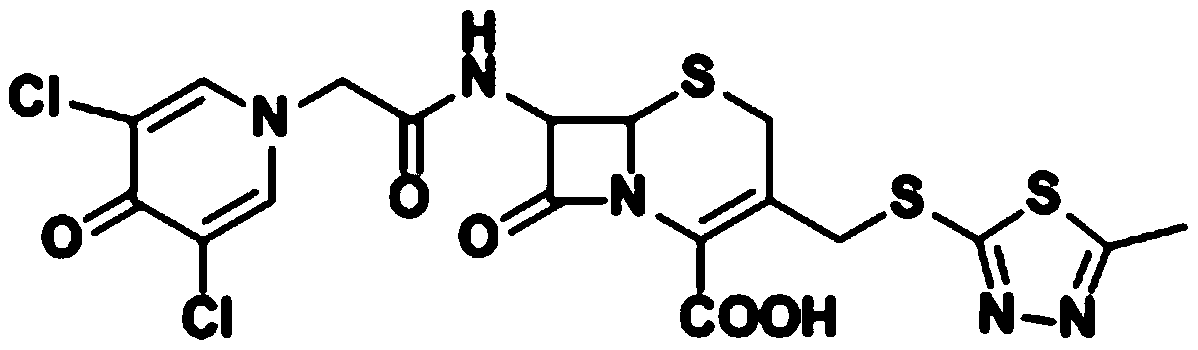
Cefazedone synthesis method
A synthesis method and a technology for cefoxidone, which are applied in the field of drug synthesis, can solve the problems of affecting the total yield and product quality of the product, being difficult to remove, and having many side reactions, so as to improve the total yield and purity of the product, the route is simple, and the product high yield effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
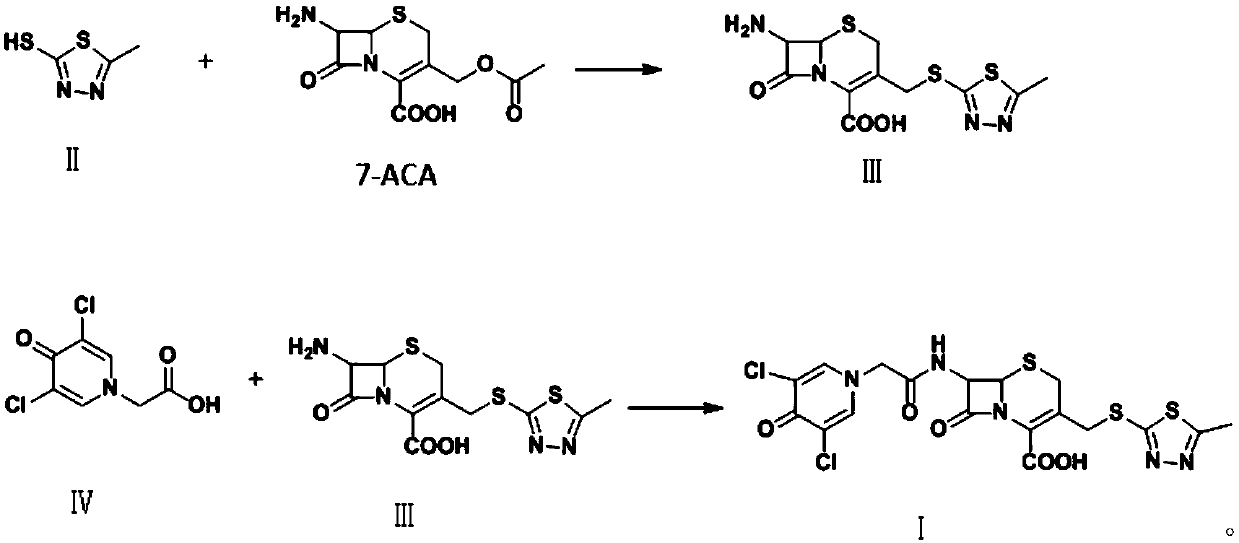
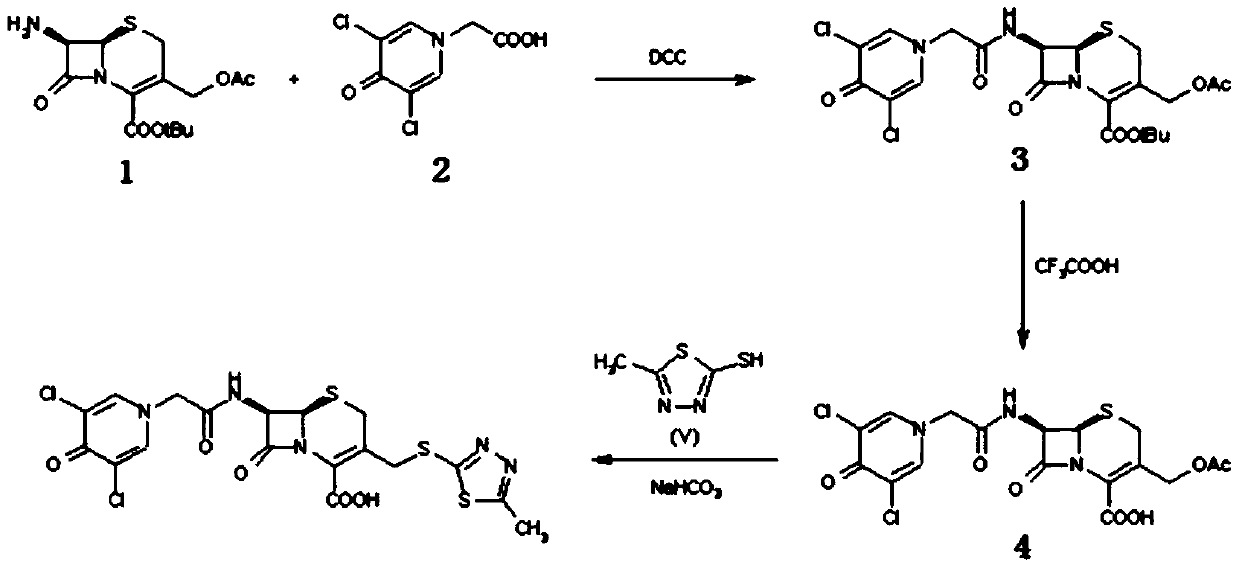
[0025] Preparation of compound Ⅲ
[0026] Dissolve boron trifluoride-dimethyl carbonate complex (2mol) in 150ml of dimethyl carbonate, then add 132.25g (1mol) of compound II, stir and dissolve at room temperature, and control the temperature at 0-5°C. Add formic acid (1mol), then slowly add 272.30g (1mol) of 7-ACA dropwise, control the reaction at 0-5°C for 1-1.5h, transfer to water, adjust the pH to 3.8 with ammonia water, precipitate crystals, grow crystals for 1h, It was filtered, washed with purified water, and vacuum-dried to obtain 20.32 g of compound III, with a molar yield of 93%, a purity of 99.3%, and a maximum of 0.07%.
Embodiment 2
[0028] Preparation of compound Ⅲ
