Combinatorial protein library screening by periplasmic expression
a technology of periplasmic expression and protein library, applied in the field of protein engineering, can solve the problems of difficult preparation of clones isolated after several rounds of panning, difficult screening of phage-displayed libraries, complex screening of large libraries consisting of tens of millions or billions of clones, etc., and achieve the effect of increasing the permeability of the outer membran
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Demonstration of Anchored Periplasmic Expression to Target Small Molecules and Peptides
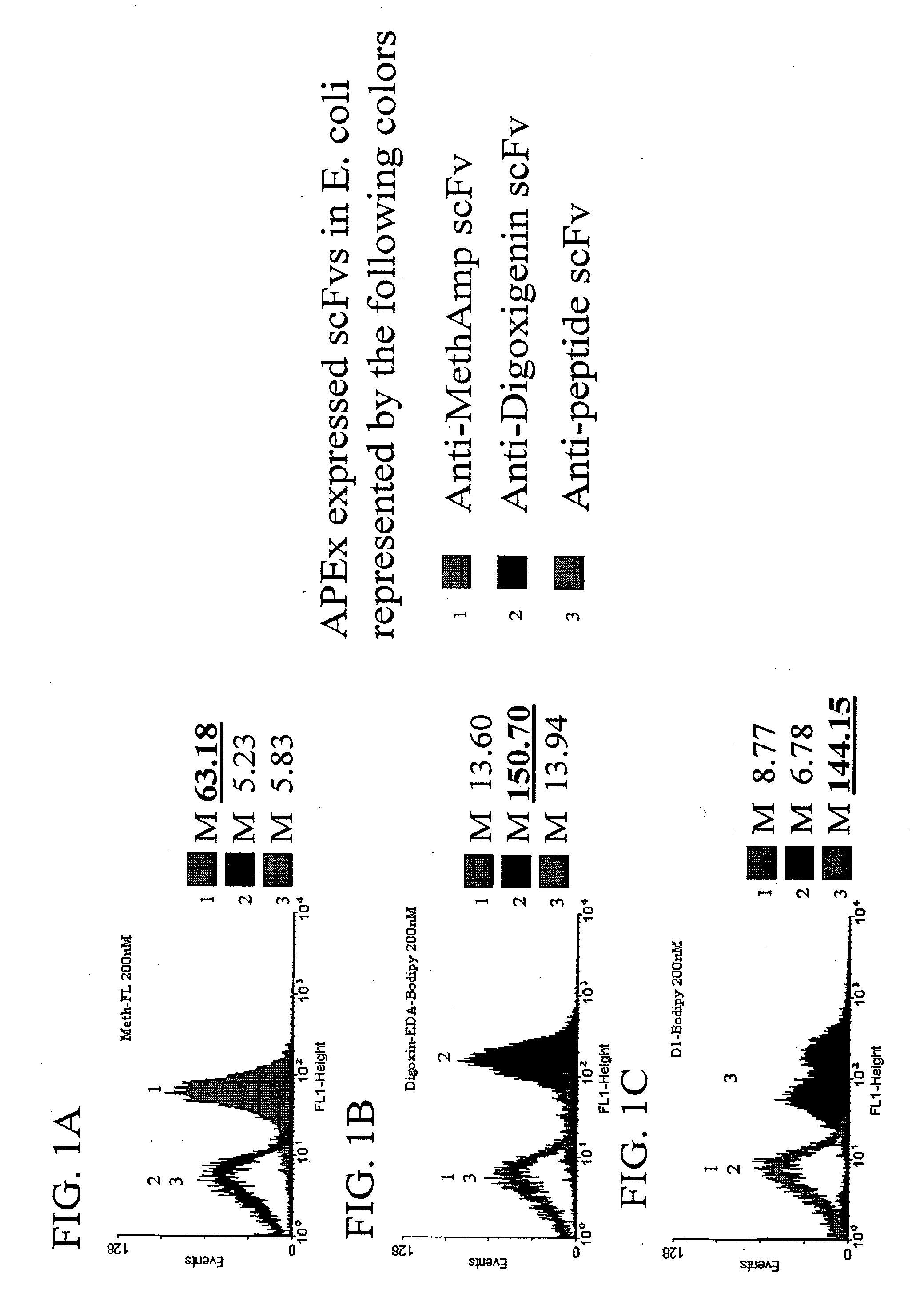
[0151] The ability of scFvs displayed by APEx to target small molecules and peptides is shown in FIGS. 1A-1B and in FIG. 1C, respectively. Three cultures of Escherichia coli containing fusions of the first six amino acids of NlpA (to serve as a inner membrane targeting sequence for APEx analysis) to either an anti-methamphetamine, anti-digoxin, or anti-peptide scfv were grown up and induced for protein expression as described below. Cells of each construct were then labeled in 5×PBS buffer with 200 nM concentrations of methamphetamine-FL (FIG. 1A), digoxigenin-bodipy (FIG. 1B), or 200 nM peptide(18mer)-BodipyFL (FIG. 1C). The data presented shows a histogram representation of 10,000 events from each of the labeled cell cultures. The results demonstrate the ability of scfvs displayed by APEx to bind to their specific antigen conjugated fluorophore, with minimal crossreactivity to non-specific liga...
