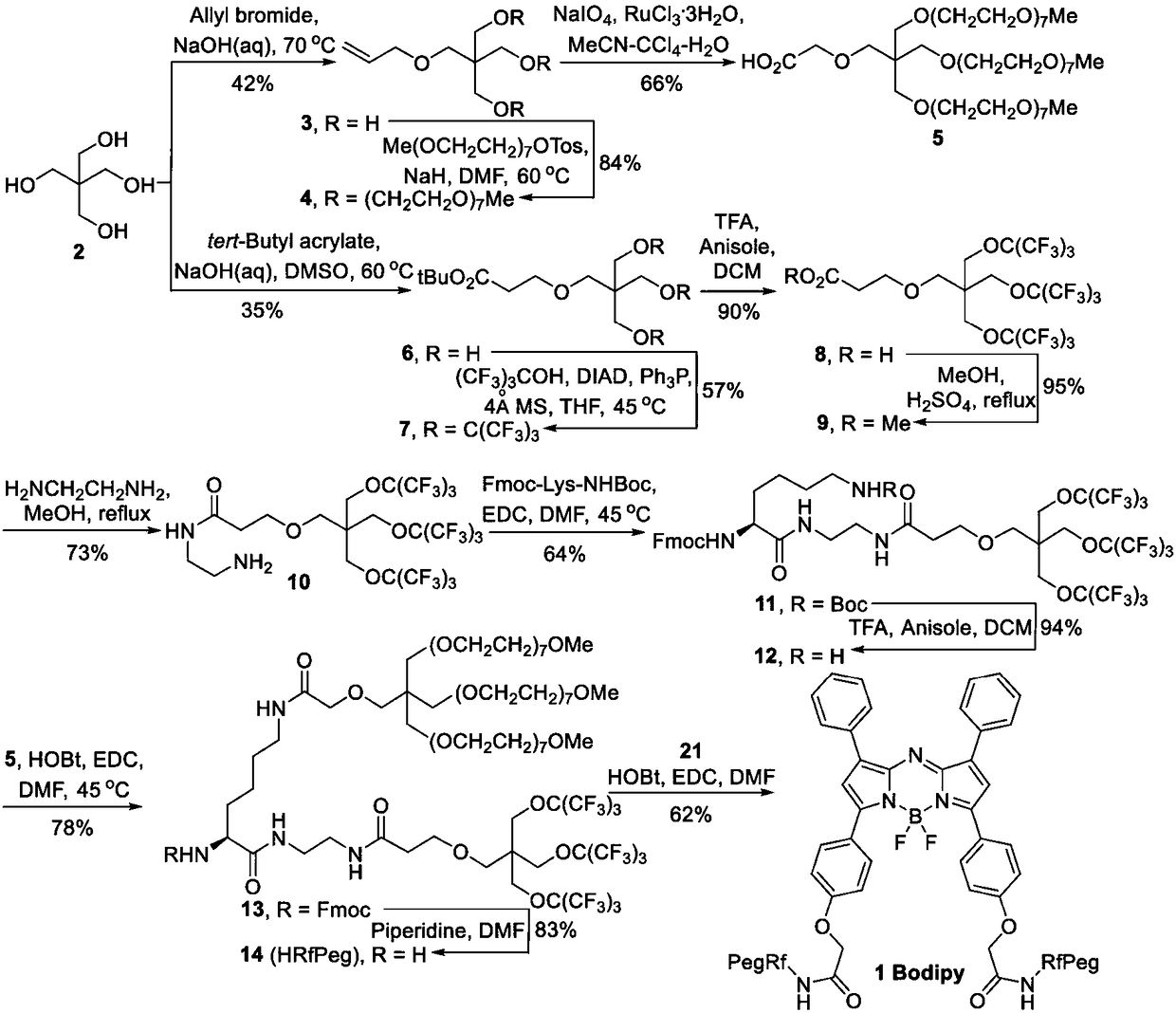
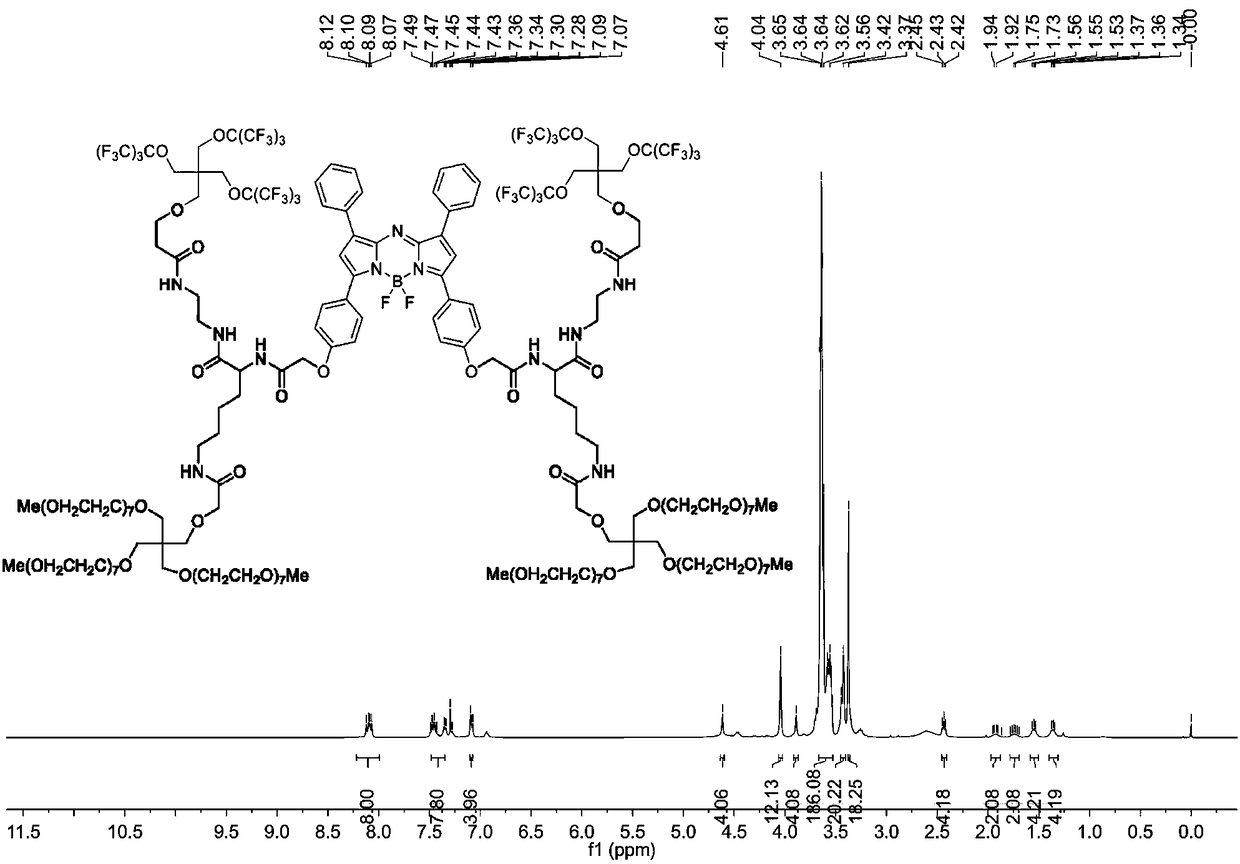
[0063] The preparation of novel Bodipy amphiphilic compound (synthetic route sees figure 1 )
[0064] 1) Preparation of compound 3:
[0065] Pentaerythritol (68.0 g, 0.5 mol) was dissolved in NaOH solution (9.6 g, 200 mL), and allyl bromide (24.2 g, 0.2 mol) was added dropwise and stirred for 2 hours. The reaction was heated to 70°C for 8 hours. The mixture was diluted with 100 mL of water and extracted with EA (150 mL x 5). The combined organic layers were dried over anhydrous sodium sulfate, concentrated in vacuo and purified by silica gel column (PE / EA=1 / 1) to obtain compound 3 as a clear oil. 1 H NMR (400MHz, CDCl 3 )δ5.82-5.92(m,1H),5.18-5.28(m,2H),3.97(d,J=8.0Hz,2H),3.76(s,3H),3.68(s,6H),3.44(s ,2H).
[0066] 2) Preparation of compound 4:
[0067] All the following operations were carried out under argon protection. A DMF solution of compound 3 (7.0 g, 40.0 mmol) was added dropwise to a DMF suspension of NaH (5.8 g, 240.0 mmol) under ice-cooling conditions. Afte...
Embodiment 2
[0091] Preparation of nanoemulsions
[0092] 1) Dissolve compound 1 in water (0.1 mg / mL), add PFH (0.5% v / v), stir for 10 minutes, and sonicate (100 W) in an ice bath to form a uniform nanoemulsion. Then, the compounds in the supernatant were removed by low-speed centrifugation to obtain a high-concentration nanoemulsion. Infuse oxygen with an oxygen pump for 5 minutes.
[0093] 2) The nanoemulsion preparation method for in vivo testing is the same as that described in step 1), the concentration of compound 1 is 1 mg / mL, and the concentration of perfluorohexane is 3% v / v.