[0029] Dissolve boron trifluoride-dimethyl carbonate complex (2.5mol) in 150ml of dimethyl carbonate, then add 132.26g (1mol) of compound II, stir and dissolve at room temperature, and control the temperature at 5-10°C , add glacial acetic acid (1mol), then slowly add 7-ACA 299.51g (1.1mol) dropwise, control the reaction at 5-10°C for 1-1.5h, transfer to water, adjust the pH to 3.8 with ammonia water, precipitate crystals, and raise After 1 hour of crystallization, filtered, washed with purified water, and vacuum-dried to obtain 327.21 g of compound III, the molar yield was 95%, the purity was 99.4%, and the maximum monoheterogeneity was 0.08%.
Embodiment 3
[0031] Preparation of compound Ⅲ
[0032] Dissolve boron trifluoride-dimethyl carbonate complex (2mol) in 150ml of dimethyl carbonate, then add 132.26g (1mol) of compound II, stir and dissolve at room temperature, and control the temperature at 10-15°C. Add formic acid (1mol), then slowly add 272.29g (1mol) of 7-ACA dropwise, control the reaction at 10-15°C for 1-1.5h, transfer to water, adjust the pH to 3.8 with ammonia water, precipitate crystals, grow crystals for 1h, It was filtered, washed with purified water, and vacuum-dried to obtain 296.21 g of compound III, with a molar yield of 86%, a purity of 98.7%, and a maximum of 0.23%.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com