Purification method of linaclotide
A technology of linaclotide and purification method, which is applied in the field of purification of polypeptide drugs, can solve the problems of low total product yield, difficulty in removing impurities, and low purity, and achieve high yield, good multimer impurities, and low cost Effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0078] Example 1: Solid phase synthesis of linaclotide
[0079] Weigh 66.67g of 2-CTC resin with a degree of substitution of 1.50mmol / g, add it to the solid phase reaction column, add it to the solid phase reaction column, wash once with DCM, and swell the resin with DCM for 30 minutes, then take 229.76g of Fmoc -Tyr(tBu)-OH (500mmol) was dissolved in DMF, activated by adding 124ml DIEA (750mmol) in an ice-water bath, then added to the above-mentioned reaction column equipped with resin, reacted for 2 hours, and then blocked by adding 1000ml of anhydrous methanol for 1 hour. Washed 3 times with DMF to obtain Fmoc-Tyr(tBu)-CTC resin. Use a mixed solution of DMF:pyridine with a volume ratio of 4:1 to remove Fmoc protection, then wash with DMF for 6 times, weigh 175.72g Fmoc-Cys(Trt)-OH (300mmol), add 40.52gHOBt (300mmol) at a volume ratio of 1 : 1 mixed solution of DCM and DMF, add 46ml DIC (300mmol) under ice-water bath to activate, then add to the above-mentioned reaction c...
Embodiment 2
[0080] Example 2: Cleavage of Linaclotide
[0081] Weigh 33.09g of linaclotide linear peptide resin and add it to a 1000mL three-necked round-bottomed flask. EDT, PhSMe, H 2 Prepare 330ml of lysate for O and TFA, add the lysate to the above resin, and react at 20~30°C for 2 hours. The reaction solution was added to glacial ether for precipitation for 1 hour, centrifuged, washed with anhydrous ether for 6 times, and vacuum-dried to obtain 14.56 g of linaclotide linear peptide.
Embodiment 3
[0082] Example 3: Cyclization of Linaclotide
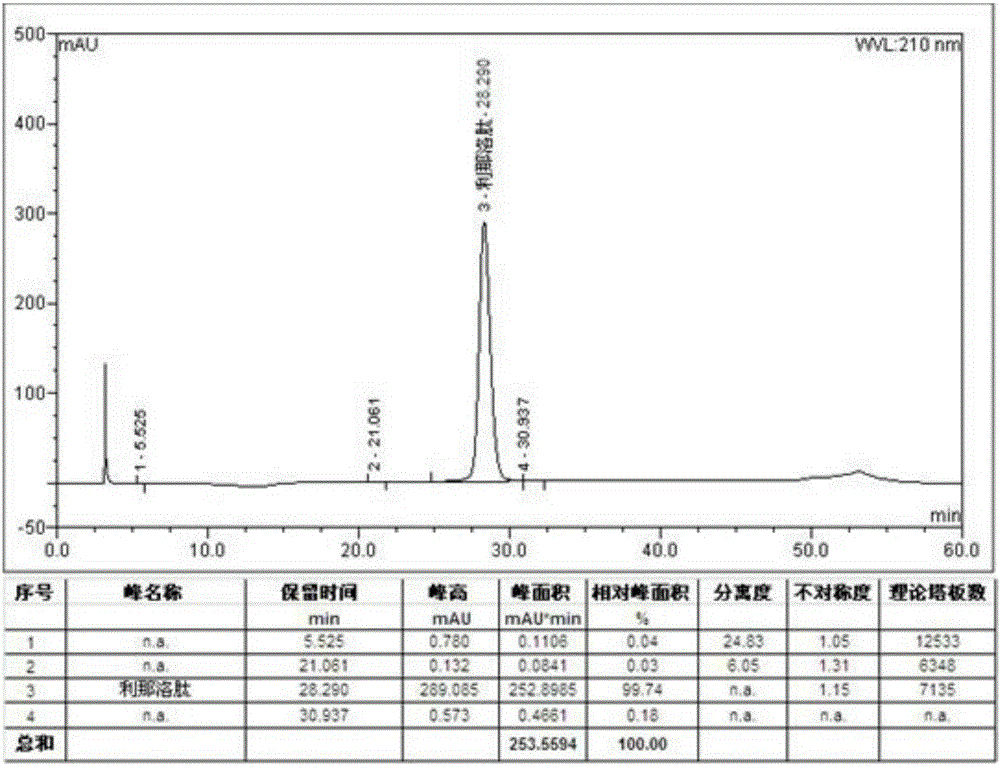
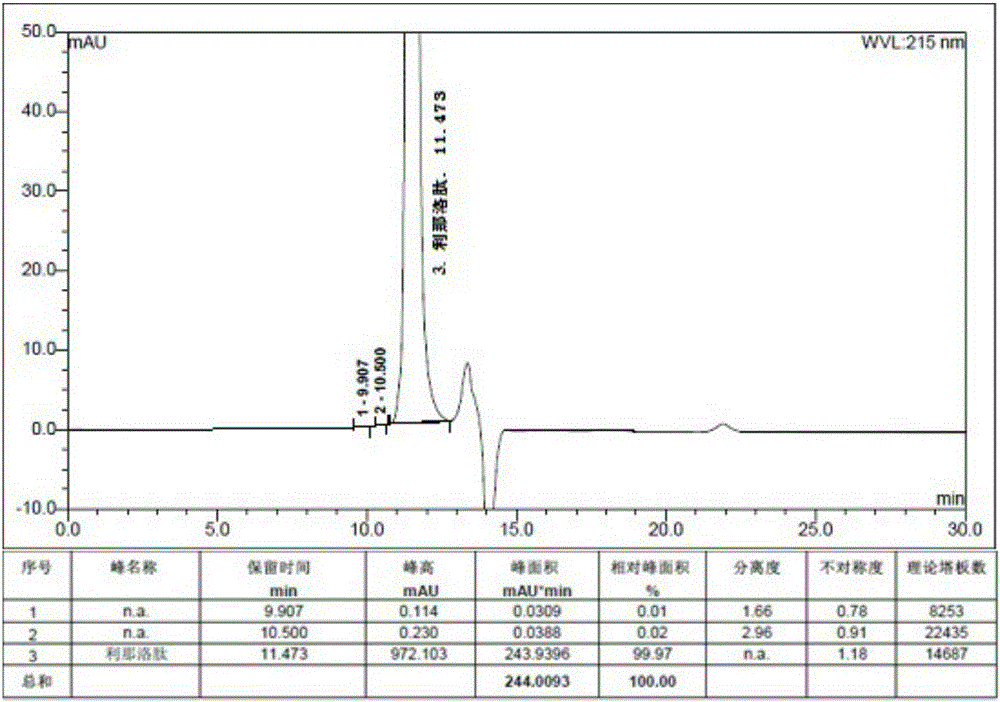
[0083] Add 120g (1mol) sodium dihydrogen phosphate and 1.91g (20mmol) guanidine hydrochloride to 10L solution (water: acetonitrile = 85:15) respectively, stir and dissolve, then add 10.2g linaclotide linear peptide, 25-30℃ After stirring and reacting for 24 hours, it was set aside. The purity of the crude linaclotide was 64.5% (HPLC detection), the polymer impurity content was 2.96% (gel chromatography detection), and the synthesis yield was 45.2%.
PUM
| Property | Measurement | Unit |
|---|---|---|
| particle diameter | aaaaa | aaaaa |
| particle diameter | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More