Ganoderma-lucidum medicinal mycoplasm, pharmaceutical preparation, pharmaceutical composition and food composition
A technology of medicinal bacteria and pharmaceutical preparations, applied in the field of bioengineering, can solve the problem of only containing active ingredients of ginseng, achieve good health care and medicinal effects, enhance physical fitness, and regulate immunity
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0065] Inoculate the Ganoderma lucidum strain preserved by the Institute of Medicinal Fungi, Changchun University of Traditional Chinese Medicine, on a PDA slant medium, and cultivate it at 27°C ± 1°C, and cultivate it for 7 days to obtain mycelium for use;
[0066] Production containing 2wt% glucose, 0.5wt% peptone, 0.1wt% yeast powder, 0.15wt% KH 2 PO 4 , and 0.1wt% MgSO 4 culture medium of bacteria. Take 100mL of strain culture solution into a 250mL Erlenmeyer flask, and sterilize at 0.1MPa and 121°C for 30min. Cut 4 pieces of 0.5cm×0.5cm mycelium into the strain culture solution of the Erlenmeyer flask in a sterile room, and vibrate and culture for 5 days under the conditions of 28°C and 150r / min to obtain a seed solution containing mycelium balls;
[0067] Take 50g of sun-dried ginseng without insects and mildew, crush it through a 60-mesh sieve, add 50g of water to moisten it for 12 hours, put the obtained ginseng into a 500mL triangular flask, sterilize it at 0.1MPa ...
Embodiment 2
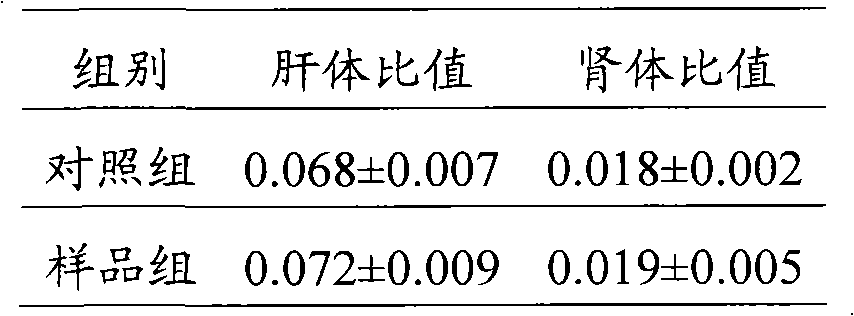
[0073] Toxicity Experiment of Ginseng Medicinal Mycoplasma
[0074] 2.1 Experimental animals and grouping
[0075] 40 Kunming mice were provided by Changchun High-tech Medical Animal Experiment Research Center, half male and half male, weighing 18g-22g.
[0076] 2.2 Experimental Instruments and Reagents
[0077] Electronic scale, mouse cage and 1mL gavage device;
[0078] Take 100g of Ganoderma lucidum and ginseng medicinal mycoplasma powder prepared in Example 1, mix it with 0.2g of Tween 80, add 400g of distilled water to prepare a test drug solution with a mass concentration of 25%;
[0079] 0.2 g of Tween 80 and 400 g of distilled water were prepared as a control solution.
[0080] 2.3 Pre-experiment
[0081] Take 20 small white mice, half male and half male, fasted without food and water for 16 hours, and divided into sample group and control group;
[0082] Give the test drug solution of 7.5g / kg body weight by intragastric administration to the mice of the sample gr...
Embodiment 3
[0096] Ginseng Medicinal Mycoplasma Efficacy Experiment of Improving Sleep
[0097] 3.1 Experimental animals
[0098] Male ICR mice, weighing 18g-26g, were provided by the Experimental Animal Center of Jilin University.
[0099] 3.2 Reagents
[0100] Take the ganoderma ginseng medicinal mycoplasma powder prepared in Example 1, add distilled water to make a test drug solution with a mass concentration of 25%;
[0101] Grind the Ganoderma lucidum provided by Jilin Province Jinyu Pharmaceutical Development Co., Ltd., and add distilled water to make a Ganoderma lucidum solution with a mass concentration of 25%;
[0102] Grind the ginseng provided by Jilin Province Jinyu Pharmaceutical Development Co., Ltd., and make a ginseng solution with a mass concentration of 25% with distilled water;
[0103] Grinding Ganoderma lucidum and Ginseng together, adding distilled water to make a mixed liquid of Ganoderma lucidum and ginseng with a mass concentration of 25%;
[0104] Sodium pent...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Diameter | aaaaa | aaaaa |
| Weight | aaaaa | aaaaa |
| Maximum tolerated dose | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More