example 2
Demonstration of Recognition of Ab Fragments by Anchored Periplasmic Expression
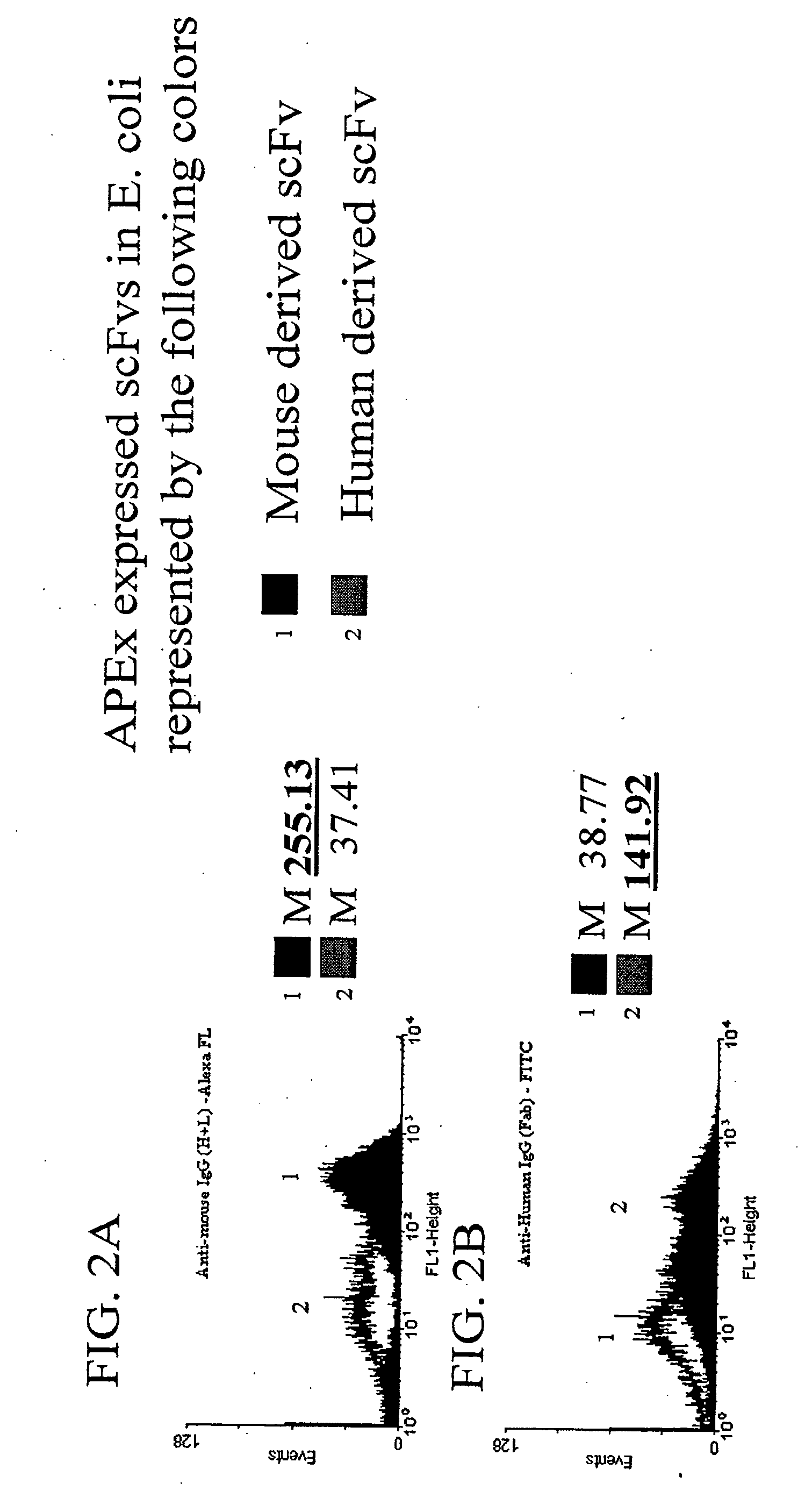
[0152] To demonstrate that the scFv is accessible to larger proteins, it was first demonstrated that polyclonal antibody serum against human Ab fragments or mouse Ab fragments would recognize scFvs derived from each displayed on the E. coli inner membrane by anchored periplasmic expression. Escherichia coli expressing a mouse derived scFv via anchored periplasmic expression (FIG. 2A) or expressing a human derived scFv via anchored periplasmic expression (FIG. 2B) were labeled as described below with either anti-mouse polyclonal IgG (H+L)-Alexa-FL or anti-human polyclonal IgG (Fab)-FITC. Results (FIG. 2A, 2B) in the form of histogram representations of 10000 events of each demonstrated that the anti-human polyclonal (approximately 150 kDa in size) recognized the human derived scFv specifically while the anti-mouse polyclonal (150 kDa) recognized the mouse derived scFv.
