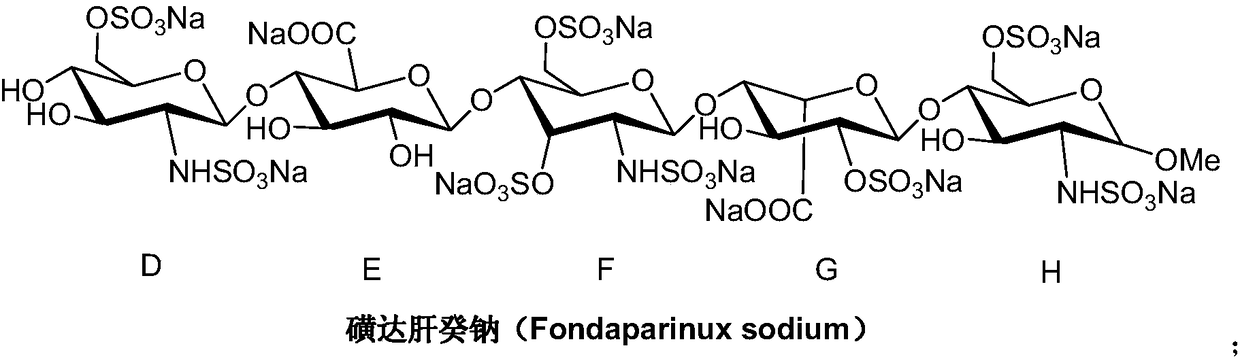
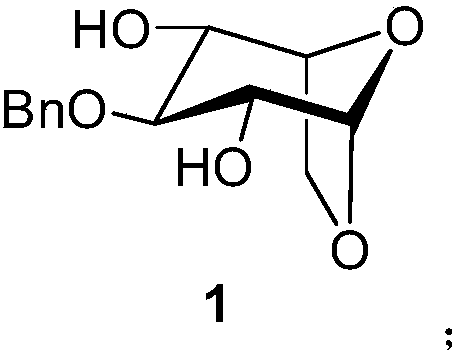
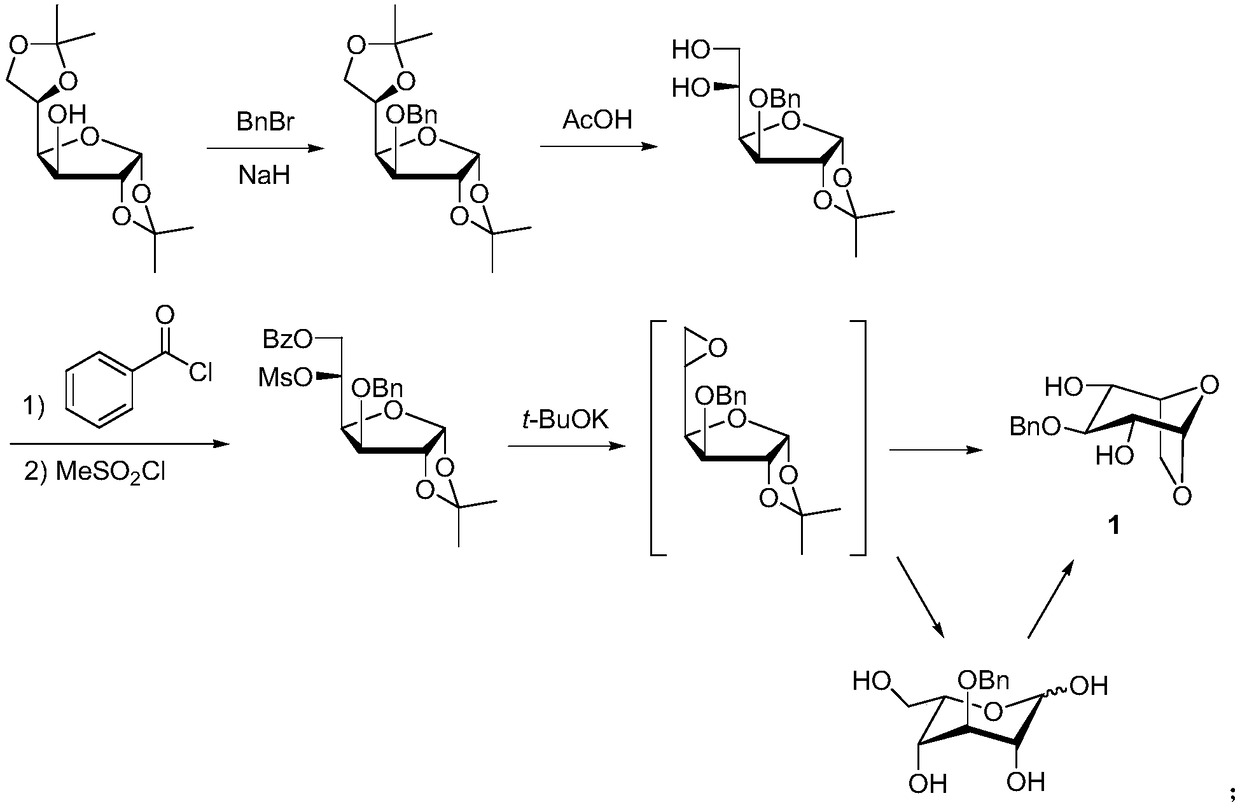
Method for synthesizing fondaparinux sodium monosaccharide intermediate
A synthesis method and compound technology, applied in the field of synthesis of fondaparinux sodium monosaccharide intermediates, can solve the problems of unfavorable purification and refinement of intermediates and products, low yield, lower product quality, etc., and achieve low cost and high yield. The effect of high yield, improving product quality and yield
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0048] A) Synthesis of 6-O-benzoyl-1,2-O-isopropylidene-3-O-benzyl-α-D-glucofuranose (compound shown in formula (3)):
[0049] 1,2-O-isopropylidene-3-O-benzyl-α-D-glucofuranose (95g, the compound shown in formula (4)) was dissolved in methyl tert-butyl ether (1L), added benzene Add formic acid (37.4g) and N,N-diisopropylethylamine (59.3g), dropwise add a solution of N,N'-carbonyldiimidazole (59.6g) in methyl tert-butyl ether (300mL), keep warm at 60°C After 12 hours of reaction, the reaction was completed, evaporated to dryness under reduced pressure, extracted with dichloromethane, washed with brine, dried over anhydrous sodium sulfate, and evaporated to dryness under reduced pressure. The crude product was recrystallized from a mixed solvent of ethyl acetate-petroleum ether to obtain 6- O-benzoyl-1,2-O-isopropylidene-3-O-benzyl-α-D-glucofuranose, white solid (111.6 g), yield 88%, purity 97.3%.
[0050] B) Synthesis of 6-O-benzoyl-1,2-O-isopropylidene-3-O-benzyl-5-O-trifluor...
Embodiment 2
[0055] A) Synthesis of 6-O-benzoyl-1,2-O-isopropylidene-3-O-benzyl-α-D-glucofuranose (compound shown in formula (3)):
