Galectin-3 Immunoassay
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Enzyme-Linked Immunosorbent Assay for Quantitative Detection of Human Galectin-3
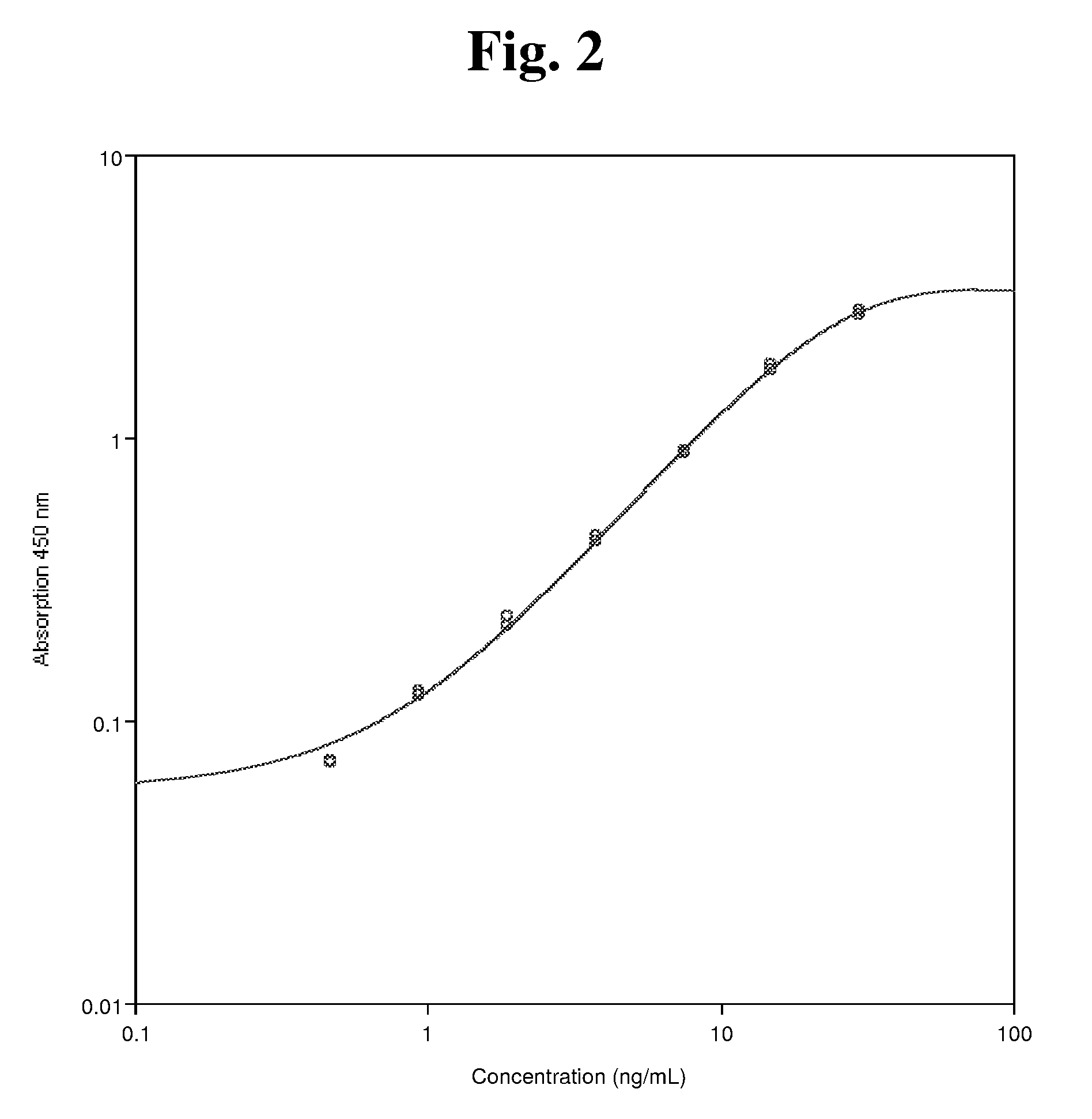
[0071]The human galectin-3 ELISA is an enzyme-linked immunosorbent assay for the quantitative detection of human galectin-3 in EDTA plasma. Human galectin-3 present in the sample or standard bound to antibodies adsorbed to the microwells. Following incubation unbound material was removed during a wash step. HRP conjugated to anti-human galectin-3 antibody was added and bound to the galectin-3 captured by the coating antibody. Following incubation unbound HRP-conjugate was removed during a wash step, and substrate solution reactive with HRP was added to the wells. A colored product was formed in proportion to the amount of human galectin-3 present in the sample or standard. The reaction was terminated by addition of acid and absorbance was measured at 450 nm. A standard curve was prepared from 7 human galectin-3 standard dilutions and human galectin-3 sample concentration determined. Plasma was removed fr...
example 2a
A Kit for Detecting Galectin-3
[0102]Table 7 shows the components of an exemplary kit for the detection of galectin-3.
