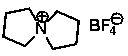Synthetic method for spiro quaternary ammonium tetrafluoroborate
A technology of tetrafluoroborate spiro and spirocyclic quaternary ammonium salts, which is applied in the direction of organic chemistry, can solve the problems of difficult industrial production, reduced yield, and instability of spirocyclic quaternary ammonium bases, and achieve cost savings and pollution reduction. Effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
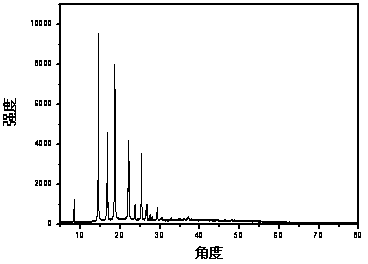
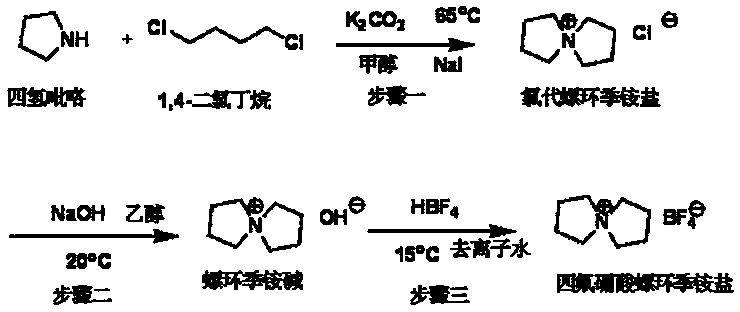
[0020] Step 2: Dissolve 10.9g of chlorospirocyclic quaternary ammonium salt in 150ml of ethanol in a 250ml reaction kettle, then add 2.7g of sodium hydroxide, and stir at -10°C for 0.5h. Then it was filtered with suction, and the filtrate was evaporated to dryness to obtain 11.1 g (yield rate: 80%) of spirocyclic quaternary ammonium base.
[0021] Step 3: Add 11.1g of spirocyclic quaternary ammonium base, 50g of deionized water, and 11.8g of fluoboric acid (40% aqueous solution) into a 100ml reactor, and stir at 0°C for 0.5h. Then add activated carbon and heat to reflux for 2h. Cool, filter with suction, and evaporate the filtrate to dryness to obtain 10.3 g of the final product (yield: 90%, purity ≥ 99%) spirocyclic ammonium tetrafluoroborate.
[0022]
[0023] Example 2:
[0024]
Embodiment 2
[0026] Step 2: Dissolve 21.5g of chlorospirocyclic quaternary ammonium salt in 150ml of ethanol in a 250ml reactor, then add 8.0g of sodium hydroxide, and stir at 20°C for 3h. Then it was filtered with suction, and the filtrate was evaporated to dryness to obtain 26.8 g (yield rate: 98%) of spirocyclic quaternary ammonium base.
[0027] Step 3: Add 26.8g of spirocyclic quaternary ammonium base, 50g of deionized water, and 42.8g of fluoboric acid (40% aqueous solution) into a 100ml reactor, and stir at 15°C for 3h. . Then add activated carbon and heat to reflux for 2h. Cool, filter with suction, and evaporate the filtrate to dryness to obtain 27.2 g (yield: 98%, purity ≥ 99%) of the final product, spirocyclic ammonium tetrafluoroborate.
[0028] Example 3:
[0029]
Embodiment 3
[0031] Step 2: Dissolve 19.2g of chlorospirocyclic quaternary ammonium salt in 150ml of ethanol in a 250ml reactor, then add 9.5g of sodium hydroxide, and stir at 60°C for 5h. Then it was filtered with suction, and the filtrate was evaporated to dryness to obtain 20.1 g (yield rate: 82%) of spirocyclic quaternary ammonium base.
[0032] Step 3: Add 20.1g of spirocyclic quaternary ammonium base, 50g of deionized water, and 42.8g of fluoboric acid (40% aqueous solution) into a 100ml reactor, and stir at 30°C for 5h. . Then add activated carbon and heat to reflux for 2h. Cool, filter with suction, and evaporate the filtrate to dryness to obtain 19.1 g (yield: 92%, purity ≥ 99%) of the final product, spirocyclic ammonium tetrafluoroborate.
[0033] Example 4:
[0034]
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More