[0056] 1,2-O-isopropylidene-3-O-benzyl-α-D-glucofuranose (30g, compound shown in formula (4)) was dissolved in toluene (400mL), added benzoic acid (14.2g) and triethylamine (19.6g), add dropwise a solution of N,N'-dicyclohexylcarbodiimide (31.9g) in toluene (100mL) and react at 20°C for 24h. After the reaction is complete, evaporate to dryness under reduced pressure , extracted with dichloromethane, washed with salt water, dried over anhydrous sodium sulfate, and evaporated to dryness under reduced pressure. The crude product was recrystallized from a mixed solvent of ethyl acetate-petroleum ether to obtain 6-O-benzoyl-1,2-O- Isopropylidene-3-O-benzyl-α-D-glucofuranose, white solid (36.1 g), yield 90%, purity 98.1%.
[0057] B) Synthesis of 6-O-benzoyl-1,2-O-isopropylidene-3-O-benzyl-5-O-methylsulfonyl-α-D-glucofuranose (shown in formula (2) compound of): ...
Embodiment 3
[0062] A) Synthesis of 6-O-benzoyl-1,2-O-isopropylidene-3-O-benzyl-α-D-glucofuranose (compound shown in formula (3)):
[0063] 1,2-O-isopropylidene-3-O-benzyl-α-D-glucofuranose (100g, the compound represented by formula (4)) was dissolved in dichloromethane (1L), and benzoic acid (59g ) and N,N-xylaniline (97.6g), slowly added 1-ethyl-(3-dimethylaminopropyl)carbodiimide hydrochloride (111.2g), and kept at 40°C for 18h, After the reaction was completed, the reaction was rotary evaporated to dryness under reduced pressure, extracted with dichloromethane, washed with salt water, dried over anhydrous sodium sulfate, and evaporated to dryness under reduced pressure. The crude product was recrystallized from a mixed solvent of ethyl acetate-petroleum ether to obtain 6-O-benzene Formyl-1,2-O-isopropylidene-3-O-benzyl-α-D-glucofuranose, white solid (114.9 g), yield 86%, purity 98.4%.
[0064] B) Synthesis of 6-O-benzoyl-1,2-O-isopropylidene-3-O-benzyl-5-O-tosyl-α-D-glucofuran (formul...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com