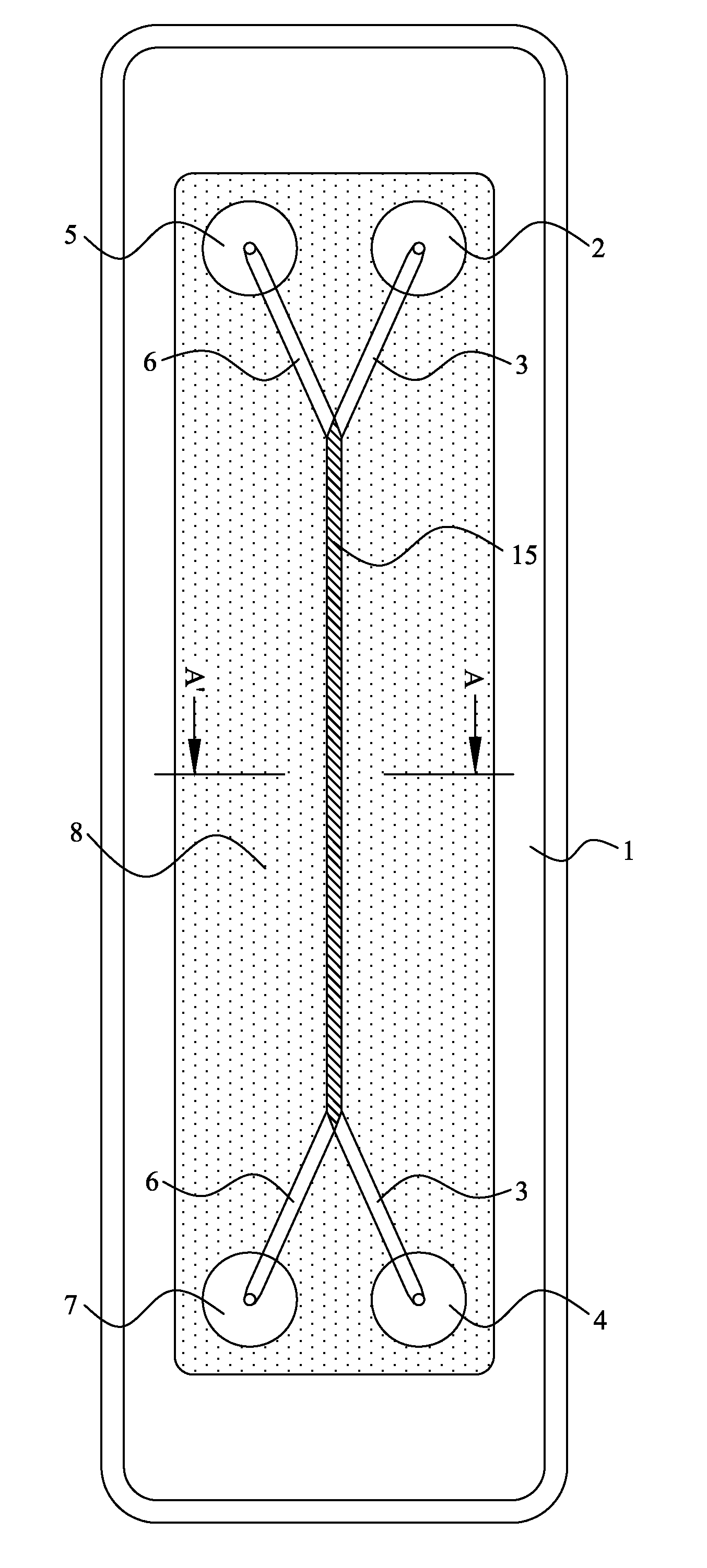
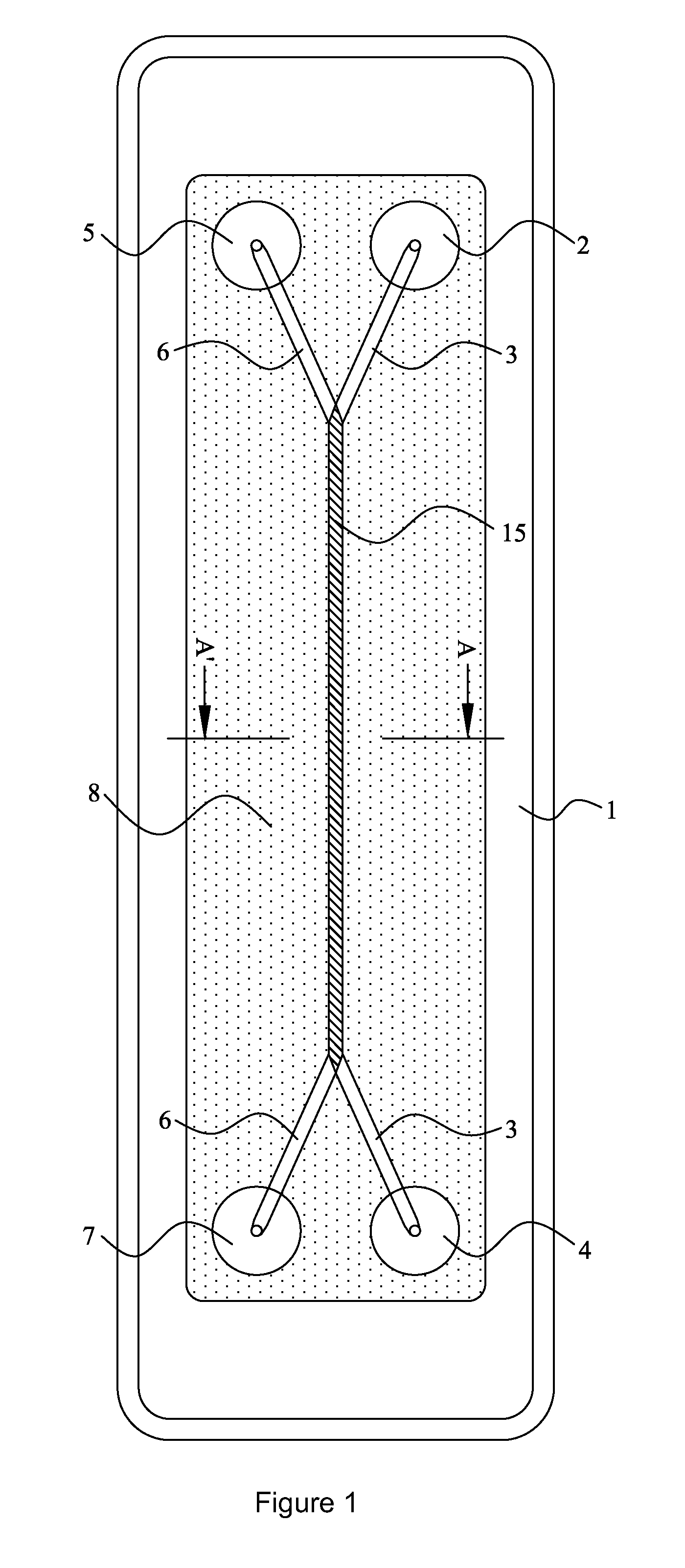
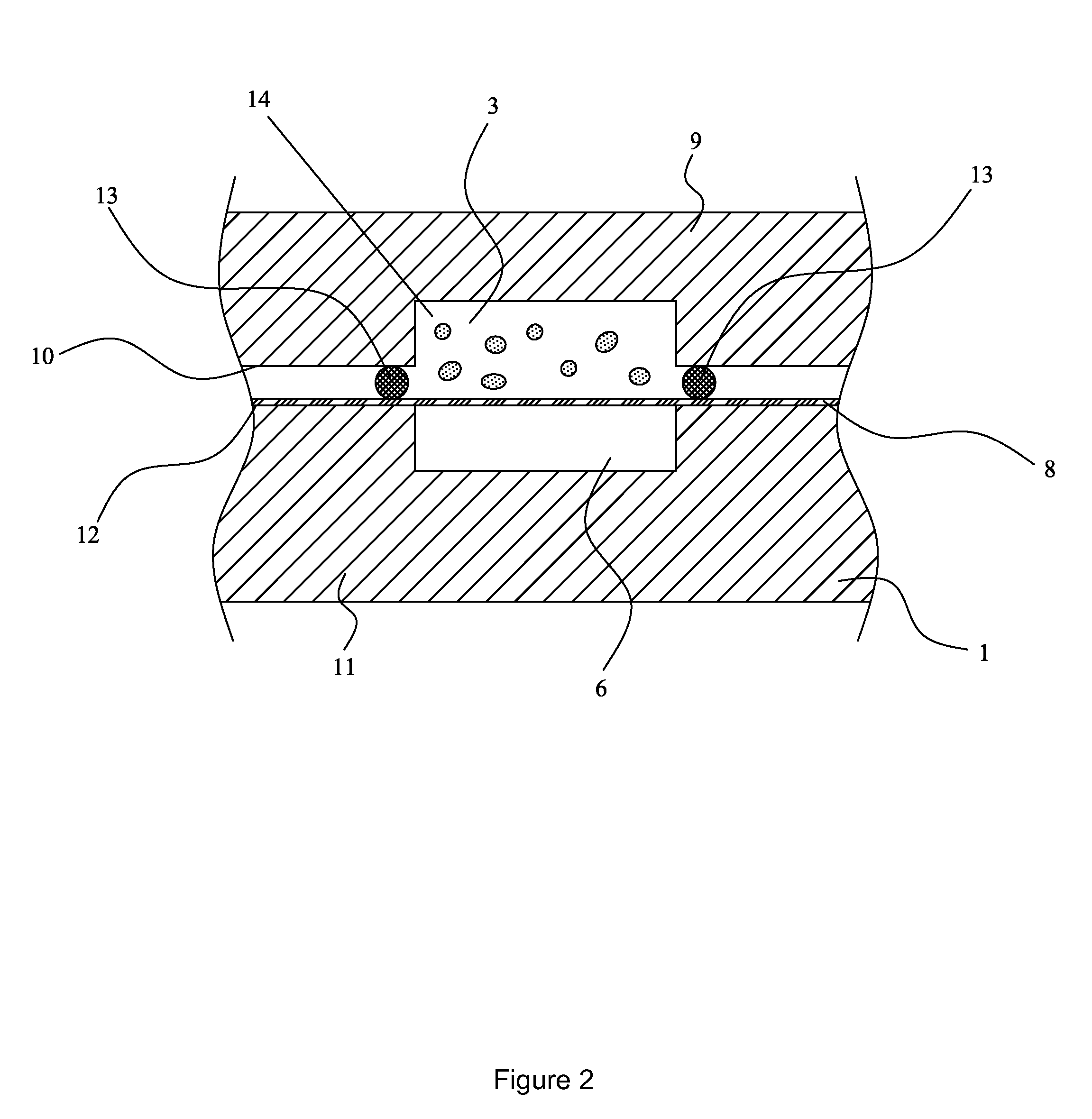
However, margination is not commonly accepted as yet in the three step migration process described above.
Although considerable effort has been invested on the study of
lymphocyte chemoattractants, they remain poorly characterized relative to
monocyte and neutrophil chemoattractants.
However, no molecule identified primarily as a
lymphocyte chemoattractive factor has been sequenced and cloned.
This excessive time span is necessary in order to get reasonable statistics of
cell migration.No physiological flow can be established in this
assay thus is not possible to monitor cells though all stages of leukocyte recruitment.There is no control of the gradient:
chemokine diffusion in the body might be different than
in vitro as it takes longer to get a
cell migration on
in vitro assays.Changes in
cell morphology during
