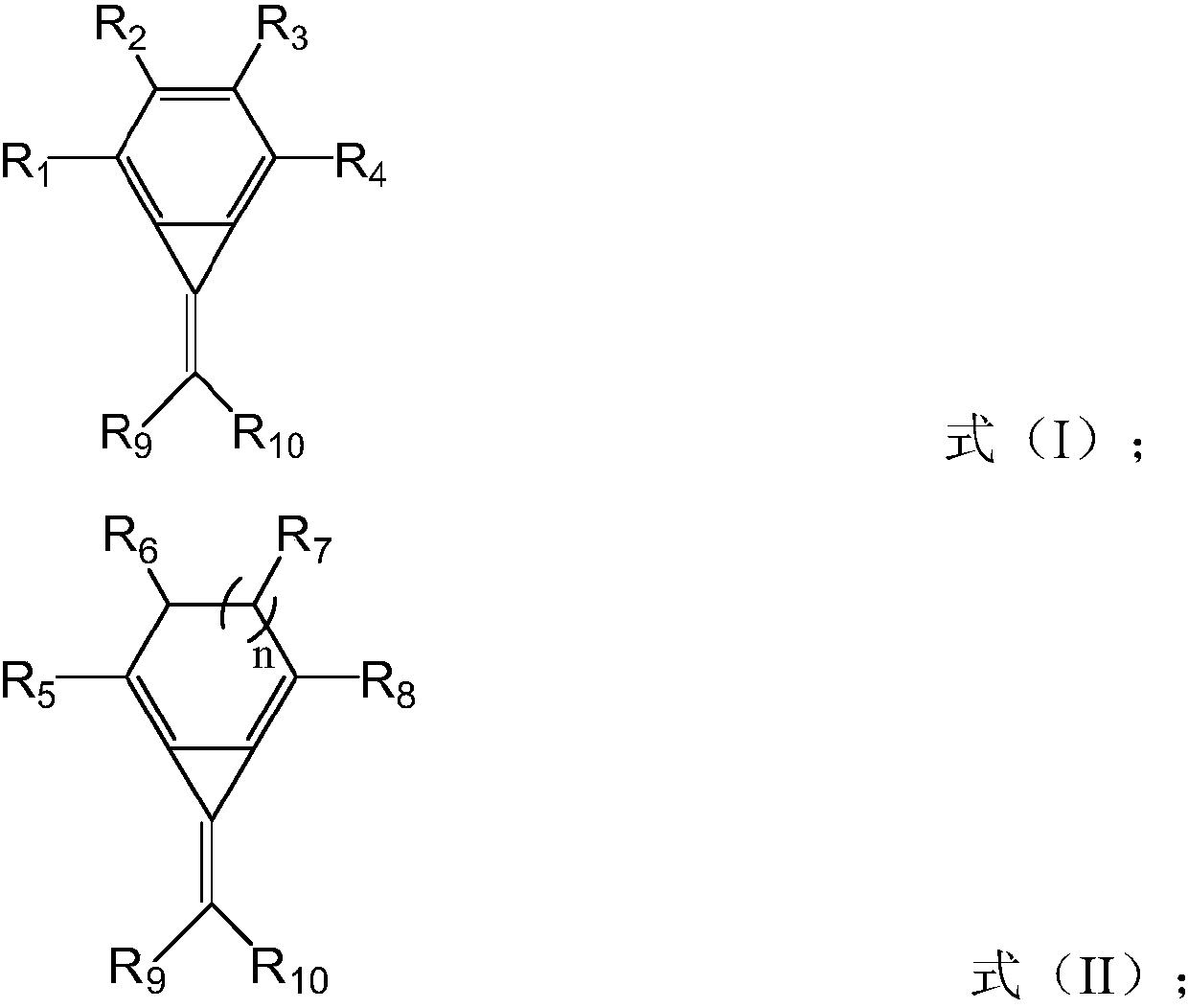
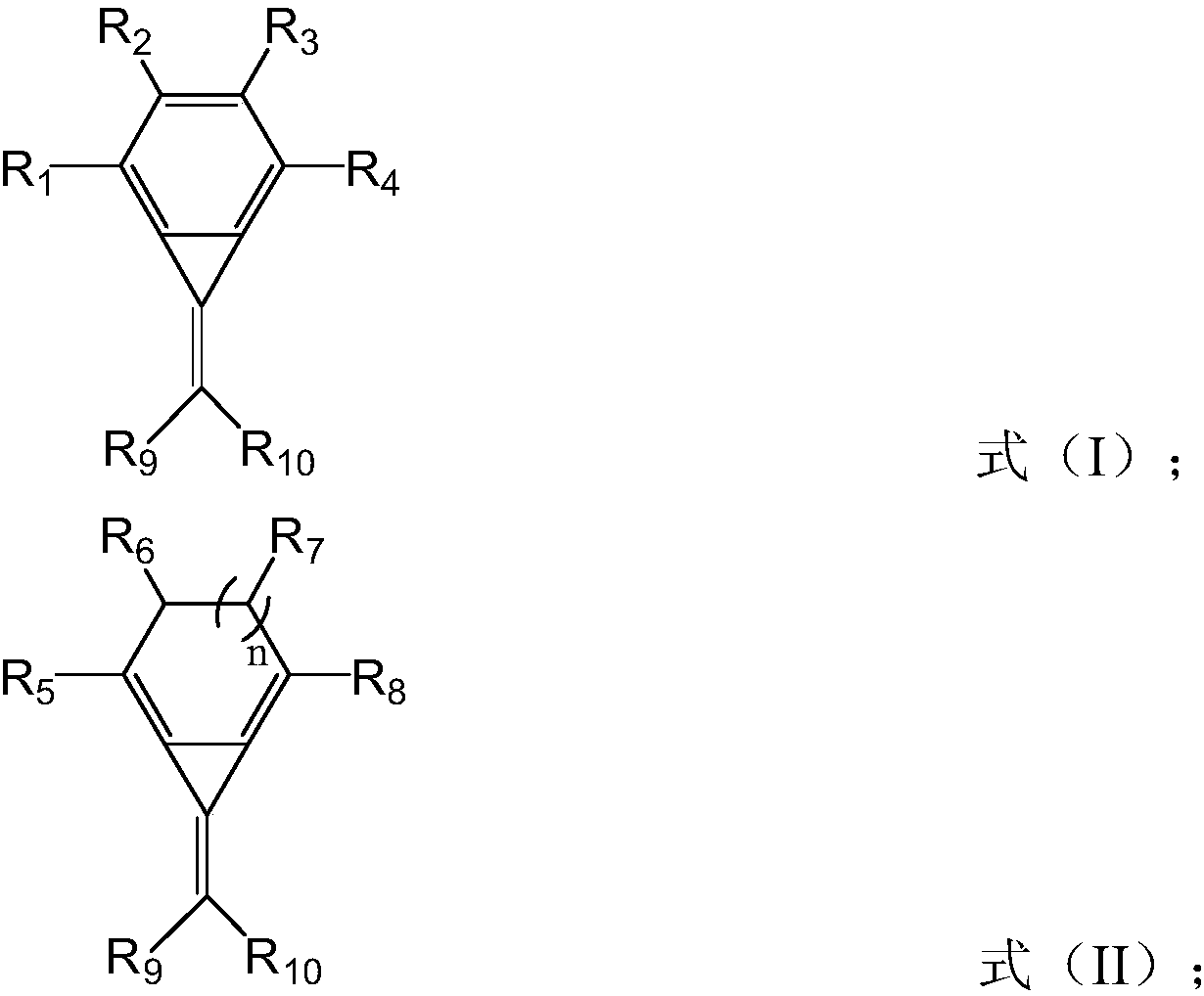
Hole injection material, hole injection layer comprising same, and OLED display panel
A technology of hole injection material and hole injection layer, which is applied in chemical instruments and methods, preparation of organic compounds, electrical components, etc., can solve problems such as hole injection performance needs to be improved, compound reduction potential is weak, etc., and achieve strong interaction effect, increase electrical conductivity, and good thermal stability
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
preparation example 1
[0064] a compound A preparation method comprising the steps of:
[0065]
[0066] Potassium tert-butoxide (112.21mg, 1mmol), a-1 (306.09mg, 1mmol) and 10mLTHF were added to the reaction flask, and a-2 was generated after stirring at room temperature for 6 hours, and the product was directly used in the next step without purification Then add dioxomalononitrile (80mg, 1mmol) and stir for 1 hour, then add potassium fluoride (58.10mg, 1mmol) and tetrabutyllithium fluoride (261.46mg, 1mmol) and continue to stir for 1 hour; Reaction solution Concentrate and column to give a-3.
[0067] Characterization:
[0068] NMR: 13 H NMR: (1H, 73.1); (2H, 117.2); (2H, 118.4); (2H, 136.8); (2H, 143.9); (1H, 176.7);
[0069] Mass spectrum: 224 (molecular weight)
[0070] The LUMO energy level of the compound of Preparation Example 1 was 5.38 eV.
preparation example 2
[0072] a compound
[0073]
[0074] A preparation method comprising the steps of:
[0075]
