Chinese medicinal composition for preventing and treating injury of gastric mucosa and preparation method thereof
A technology for gastric mucosal injury and composition, which is applied in the field of traditional Chinese medicine composition for preventing and treating gastric mucosal injury and its preparation field, can solve the problems of easy generation of drug resistance, large side effects, easy recurrence and the like, achieves prevention and treatment of gastric mucosal diseases, protection The effect of gastric mucosa
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0033] 1. Composition (weight):
[0034] Astragalus 50g, Poria 22g, Dandelion 17g, Evodia 10g, Angelica dahurica 10g, calcined oyster 5g.
[0035] 2. Preparation method:
[0036] 1) Screening, picking, arranging, and processing the various medicines in the above weight ratio;
[0037] 2) Astragalus, Poria cocos, dandelion, and Evodia rutaecarpa were decocted twice, adding 10 times the amount of water each time, and decocted for 2 hours. Concentrate under reduced pressure to (-0.065Mpa, 75°C) to a clear paste with a relative density of 1.30-1.35 (measured at 60°C);
[0038] 3) The concentrated solution is vacuum-dried (-0.085Mpa, 65°C) to obtain a dry extract, which is crushed through a 100-mesh sieve to obtain a fine powder A;
[0039] 4) Angelica dahurica is crushed into fine powder, passed through a 100-mesh sieve; oyster (calcined) is crushed, passed through a 80-mesh sieve, and mixed to obtain fine powder B;
[0040] 5) Mix A and B powders, add 9.1 g of dextrin and 0.44 ...
Embodiment 2
[0041] Embodiment 2: Determination of extraction process parameters
[0042] According to the summary and induction of the literature and materials, combined with the actual production experience, the preparation is formulated to operate as follows:
[0043] 1) Angelica dahurica and oyster (calcined) are powdered and used as medicine;
[0044] 2) Astragalus, Poria, Dandelion, and Evodia are decocted with water, filtered, and the filtrate is concentrated into a clear ointment;
[0045] 3) The above-mentioned Qinggao is dried under reduced pressure, pulverized, mixed, granulated, sized, filled, and packaged.
[0046] In order to make some technical parameters of crushing process and extraction process more specific, the process parameters such as powder yield, solvent addition, extraction time, and extraction times were investigated respectively.
[0047] 1. Inspection of crushing process
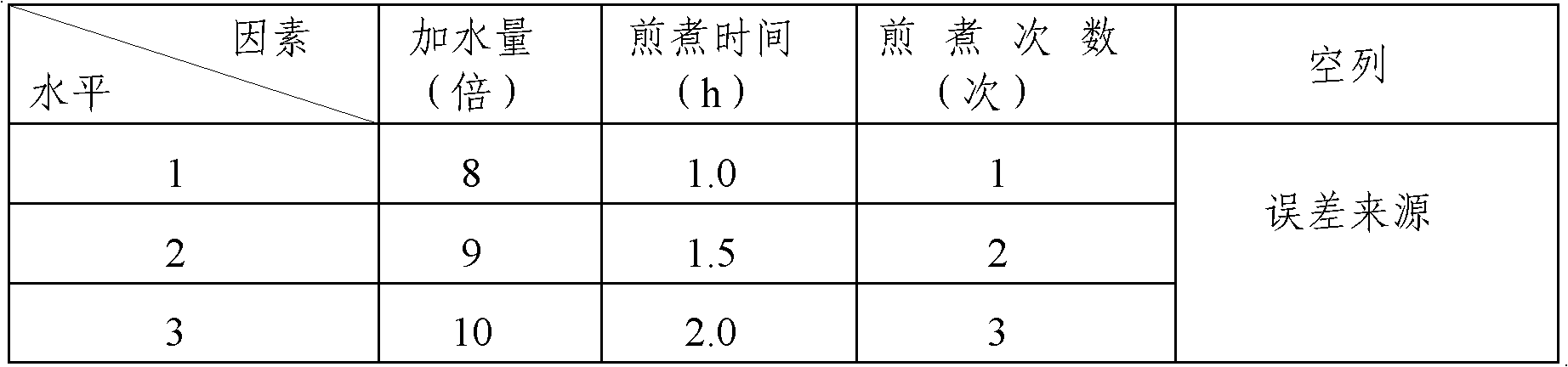
[0048] 1.1 Investigation on the crushing process of Angelica dahurica medicinal materi...
Embodiment 3
[0098] Embodiment 3: Safety investigation
[0099] 1. Materials
[0100] 1.1 Experimental drug: The drug prepared according to Example 1, named Huangpu Fuling Capsule or the test substance, was provided by Shijiazhuang Zangnuo Biotechnology Co., Ltd., and the contents were brown granules and powder. The recommended daily dose for adults is 2100mg / 60kg.BW.
[0101] 1.2 Experimental animals: Kunming mice and SD rats were provided by the Institute of Experimental Animals, Sichuan Academy of Medical Sciences, production license number: SCXK (Sichuan) 2004-16.SPF grade.
[0102] 2. Test method
[0103] 2.1 Rat and mouse acute toxicology test:
[0104] 20 Kunming mice and 20 SD rats were used, half male and half female, the weight of the mice was 18-22g, and the weight of the rats was 180-220g. The large and small mice were randomly divided into two groups, 10 in each group, half male and half male . The test set up a dose group of 15000mg / kg·BW according to the maximum tolerat...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com