Quench liquids and washing systems for production of microparticles
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
[0103]Microparticles were prepared using a 105 gram batch size coacervation process. The composition and volume of the quench liquid and the composition of the microparticle washing system were varied. The resulting microparticles were characterized with respect to yield, particle size, and residual solvents.
A. Inner Water-In-Oil Emulsion Formation
[0104]A water-in-oil emulsion was created with the aid of a sonicator (Vibracell VCX 750 with a ½″ probe (part #A07109PRB; Sonics and Materials Inc., Newtown, Conn.). The water phase of the emulsion was prepared by dissolving 2.1 g sucrose in 63 g water. The oil phase of the emulsion was prepared by dissolving PLG polymer (97.7 g of purified 50:50 DL4A PLG (Alkermes)) in methylene chloride (1530 g or 6% w / v)). The inner emulsion was formed by adding the water phase to the oil phase while stirring at 1400 to 1600 rpm and sonicating at 100% amplitude over about a five minute period at 2-8° C. The sonication scheme was 2 minutes of sonication...
example 2
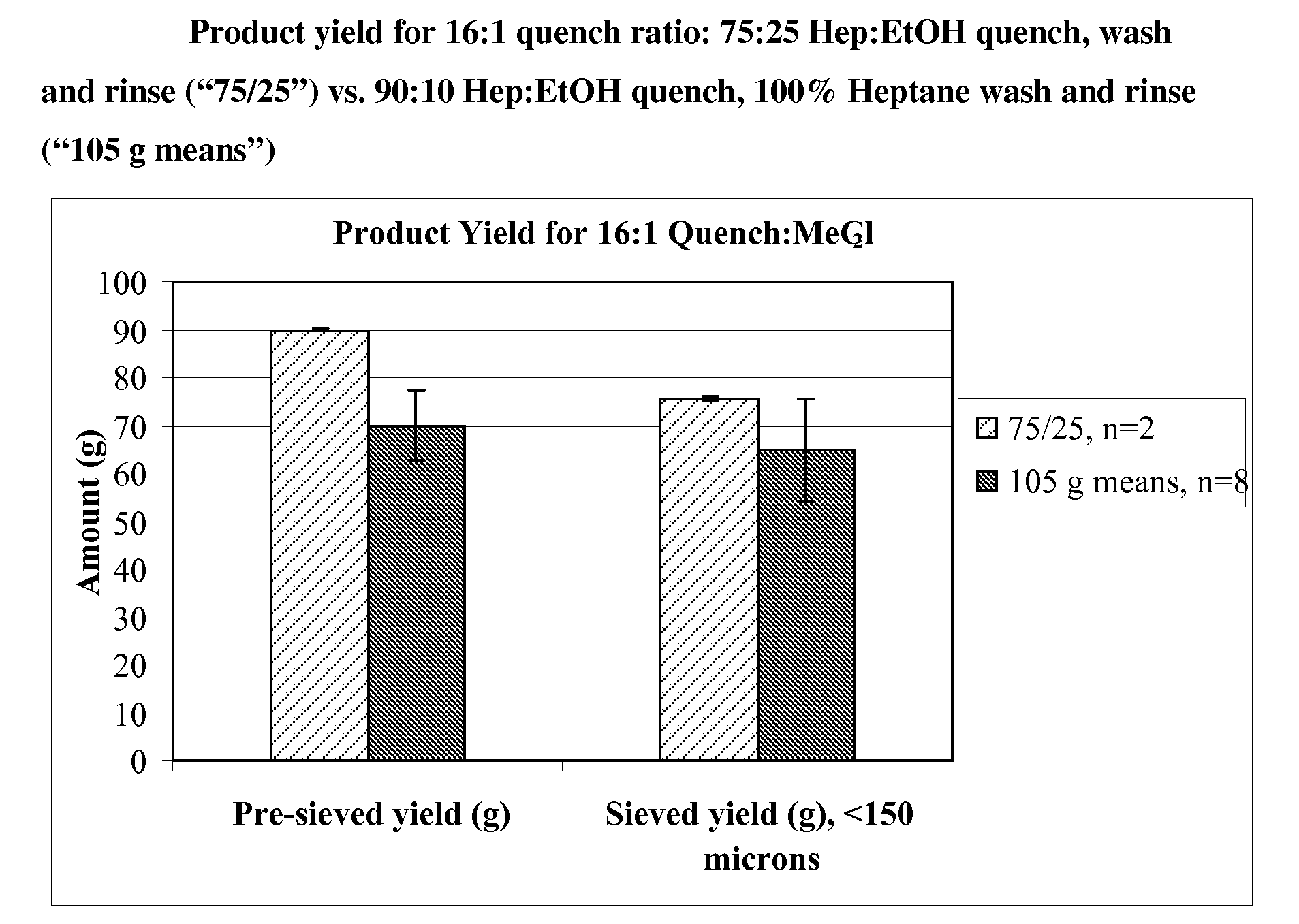
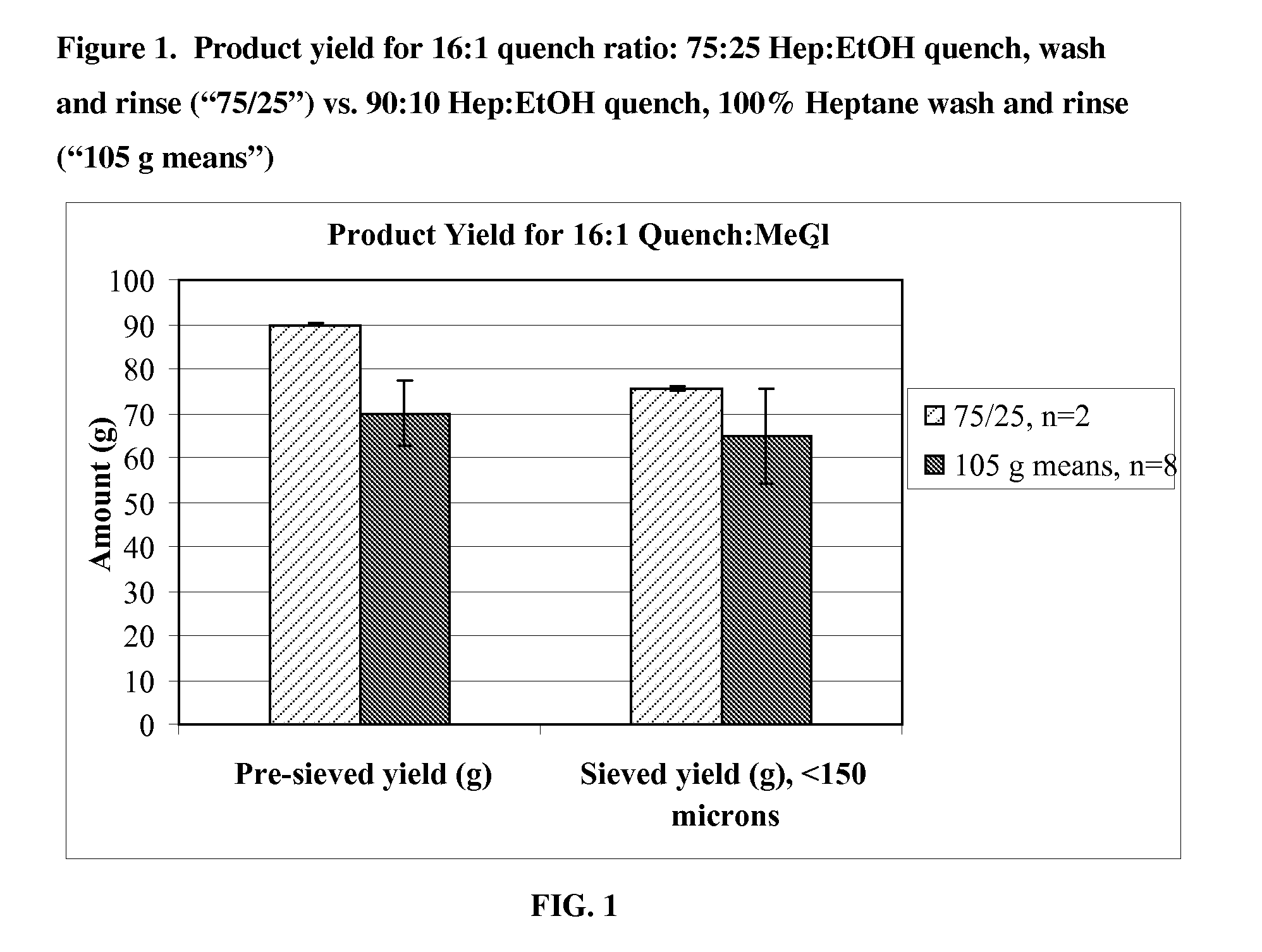
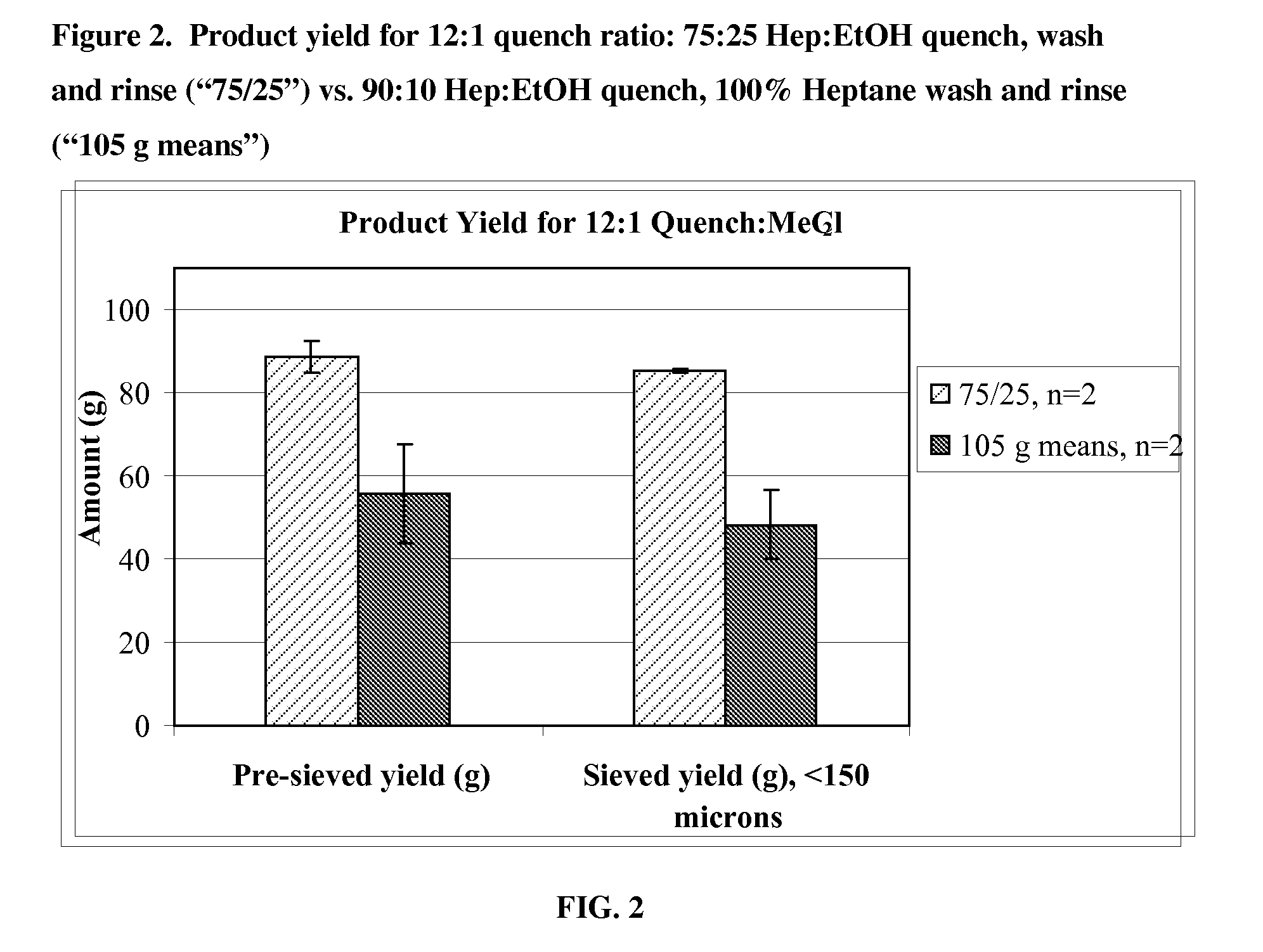
[0115]Microparticles were prepared using a large scale coacervation process. A 20 kg batch was produced using a 75:25 heptane-ethanol mixture for the quench, wash and rinse steps, and a quench liquid to methylene chloride ratio of 12:1. For comparison, four 15 kg reference batches were produced using a 90:10 heptane-ethanol mixture for the quench step, 100% heptane for wash and rinse steps, and a quench liquid to methylene chloride ratio of 16:1. The resulting microparticles were characterized with respect to yield, particle size, and residual solvents.
A. Inner Water-In-Oil Emulsion Formation
[0116]A water-in-oil emulsion was created with the aid of an in-line Megatron homogenizer MT-V 3-65 F / FF / FF, Kinematica AG, Switzerland. For the 20 kg batch, the water phase of the emulsion was prepared by dissolving 400 g sucrose in 12 kg water for irrigation (WFI). The oil phase of the emulsion was prepared by dissolving PLG polymer (e.g., 18,600 g of 50:50 DL4A PLG (Alkermes, Inc.)) in methyl...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Fraction | aaaaa | aaaaa |
| Fraction | aaaaa | aaaaa |
| Fraction | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More