Modified ADAMTS4 molecules and method of use thereof
a technology of aggrecanase and adamts4, which is applied in the field of modified aggrecanases, can solve the problems of hampered research effects on adamts4 and achieve the effects of facilitating the production of anti-adamts antibodies, reducing binding affinity, and improving stability
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Cloning and Purification of Full-Length Human ADAMTS4
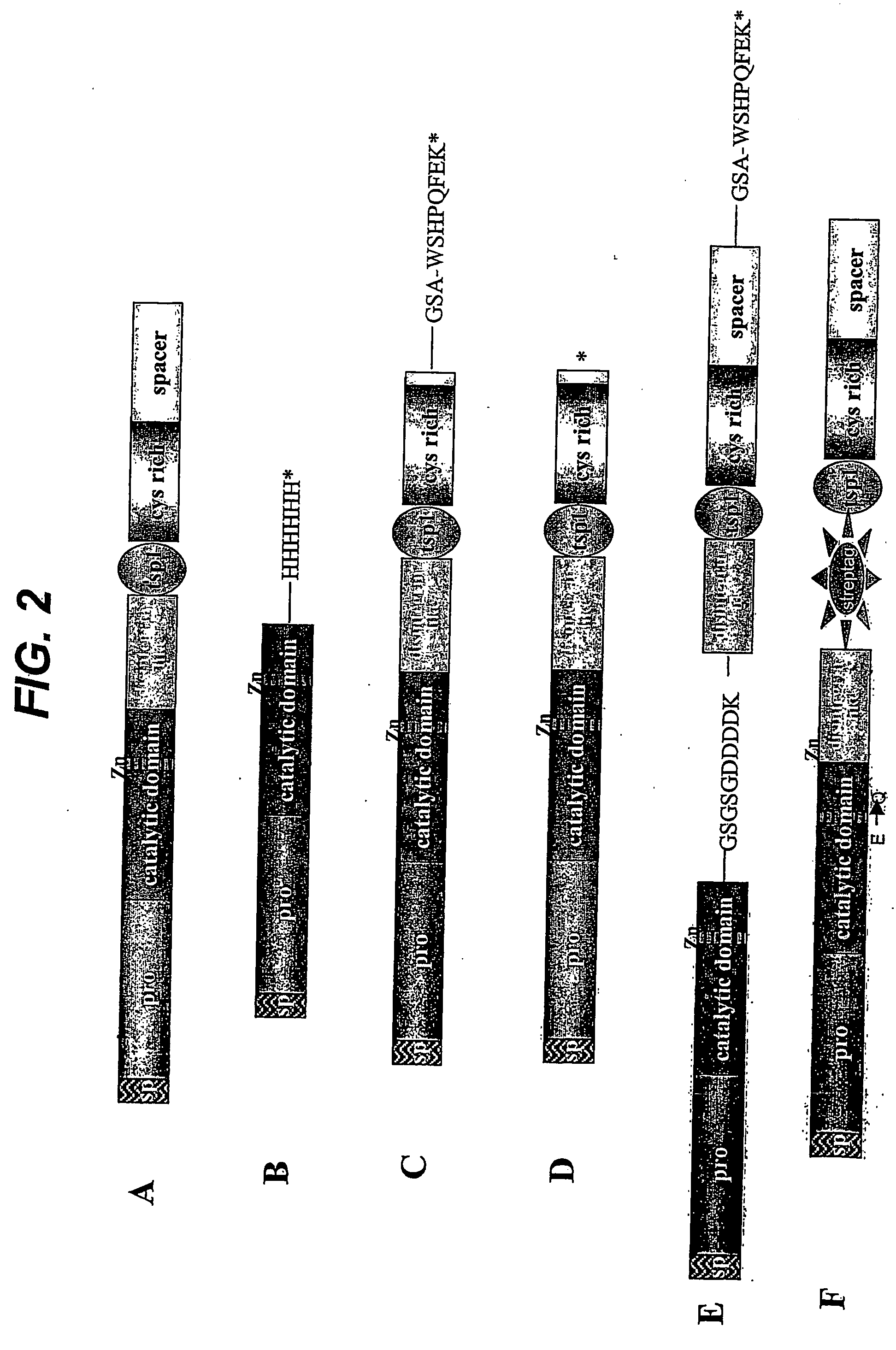
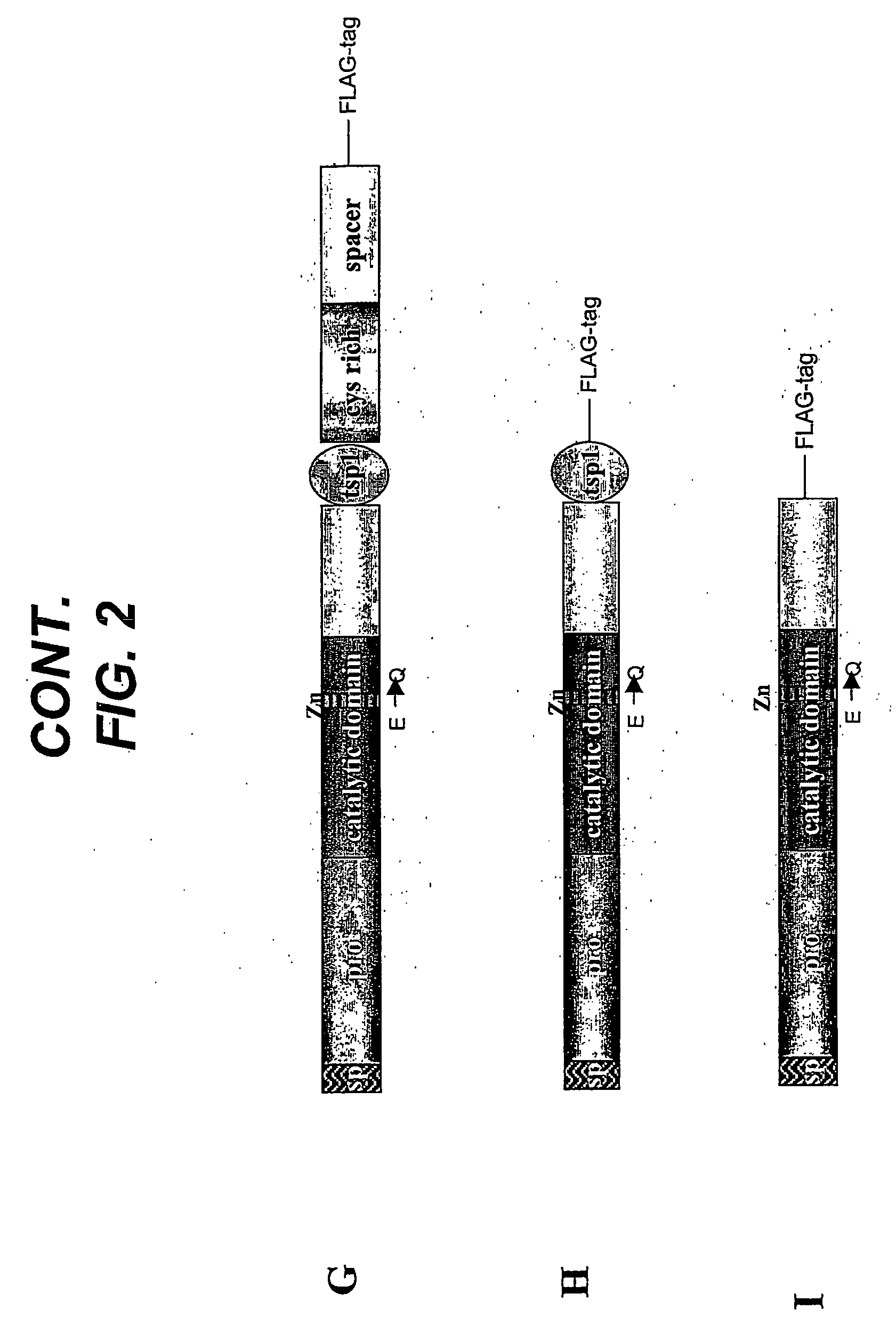
[0118] Human ADAMTS4 cDNA was cloned using a PCR strategy. Two sets of oligonucleotide primers were designed to amplify overlapping portions of the 5′- and 3′-halves of the cDNA. Of the seven human multiple-tissue cDNA libraries that were used as PCR templates, only the uterus cDNA library resulted in PCR products of the appropriate size (5′-amplimer of 1294bp (SEQ ID