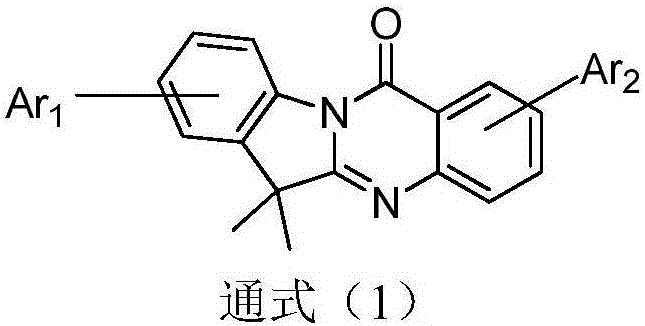
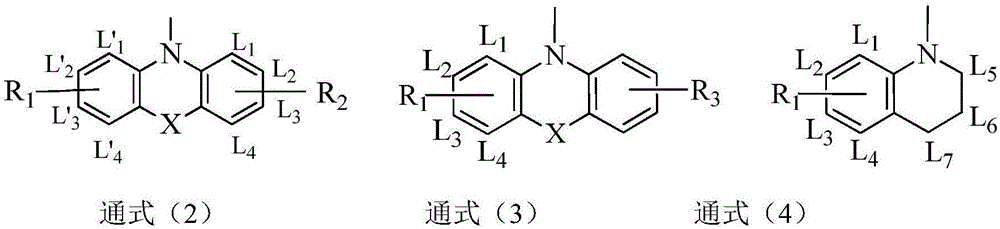
Compound employing quinazolinone derivative as mother nucleus and application of compound
A quinazolinone and compound technology, applied in organic chemistry, chemical instruments and methods, electrical components, etc., can solve problems such as limited applications and lower triplet energy levels
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
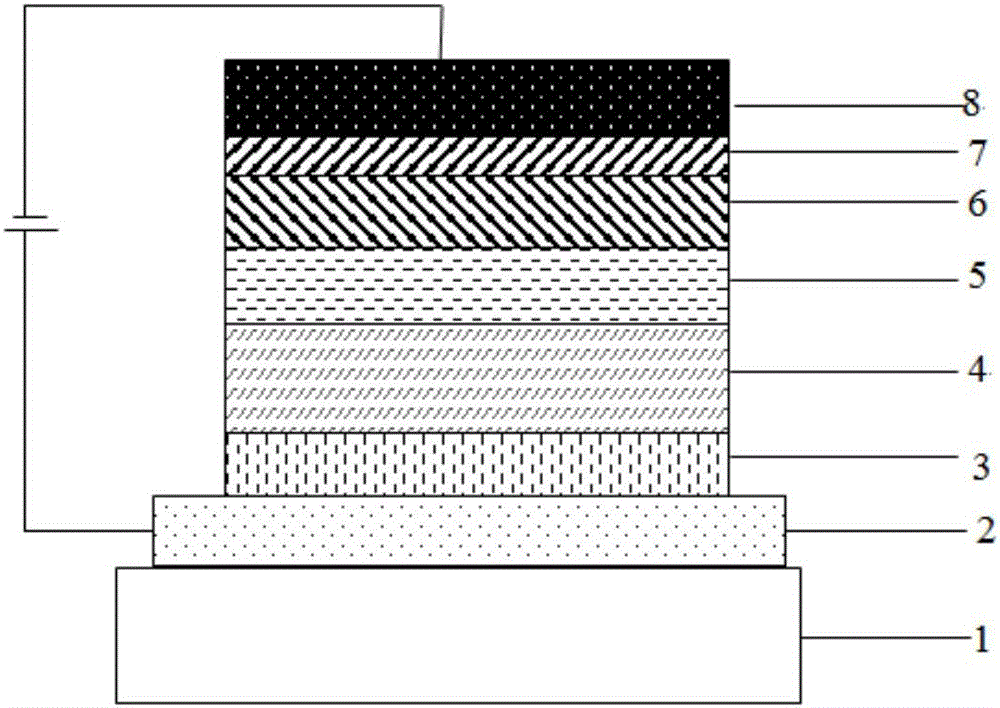
Image
Examples
Embodiment 1
[0075] The synthesis of embodiment 1 compound 22
[0076]
[0077] In a 250ml four-neck flask, add 0.01mol 3-bromo-6,6-dimethylindolo[2,1-b]quinazolin 12(6H)-one, 0.015mol Compound B1, 0.03mol sodium tert-butoxide, 1×10 -4 mol Pd 2 (dba) 3 , 1×10 -4 mol of tri-tert-butylphosphine, 150ml of toluene, heated to reflux for 24 hours, sampling point plate, complete reaction, natural cooling, filtration, filtrate rotary evaporation, silica gel column, to obtain the target product with a purity of 97.36% and a yield of 66.4%. HPLC-MS (m / z): The theoretical value is 559.23, and the measured value is 559.42.
Embodiment 2
[0078] The synthesis of embodiment 2 compound 26
[0079]
[0080] In a 250ml four-neck flask, add 0.01mol 3-bromo-6,6-dimethylindolo[2,1-b]quinazolin 12(6H)-one, 0.015mol Compound B2, 0.03mol sodium tert-butoxide, 1×10 -4 mol Pd 2 (dba) 3 , 1×10 -4 mol of tri-tert-butylphosphine, 150ml of toluene, heated to reflux for 24 hours, sampling plate, reaction complete, natural cooling, filtration, filtrate rotary evaporation, silica gel column, to obtain the target product with a purity of 96.59% and a yield of 59.8%. HPLC-MS (m / z): The theoretical value is 559.23, and the measured value is 559.67.
Embodiment 3
[0081] The synthesis of embodiment 3 compound 42
[0082]
[0083]In a 500ml four-neck flask, under nitrogen atmosphere, add 0.01mol 2-bromo-benzo[d]benzo[4,5]imidazo[2,1-b]oxazole, 0.015mol compound C1, and use The mixed solvent is dissolved (180ml toluene, 90ml ethanol), then add 0.03mol Na 2 CO 3 aqueous solution (2M), then add 0.0001mol Pd (PPh 3 ) 4 , heated to reflux for 10-24 hours, sampled and plated, the reaction was complete. Naturally cooled, filtered, the filtrate was rotary evaporated, and passed through a silica gel column to obtain the target product with a HPLC purity of 97.80% and a yield of 67.5%. HPLC-MS (m / z): The theoretical value is 635.26, and the measured value is 635.68.
PUM
| Property | Measurement | Unit |
|---|---|---|
| Thickness | aaaaa | aaaaa |
| Thickness | aaaaa | aaaaa |
| Thickness | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More