chemotaxis cannot be observed in real-time (because cells transmigrate through the filter).
In addition, Boyden chamber assays cannot readily answer many questions related to the leukocyte migration.
However, in blood even a short persistence of a soluble
chemokine gradient is not feasible because the constant flow of
plasma removes the soluble chemokines from the site of their production.
However, many of these cells are difficult to grow
in vitro and, when grown, are not morphologically similar to
in vivo tissue.
However, animal studies are costly, time-consuming and are limited, by
throughput, to characterize no more than a few compounds.
Furthermore, several drugs have shown unanticipated or unpredicted side effects only after reaching clinical trials or wide-scale release to the public.
The complexity and interplay of biological processes that must be simulated to predict the ADMET properties of a compound far exceed the capabilities of currently available methods and tools.
Even if these biological functions could be faithfully reproduced in vitro, a difficulty remains in getting the capacity and format of the
assay to facilitate testing and analysis of thousands of compounds.
However, there are several disadvantages when using the parallel chamber.
For example, the parallel flow chamber requires a substantial amount of the
drug candidate for the experiment.
Furthermore, setting up the experiment is often
time consuming and rather complex.
Efficient
metabolism of a candidate
drug by a CYP450
enzyme may lead to poor pharmacokinetic properties, while
drug candidates that act as potent inhibitors of a CYP450
enzyme can cause undesirable drug-drug interactions when administered with another drug that interacts with the same CYP450.
Consequently, such earlier-available, reliable pharmacokinetic information may result in greatly reduced
drug development costs and / or increased profits from earlier market entrance.
 Login to View More
Login to View More  Login to View More
Login to View More