NO:2) and 3′-amplimer of 1421bp (SEQ ID NO:3)). PCR-amplified fragments were digested with EcoRI and BamHI (5′-product) or BamHI and NotI (3′-product), ligated into EcoRI- and NotI-digested COS expression vector pED6-dpc2, and transformed into ElectroMAX DH10B cells (Invitrogen). Cloned PCR fragments of ADAMTS4 were sequenced and found to have three silent changes as compared with the published nucleotide sequence for ADAMTS4 cDNA (SEQ ID NO:4) (Tortorella et al., Science 284:1664-1666, 1999). These changes were C to T at base pair 466, A to G at base pair 2131, and...
example 2
Generation of Truncated ADAMTS4 Molecules by Auto-Digestion
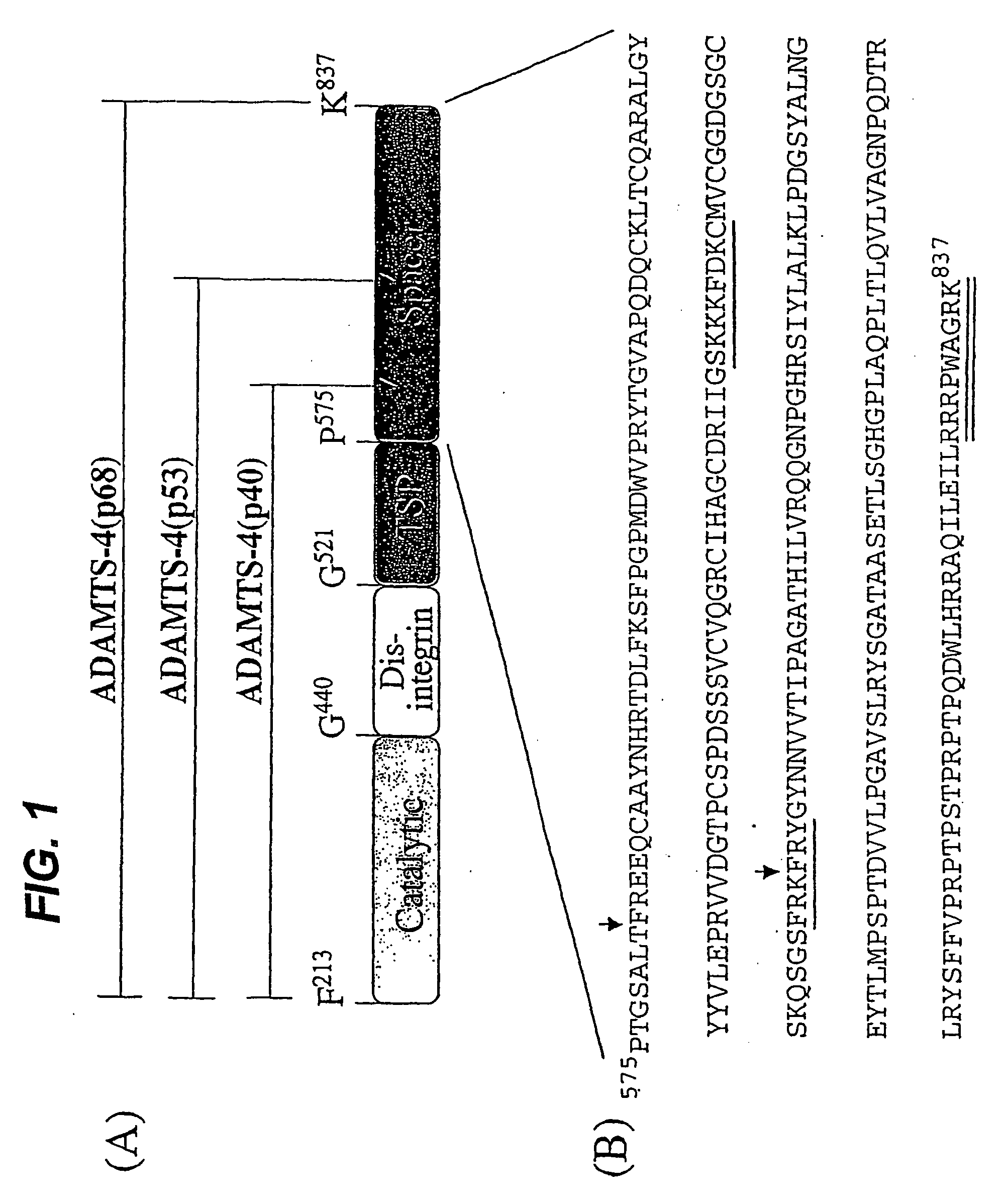
[0121] Purified recombinant human ADAMTS4 migrated on SDS-PAGE gels predominantly as a 68 kD band, together with a small amount (2 and 0.1-1.0M NaCl. Auto-digested products were visualized by Coomassie blue staining, by silver staining, or by Western immunoblot analysis with the L9026 antibody.
