Method for constructing HPLC (high performance liquid chromatography) fingerprint spectrum of medicinal leaves and twigs of rhododendron mariae
A technology of Rhododendron violaceum and fingerprint spectrum, which is applied in the field of medicine and achieves the effects of easy identification, high technical content and high similarity
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0028] Example 1: Detecting the fingerprints of different batches of Rhododendron azalea
[0029] 1 Instruments and reagents
[0030] 1.1 Agilent 1220 high performance liquid chromatograph (Agilent, USA); 124S electronic balance (Beijing Sartorius Balance Co., Ltd.).
[0031] 1.2 Purified water (Wahaha), acetonitrile (chromatographically pure), and other reagents are analytically pure. Rutin, quercetin, hyperoside, quercetin, kaempferol, zidonolic acid reference substance (batch number 100080-200306, 111538-200302, 111521-201205, 100081-200907, 110861-200304, 110709- 200304, China National Institutes for Food and Drug Control). Rhododendron violet (lot numbers: 181001, 181002, 181003, 181004, 181005, 181101, 181102, 181103, 181104, 181105).
[0032] 2 Determination of fingerprint
[0033] 2.1 Chromatographic conditions: Octadecylsilane bonded silica gel as filler, acetonitrile as mobile phase A, 0.1% phosphoric acid solution as mobile phase B, gradient elution, flow rate 1...
Embodiment 2
[0044] Example 2: Determination and data analysis of fingerprints of 13 batches of Rhododendron violet
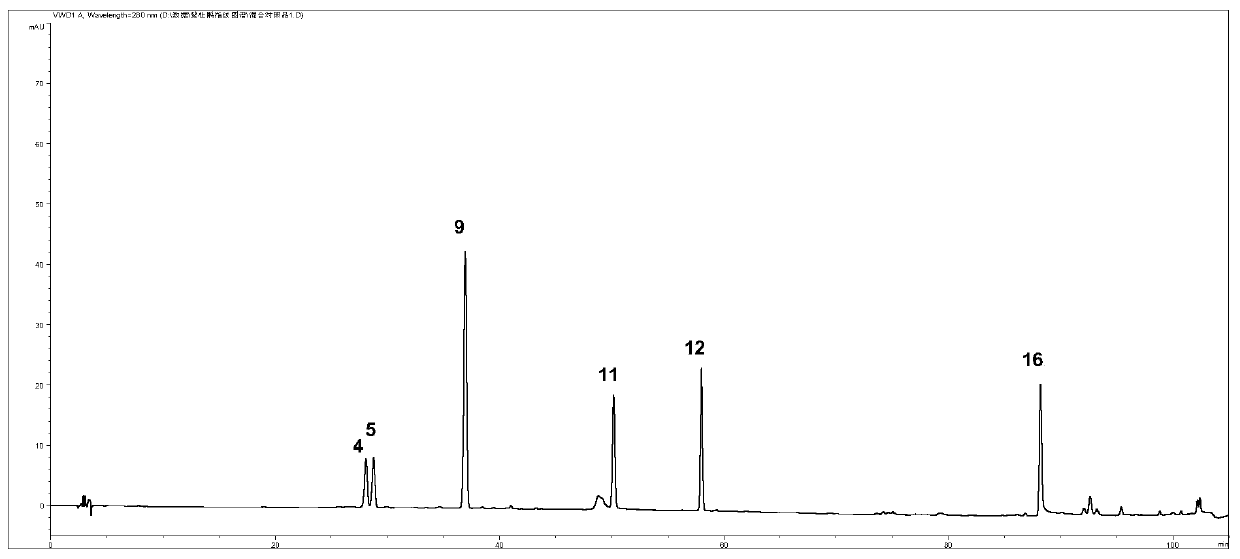
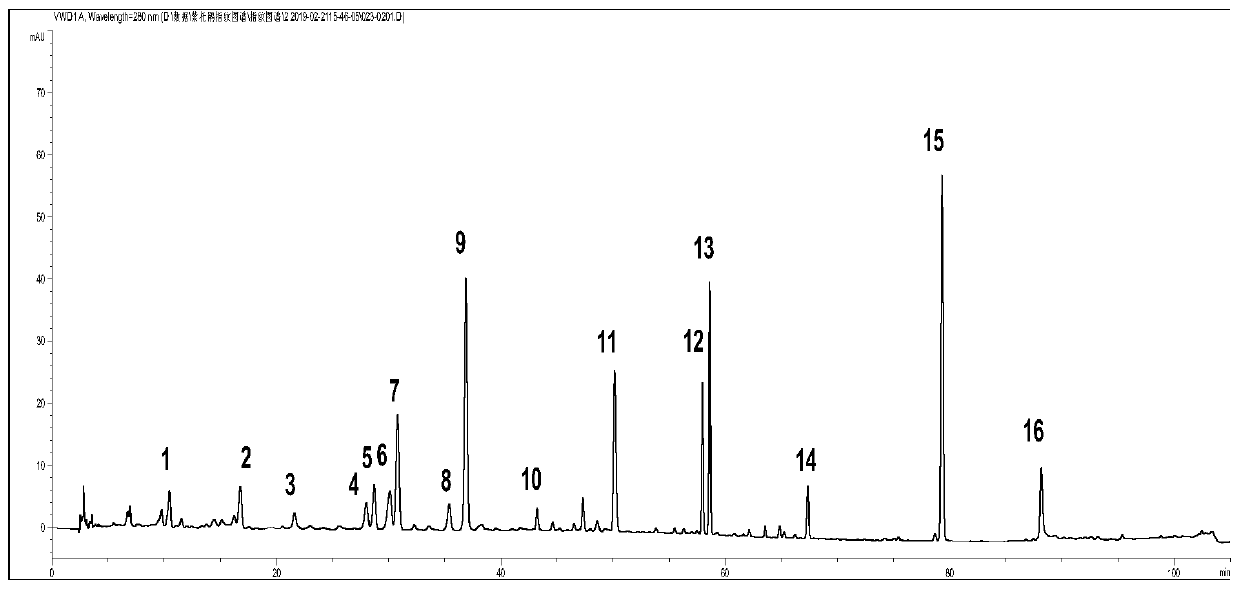
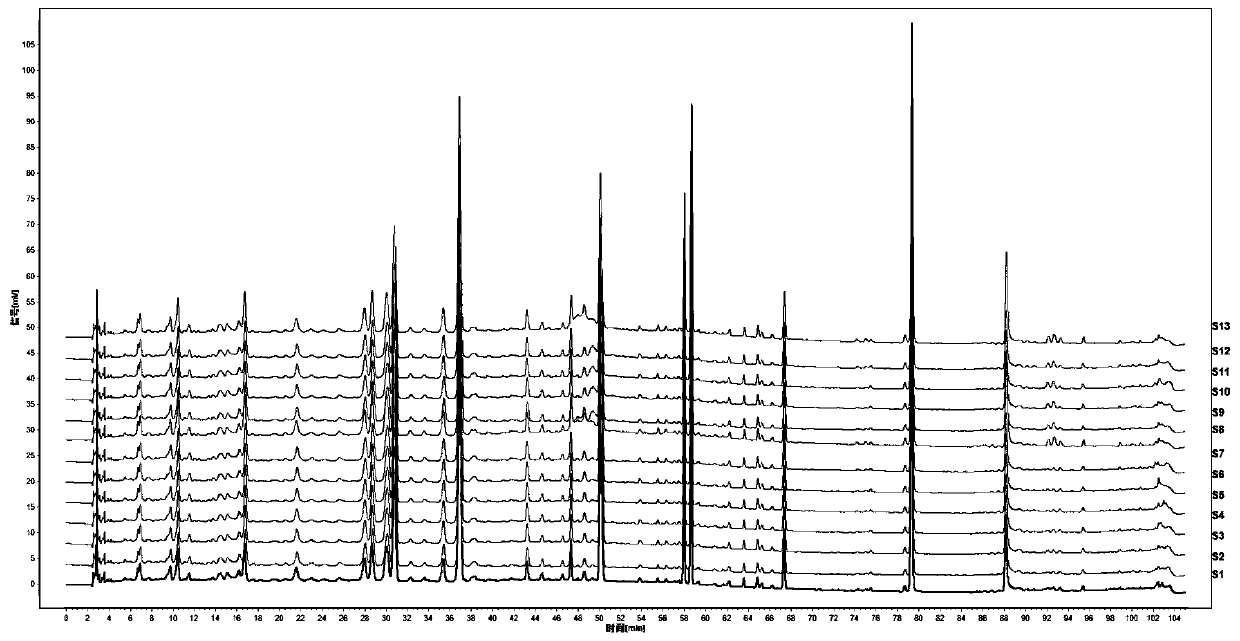
[0045] Get 13 batches of Rhododendron viola, detect according to the conditions of Example 1, and obtain the HPLC spectra of 13 batches of samples. Such as image 3 .
[0046] Through the comparison of 13 batches of PHLC spectra, the similarity evaluation was carried out to determine its characteristic common peaks: import the chromatograms of 10 batches of Rhododendron violet into the "Chinese Medicine Chromatographic Fingerprint Similarity Evaluation System" 2004A Edition (National Pharmacopoeia Commission), and use the common peaks mode, set the time window width to 0.1min, and generated standard fingerprints by the average method, and determined 16 common peaks. The similarities of the 13 batches of Rhododendron viola were all greater than 0.9, as shown in Table 2. The relative retention times of the 16 common peaks were basically the same, but there were large diff...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More