[0076] Potassium tert-butoxide (112.21mg, 1mmol), a-1 (306.09mg, 1mmol) and 10mLTHF were added to the reaction flask, and a-2 was generated after stirring at room temperature for 6 hours, and the product was directly entered into the next step without purification . Add oxo(2,3,5,6-tetrafluoro-4-trifluoromethyl-phenyl)propionitrile (270.99 mg, 1 mmol) and stir for 1 hour, followed by potassium fluoride (58.10 mg, 1 mmol) and Tetrabutyllithium fluoride (261.46 mg, 1 mmol) was stirred for an additional 1 hour. The reaction solution was concentrated and passed through a column to obtain a-3.
[0077] Characterization:
[0078] NMR: 13 H NMR: (1H, 97.7); (1H, 102.7); (1H, 107.4); (1H, 115.4); (1H, 117.2); (1H, 118.4); (2H, 136.8); (1H, 97.7) ;(2H,118.4);(2H,143.0);(2H,143.9);(2H,144.3),(1H,151.5);
[0079] Mass spectrum: 414.99 (molecular weight).
[0080] The LUMO energy lev...
preparation example 3
[0082] a compound
[0083]
[0084] A preparation method comprising the steps of:
[0085]
[0086] Add c-1 (229.93mg, 1mmol) and 10mL cyclohexane to the reaction flask, then add n-butyllithium (32.03mg, 0.5mmol) dropwise to the reaction system to obtain intermediate c-2, and finally the reaction system continues Stir for 6 hours. The reaction solution was concentrated and passed through a column to obtain c-3.
[0087] Characterization:
[0088] NMR: 13 H NMR: (4H, 118.4); (2H, 132.3); (4H, 136.8); (4H, 143.9);
[0089] Mass spectrum: 319.99 (molecular weight).
[0090] The LUMO energy level of the compound of Preparation Example 3 was 5.20 eV.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com