Preparation method of trimethylphenylammonium tribromide and recycling method of trimethylphenylammonium tribromide in naproxen production
A technology of trimethylphenylammonium tribromide and methylphenylammonium tribromide, which is applied to the preparation of amino compounds from amines, the preparation of functional groups replaced by amino groups, the purification/separation of amino compounds, etc., which can solve the problems of losing market competition, Naproxen industrialization cost increase and other problems, to achieve the effect of reducing production cost, low cost and high yield
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
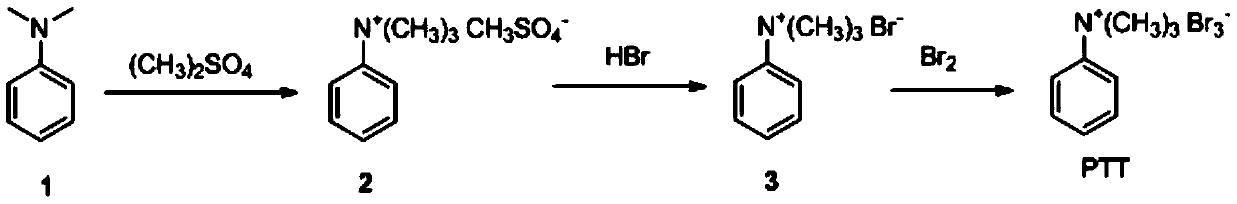
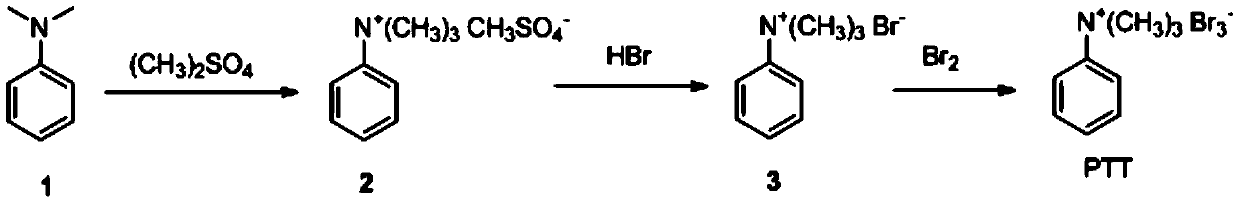
[0026] The preparation of PTT, reaction formula is:
[0027]
[0028] In an Erlenmeyer flask equipped with a thermometer and a magnetic stirrer, a solution of 248 g (260 mL, 2 moles) of freshly distilled xylidine and 1000 mL of toluene was added. Stir to dissolve and heat to 35°C with stirring. The heating was stopped and 190 ml (2 moles) of freshly distilled dimethyl sulfate were added over 20 minutes via the dropping funnel. After a few minutes, the colorless ammonium methyl sulfate salt began to crystallize. During the dropwise addition, the temperature changed slightly, and after 1 hour after the dropwise addition, the temperature rose slowly to 50° C. due to the exothermic reaction. It was then reacted for an additional 1.5 hours at room temperature (i.e. without heating the reaction), followed by heating on a steam bath for 1 hour. After cooling, the ammonium phenyltrimethylmethylsulfate was filtered, washed with 200 ml of dry toluene and dried under vacuum. Obtai...
Embodiment 2
[0032] Preparation of PTT
[0033] In an Erlenmeyer flask equipped with a thermometer and a magnetic stirrer, a solution of 248 g (260 mL, 2 moles) of freshly distilled xylidine and 1000 mL of toluene was added. Stir to dissolve and heat to 45°C with stirring. The heating was stopped and 228 ml (2.4 moles) of freshly distilled dimethyl sulfate were added over 20 minutes via the dropping funnel. After a few minutes, the colorless ammonium methyl sulfate salt began to crystallize. During the dropwise addition, the temperature changed slightly, and one hour after the dropwise addition, the temperature rose slowly to 55°C. It was then reacted for a further 1.5 hours at room temperature and then heated on a steam bath for 1 hour. After cooling, ammonium phenyltrimethylmethylsulfate was filtered, washed with 200 ml of dry toluene, and dried under vacuum. Obtain 465 grams of phenyl trimethyl ammonium methyl sulfate, yield 94%.
[0034] In an Erlenmeyer flask, 100 g (0.4 mol) of ...
Embodiment 3
[0037] Preparation of PTT
[0038] In an Erlenmeyer flask equipped with a thermometer and a magnetic stirrer, a solution of 248 g (260 mL, 2 moles) of freshly distilled xylidine and 1000 mL of toluene was added. Stir to dissolve and heat to 40°C with stirring. The heating was stopped and 209 ml (2.2 moles) of freshly distilled dimethyl sulfate were added over 20 minutes via the dropping funnel. After a few minutes, the colorless ammonium methyl sulfate salt began to crystallize. During the dropwise addition, the temperature changed slightly, and one hour after the dropwise addition was completed, the temperature rose slowly to 45°C. It was then reacted for a further 1.5 hours at room temperature and then heated on a steam bath for 1 hour. After cooling, ammonium phenyltrimethylmethylsulfate was filtered, washed with 200 ml of dry toluene, and dried under vacuum. Obtain 440 grams of phenyl trimethyl ammonium methyl sulfate, yield 89%.
[0039] In an Erlenmeyer flask, 100 g...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More