TABLE 7Galectin-3 assay reagentsQtyNameDescriptionAbbreviation1 platePlateReady-to-use microtiter plate coated with(P)anti-galectin-3 monoclonal antibody(M3 / 38)1 bottleAssay Diluent*Phosphate buffered saline with 1% bovine(AD)serum albumin (45 mL)1 bottleTMB substrateTetramethyl benzidine (15 mL)(TS)1 bottleStop solution0.5M sulfuric acid (10 mL)(ST)2 bottlesWash buffer0.5M Tris buffered saline (2 × 50 mL; 10×(WC)concentrate*concentrate)1 bottleDetectionHorseradish peroxidase (HRP) labeled(DC)concentrate*mouse anti-human galectin-3 antibody87B5 (0.4 mL)2 vialsStandardRecombinant human galectin-3, 12 ng per(S1)vial (lyophilized)2 vialsLow Quality ControlLow QC material, Recombinant human(C1)(QC)\galectin-3 in protein matrix (lyophilized)2 vialsHigh Quality ControlHigh QC material, Recombinant human(C2)(QC)\galectin-3 in protein matrix (lyophilized)2Plate sealsAdhesive ...
example 2b
Detection of Recombinant Galectin-3 Controls
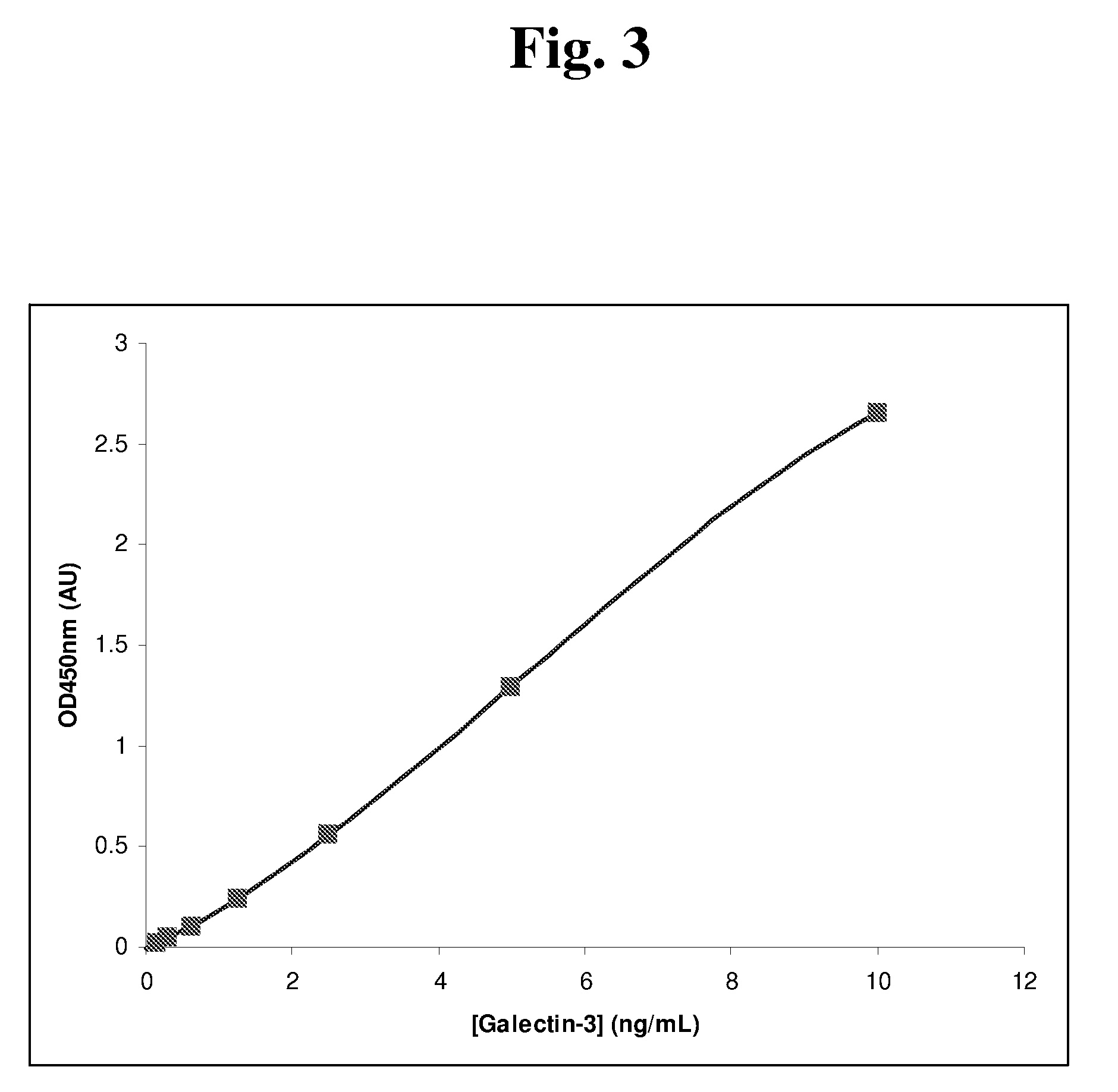
[0105]The kit of Example 2A was used in a microtiter plate-based ELISA assay to quantitate galectin-3 levels. Included in the kit were two monoclonal antibodies against galectin-3. In the assay, described in greater detail in the following paragraph, standards and quality control materials were introduced into the wells and incubated for 60 minutes. During this incubation, the galectin-3 present in the standards was bound to the capture antibody coated onto the well surface. A subsequent wash step removed all unbound material introduced with the sample including unbound galectin-3. The detection antibody was then introduced into the well and incubated for 60 minutes. During this time, an antibody-antigen-antibody complex was formed. After a wash step to remove any unbound detection antibody, the Tetramethyl benzidine (TMB) substrate was added, yielding a blue color in the presence of HRP. The color development was stopped after 20 minutes ...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Fraction | aaaaa | aaaaa |
| Concentration | aaaaa | aaaaa |
| Concentration | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More