Preparation method of axitinib intermediate
A technology of axitinib and intermediates, applied in the field of pharmaceutical synthesis, can solve the problems of high production cost, low purity and high technical requirements, and achieve the effects of mild reaction conditions, high reaction yield and high product purity
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
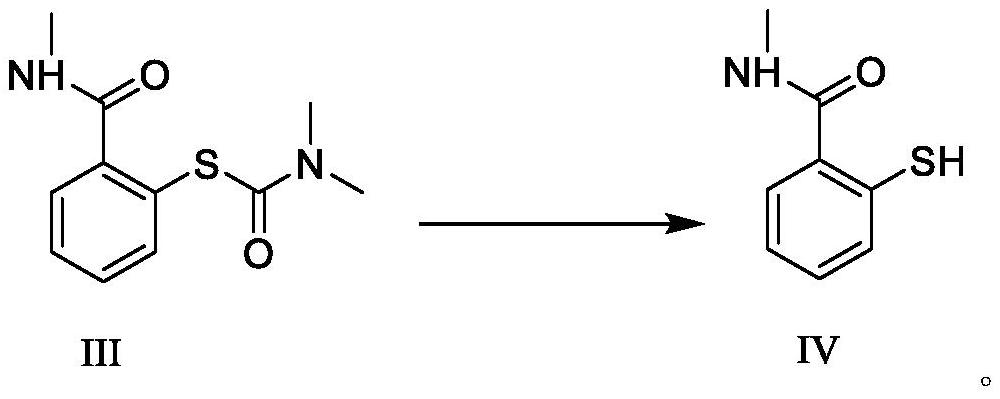
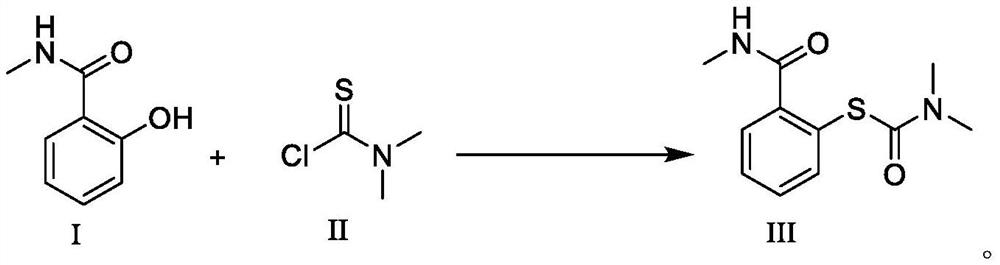
[0039] Dissolve 30.23g (0.2mol) of 2-hydroxy-N-methylbenzamide in tetrahydrofuran (50ml), slowly add 29.66g (0.24mol) of dimethylaminothioformyl chloride and 31.39g (0.28mol) of triethylene Diamine, temperature control 40 ℃ reaction, after TLC detects reaction, add ethyl acetate and purified water (V 乙酸乙酯 :V 纯化水 =3: 1) 40ml, the organic layer was concentrated under reduced pressure to obtain 2-mercapto-N-methylbenzamide with a yield of 97.2% and a purity of 99.95% by HPLC.
Embodiment 2
[0041] Dissolve 30.23g (0.2mol) of 2-hydroxy-N-methylbenzamide in acetonitrile (50ml), slowly add 27.20g (0.22mol) of dimethylaminothioformyl chloride and 36.19g (0.28mol) of N, N-diisopropylethylamine, warming up to 40 ° C, after the TLC detection reaction is completed, the reaction solution is down to room temperature, ethyl acetate and purified water (V 乙酸乙酯 :V 纯化水 =3:1) 40ml, the organic layer was concentrated under reduced pressure to obtain 2-mercapto-N-methylbenzamide with a yield of 94.4% and a purity of 99.88% by HPLC.
Embodiment 3
[0043] Dissolve 30.23g (0.2mol) of 2-hydroxy-N-methylbenzamide in N,N-dimethylformamide (40ml), slowly add 24.72g (0.2mol) of dimethylaminothioformyl chloride and 34.21g (0.28mol) of 4-dimethylaminopyridine was heated up to 60°C for reaction. After the reaction was detected by TLC, the reaction solution was cooled to room temperature, and ethyl acetate and purified water (V 乙酸乙酯 :V 纯化水=3:1) 40ml, the organic layer was concentrated under reduced pressure to obtain 2-mercapto-N-methylbenzamide with a yield of 92.3% and a purity of 99.90% by HPLC.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More