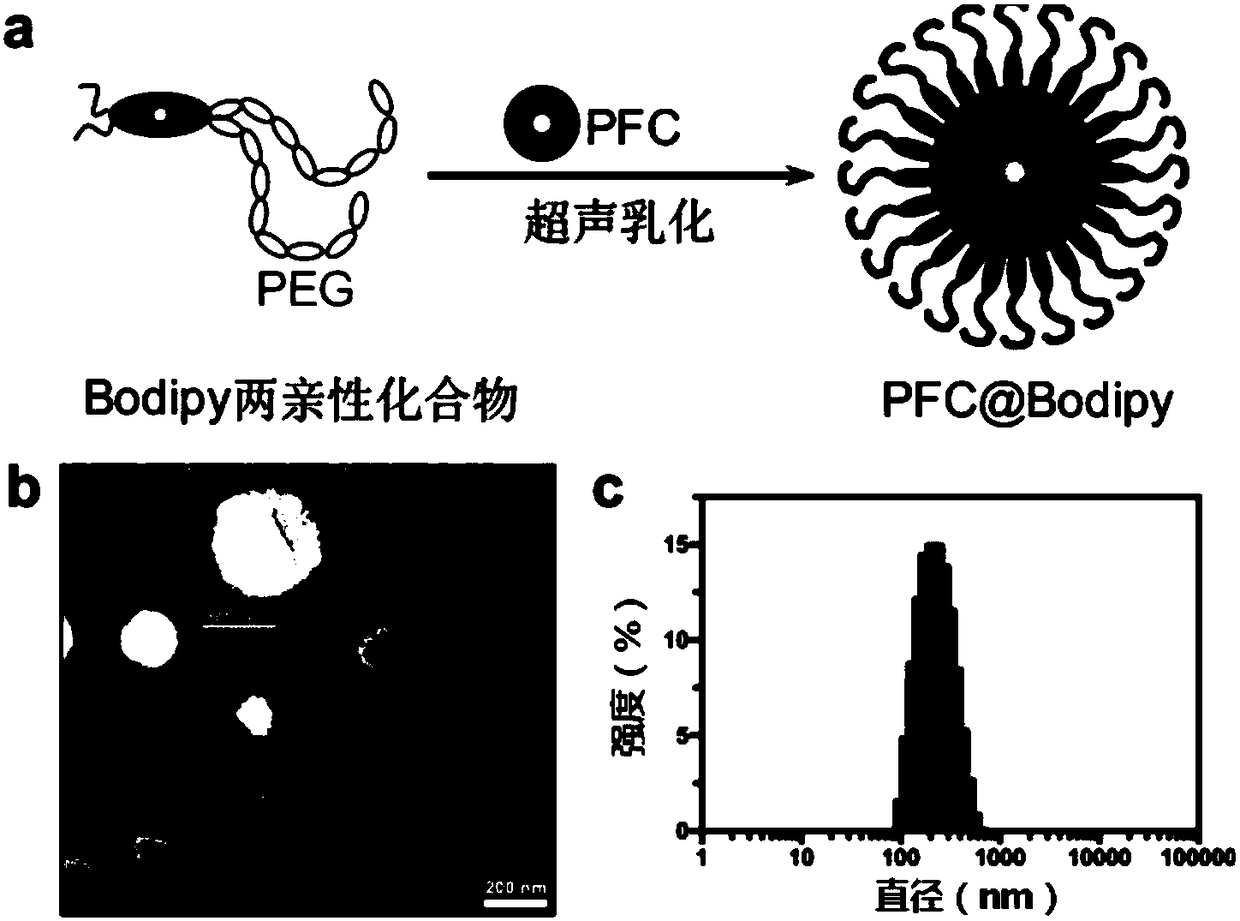
[0094] The preparation method of nanoemulsion (PFC@Bodipy) and the corresponding TEM image, particle size distribution such as image 3 shown.
[0095] image 3 (a) represents the roadmap for the preparation of nanoemulsions loaded with perfluorocarbons (PFC@Bodipy), where Bodipy represents bodipyr, and PFC represents perfluorocarbons; (b) and (c) represent nanoemulsions (PFC @Bodipy) TEM images and pa...
Embodiment 3
[0097] Performance evaluation of nanoemulsion photodynamic therapy in vitro
[0098] A375 cells were cultured in DMEM (10% fetal calf serum), and 1 × 10 per well 4 cells at a density of 96 wells. After 24 hours of incubation in the incubator, the cells were washed once with PBS. Cells were incubated with PFH@Bodipy and free Bodipy (0, 0.1, 0.25, 0.5, 2.5, 5 and 10 μM Bodipy) at 37°C under the same conditions for 12 hours. To evaluate cytotoxicity, the cells of each group were washed twice with PBS. A standard CCK-8 assay was performed to assess cell viability. Cells in each group were rinsed twice with PBS. Then use a power density of 200mW / cm 2 Irradiate the cells with a 660nm laser for 5 min. After irradiation, cells were incubated for an additional 24 hours. The control group was protected from light, and the other conditions were the same. Subsequently, a standard CCK-8 assay was performed to assess cell viability.
[0099] Figure 4 Indicates the effect of diffe...
PUM
| Property | Measurement | Unit |
|---|---|---|
| The average diameter | aaaaa | aaaaa |
| Number average molecular weight | aaaaa | aaaaa |
| Number average molecular weight | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com