Deep red photothermally activated delayed fluorescence material as well as preparation method thereof and electroluminescent devices
A technology of thermal activation delay and fluorescent materials, applied in the direction of luminescent materials, electric solid devices, chemical instruments and methods, etc., can solve the problem of inability to obtain device performance
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
preparation example Construction
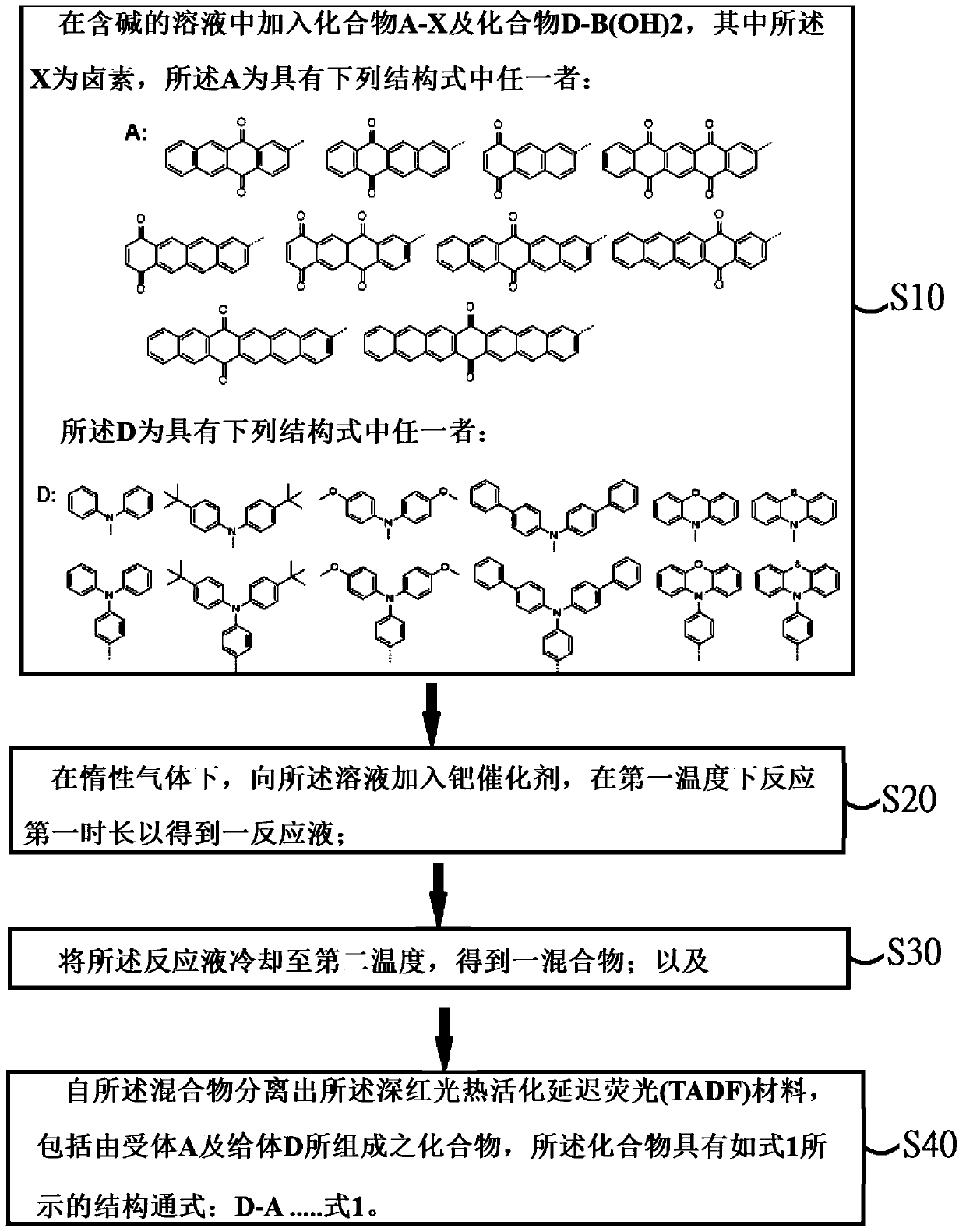
[0044] see figure 1 , figure 1 It is a flowchart of a preparation method of a deep red photothermally activated delayed fluorescence (TADF) material according to an embodiment of the present invention. Such as figure 1 As shown, the embodiment of the present invention also provides a preparation method of a deep red photothermally activated delayed fluorescence (TADF) material, comprising the following steps:
[0045] Step S10, adding compound A-X and compound D-B(OH)2 into the alkali-containing solution, wherein X is halogen, and A has any one of the following structural formulas:
[0046]
[0047] The D is any one of the following structural formulas:
[0048]
[0049] Step S20, under an inert gas, add a palladium catalyst to the solution, and react at a first temperature for a first time to obtain a reaction solution;
[0050] Step S30, cooling the reaction solution to a second temperature to obtain a mixture; and
[0051] Step S40, separating the deep red photot...