example 3
Demonstration of the Ability of scFvs Displayed by Anchored Periplasmic Expression to Specifically Bind Large Antigen Conjugated Fluorophores
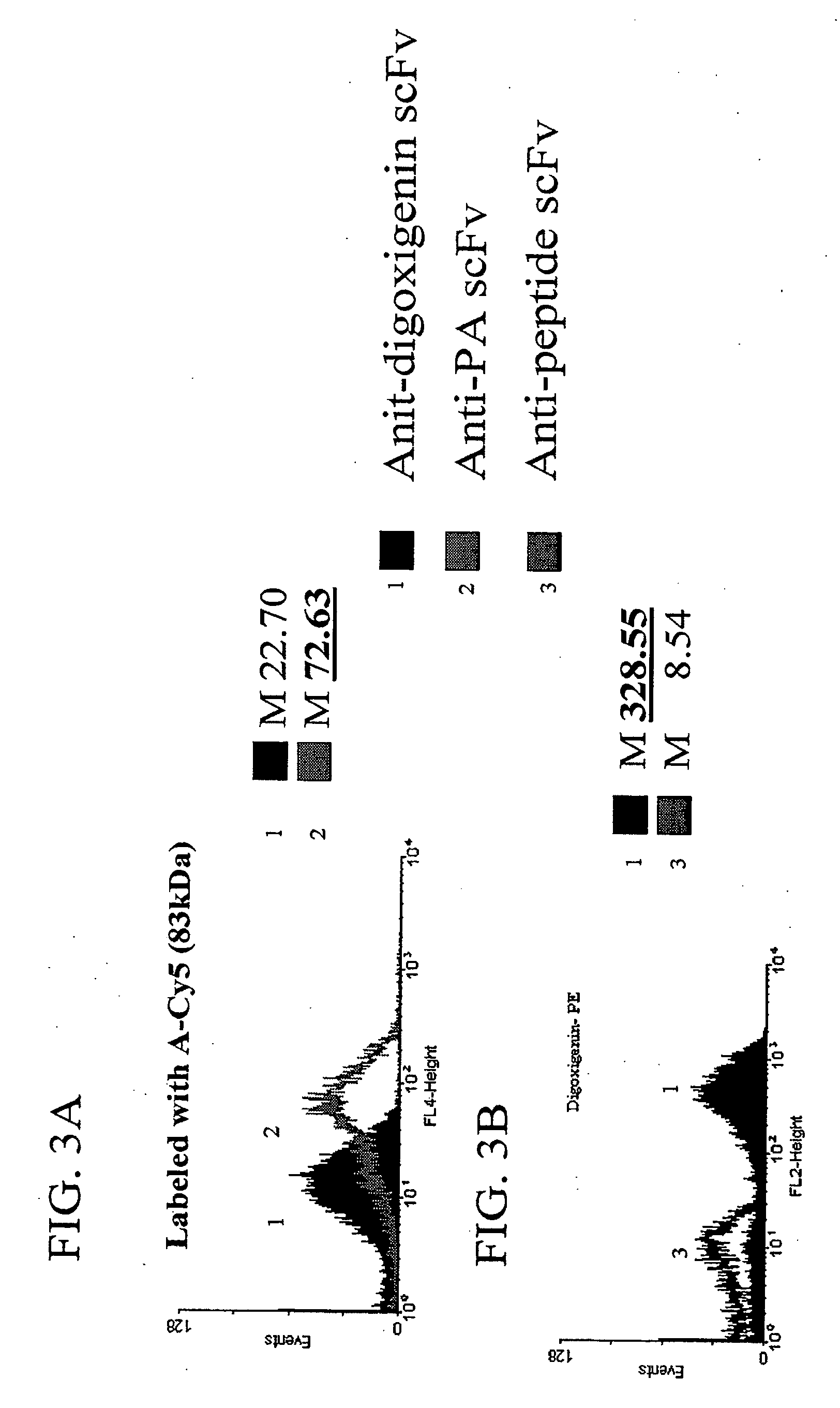
[0153] To demonstrate the ability of scFvs displayed via anchored periplasmic expression to specifically bind to large antigen conjugated fluorophores, E. coli were induced and labeled as described below expressing, via anchored periplasmic expression, an anti-protective antigen(PA) scFv (PA is one component of the anthrax toxin: a 83 kDa protein) or an anti-digoxigenin scFv. Histogram data of 10,000 events demonstrated specific binding to a PA-Cy5 antigen conjugated flourophore as compared to the cells expressing the an anti-digoxigenin scFv (FIG. 3A). To further illustrate this point, digoxigenin was coupled to phycoerythrin(PE), a 240 kDa fluorescent protein. Cells were labeled with this conjugate as described below. It was found that E. coli (10,000 events) expressing the anti-digoxigenin scFv via anchored periplasmic expression were label...
PUM
| Property | Measurement | Unit |
|---|---|---|
| sub-physiological temperature | aaaaa | aaaaa |
| concentration | aaaaa | aaaaa |
| pH | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More