Synthesis and preparation method of phenobarbital-d5 for qualitative and quantitative use in forensic science
A phenobarbital and scientific technology, applied in the field of preparation of a D5-deuterated phenobarbital standard in the field of forensic science, can solve the problem of poor stability of the deuterated internal standard, dependence on imports, restrictions on widespread use, etc. problems, to achieve the effect of eliminating hidden safety hazards, good quality and low cost
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
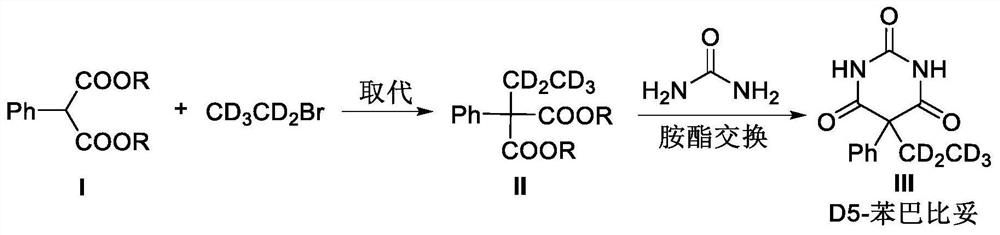
[0032] (1) Dimethyl 2-phenylmalonate (10.00mmol, 2.08g) was placed in a 50mL round bottom flask, the solvent MeOH (30mL) was added, and MeONa (15.00mmol, 0.81g) was added, and stirred at room temperature for 1h . Then deuterated bromoethane (14.00 mmol, 1.60 g) was added, stirred at room temperature for 5 h, and the reaction was monitored by TLC until dimethyl 2-phenylmalonate disappeared.
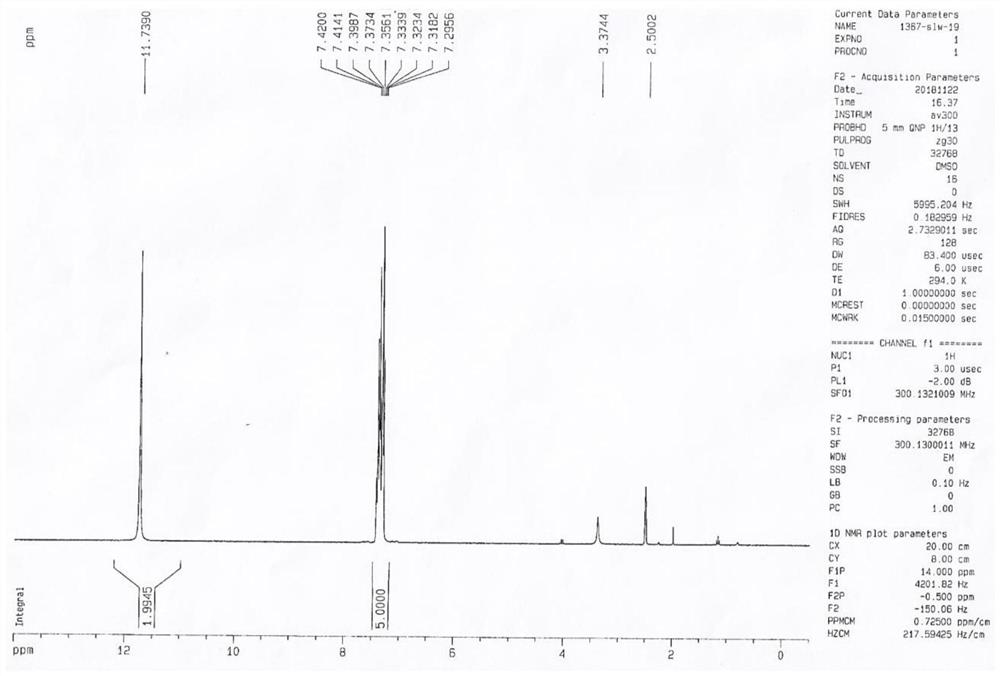
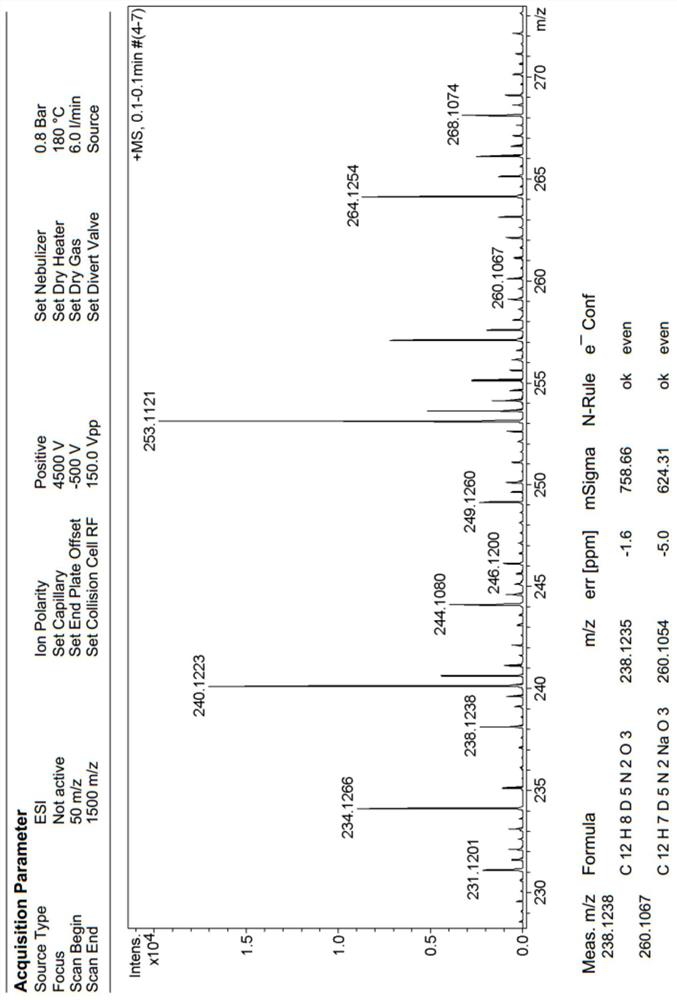
[0033] (2) Continue to add MeONa (25.00mmol, 1.35g) and urea (16mmol, 0.96g) to the reaction system, heat the reaction mixture to reflux rapidly, stir for 1h, recover methanol by distillation under reduced pressure, and add 30mL of urea to the residue Stir the water evenly, add hydrochloric acid (concentration: 6mol / L) to adjust the pH to 2. Then extract three times with ethyl acetate, extract with 30mL ethyl acetate each time, combine the organic phases and dry with anhydrous sodium sulfate (40g), after filtering, concentrate under reduced pressure, column chromatography purification, el...
Embodiment 2
[0035](1) Diethyl 2-phenylmalonate (100.00mmol, 23.6g) was placed in a 500mL round bottom flask, the solvent EtOH (300mL) was added, the temperature was cooled to 0°C in an ice bath, and then EtONa (140.0mmol, 9.5g), stirred for 1h. The reaction system was returned to room temperature, deuterated bromoethane (140.0 mmol, 16.0 g) was added, stirred at room temperature for 5 h, and the reaction was monitored by TLC until the disappearance of diethyl 2-phenylmalonate.
[0036] (2) Continue to add EtONa (250.0mmol, 17.0g) and urea (140mmol, 8.4g) to the reaction system, heat the reaction mixture to reflux rapidly, stir for 1h, reclaim ethanol by distillation under reduced pressure, and add 300mL of urea to the residue Stir the water evenly, add hydrochloric acid (concentration: 6mol / L) to adjust the pH to 2. Then it was extracted three times with ethyl acetate, each extraction was extracted with 300mL ethyl acetate, the combined organic phases were dried with anhydrous sodium sul...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More