Embodiment 1
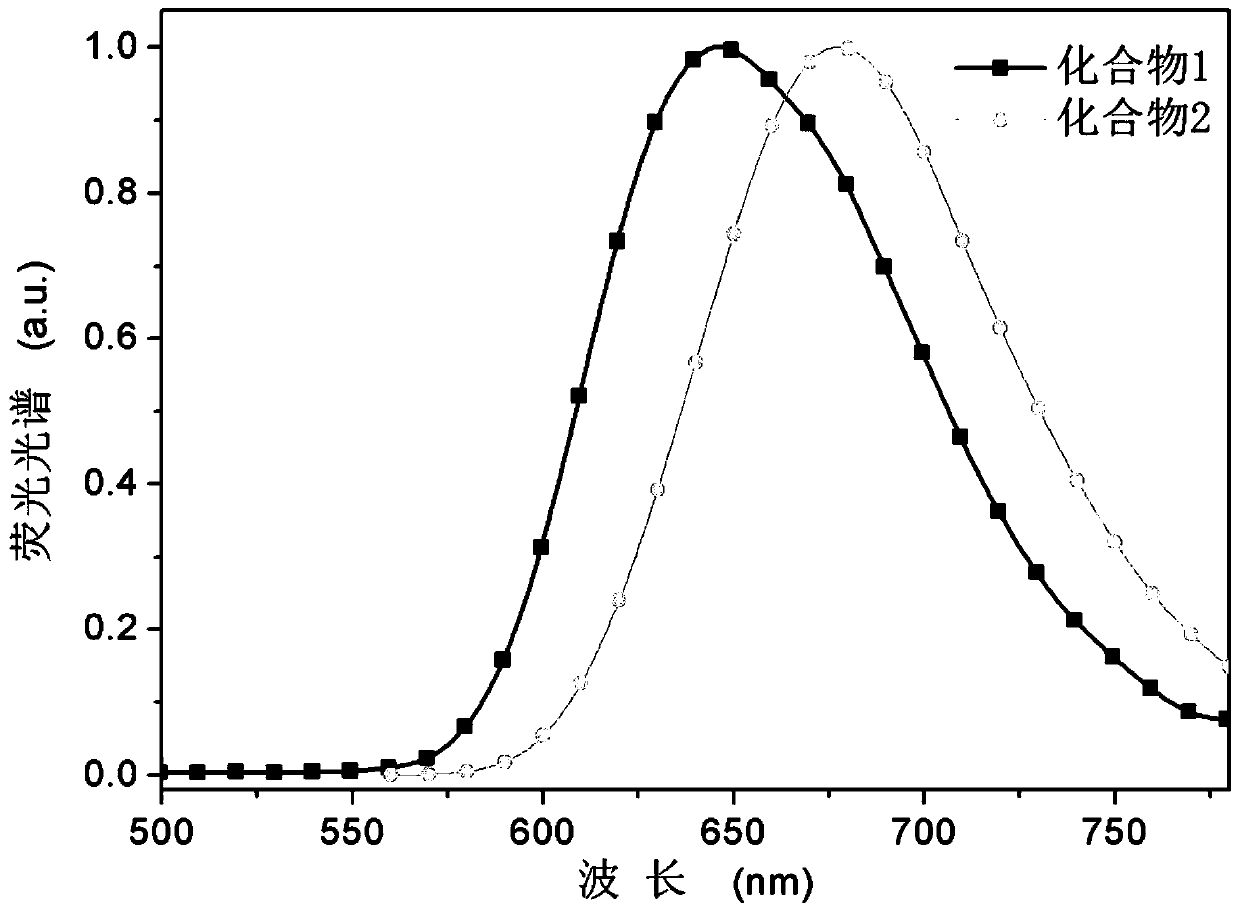
[0060] In specific example 1 of the present invention, the target deep red photothermally activated delayed fluorescence (TADF) material to be synthesized includes compound 1 of the following structural formula 2:
[0061]
[0062] The synthetic route of compound 1 of structural formula 2 is shown in following reaction formula 1:
[0063]
[0064] The detailed synthetic steps of compound 1 are as follows:
[0065] 2-Bromotetracene-5,12-dione (3.36 g, 10 mmol), 4-(diphenylamino)-phenylboronic acid (3.18 g, 11 mmol), 30 mL of toluene and 10 mL of 2.5 M aqueous potassium carbonate Added to a 100mL Schlenk (Schlenk) bottle, and evacuated with argon. Then tetrakistriphenylphosphopalladium (0.48g, 0.4mmol) was added, and the reaction was refluxed at 80°C for 24h. After cooling to room temperature, the reaction solution was extracted three times with DCM, washed three times with water, dried over anhydrous sodium sulfate, filtered, and spin-dried. Column chromatography was p...
Embodiment 2
[0067] In specific example 2 of the present invention, the target deep red photothermally activated delayed fluorescence (TADF) material to be synthesized includes compound 2 of the following structural formula 3:
[0068]
[0069] The synthetic route of compound 2 of structural formula 3 is shown in following reaction formula 2:
[0070]
[0071] The detailed synthetic steps of compound 2 are as follows:
[0072] 2-Bromopentacene-5,7,12,14-tetraketone (4.16g, 10mmol), 4-(diphenylamino)-phenylboronic acid (3.18g, 11mmol), 30mL of toluene and 10mL of 2.5 M Potassium carbonate aqueous solution was added to a 100 mL Schlenk bottle, and argon gas was used for gas exchange. Then tetrakistriphenylphosphopalladium (0.48g, 0.4mmol) was added, and the reaction was refluxed at 80°C for 24h. After cooling to room temperature, the reaction solution was extracted three times with DCM, washed three times with water, dried over anhydrous sodium sulfate, filtered, and spin-dried. Col...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Thickness | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com