[0122] Following incubation at 37° C. for various times up to 16 h, ADAMTS4 was detected as isoforms of 68 kD (ADAMTS4(p68)), 53 kDa (ADAMTS4(p53)) and 40 kD (ADAMTS4(p40)). Results from incubations performed using ADAMTS4 at concentrations ranging from 10 pg / ml to 569 pg / ml, and at salt concentrations up to 1.0M, were essentially identical. Incubation of ADAMTS4 ASM under the same condition resulted in no detectable isoforms, thus confirming that the processing of ADAMTS4 was autocatalytic (Flannery et al., J. Biol. Chem. 277:42775-42780).
example 3
Amino Acid Sequencing and Mass Spectrometry Analyses of Auto-Digested ADAMTS4 Isoforms
[0123] For N-terminal sequence analysis, aliquots of auto-digested ADAMTS4 isoforms were separated on 10% Bis-Tris NuPage SDS-PAGE gels and transferred to PVDF membranes which were stained with Coomassie blue. Excised bands corresponding to ADAMTS4(p68), ADAMTS4(p53) and ADAMTS4(p40) were subjected to automated sequencing on a PE-Biosystems 491A Pulsed-Liquid Sequencer on-line with a PE-Biosystems 140S PTH Analyzer (Procise-HT).
[0124] For C-terminal sequence analysis, auto-digested ADAMTS4 isoforms were separated by fractionation on a column of Poros HQ. Unbound ADAMTS4(p53) and ADAMTS4(p40) were subsequently fractionated on a column of Poros HS eluted using an isocratic gradient of 0.05-1.0 M NaCl in 25mM HEPES, pH 6.8, 5 mM CaCl2 and 5 pM ZnCl2. Mass spectrometry analyses were performed using a Micromass LCT (LC-TOF-MS) analyzer (Micromass UK, Ltd, Manchester, U.K.). Samples were concentrated a...
PUM
| Property | Measurement | Unit |
|---|---|---|
| concentrations | aaaaa | aaaaa |
| pH | aaaaa | aaaaa |
| pH | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More