Method for quantitatively analyzing vorolanib and metabolite X297 thereof by using liquid chromatography-tandem mass spectrometry technology
A tandem mass spectrometry and quantitative analysis technology, which is applied in the direction of analyzing materials, measuring devices, and material separation, can solve the problems of simultaneous quantitative analysis of vronib, and achieve high practicability and reliability, short analysis time, and high sensitivity Effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0052] Example 1: Quantitative analysis of vronib and its metabolite X297
[0053] This example illustrates the quantitative analysis of voronib and its metabolite X297 using high performance liquid chromatography-tandem mass spectrometry.
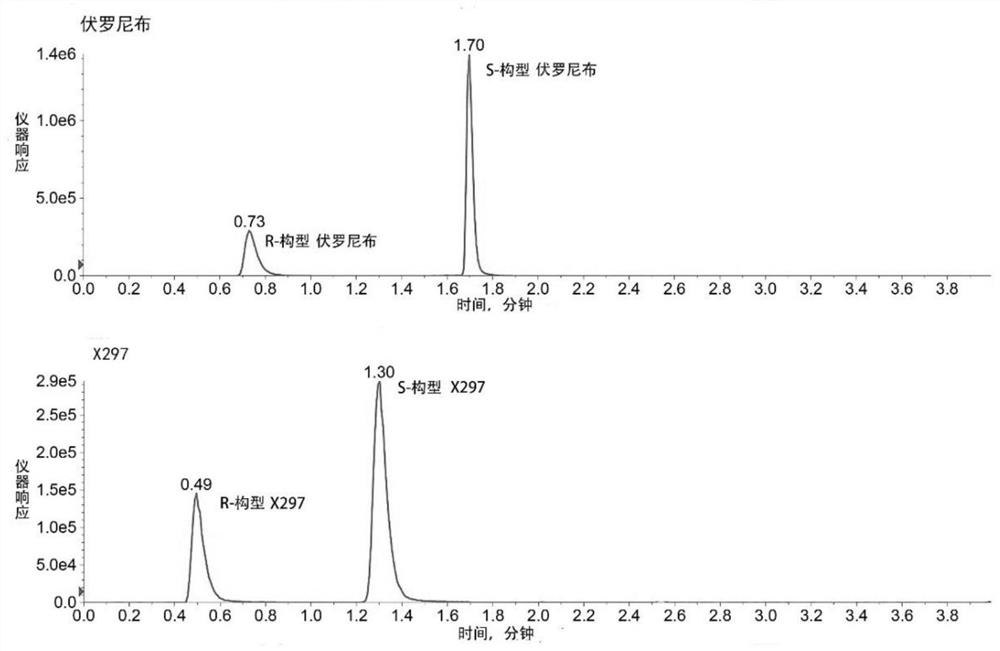
[0054] Solution preparation: Precisely weigh 1mg each of voronib and X297 powder respectively, dissolve in 1mL dimethyl sulfoxide, prepare a solution with a concentration of 1mg / mL, use diluent (acetonitrile:water=1:2(v / v )) be diluted to the solution of 1.5 / 0.75ng / mL (voronib / X297), get 10 μ L and inject in the chromatographic column of high performance liquid chromatography, adopt the analytical method provided by the invention, obtain mass chromatogram ( figure 1 ). Voronib and X297 can be completely separated by the method of the present invention, and can be quantitatively analyzed at the same time.
[0055] University liquid chromatography conditions:
[0056] Chromatographic column: Acquity BEH C18 column, specification: 1.7μm ...
Embodiment 2
[0074] Example 2: Methodological verification
[0075] This example illustrates the methodological verification of the method for the quantitative analysis of voronib and its metabolite X297 in plasma.
[0076] Plasma sample preparation method:
[0077] Take 50 μL plasma containing voronib and its metabolite X297, add 50 μL d 6 - Erlotinib (internal standard) working solution (50ng / mL), add 900 μ L of precipitant (acetonitrile), centrifuge for 10 minutes, take 200 μ L of supernatant, blow dry with nitrogen, add 400 μ L of complex solution (acetonitrile: water = 1: 2(v / v)), mix well, and take 10μL for injection.
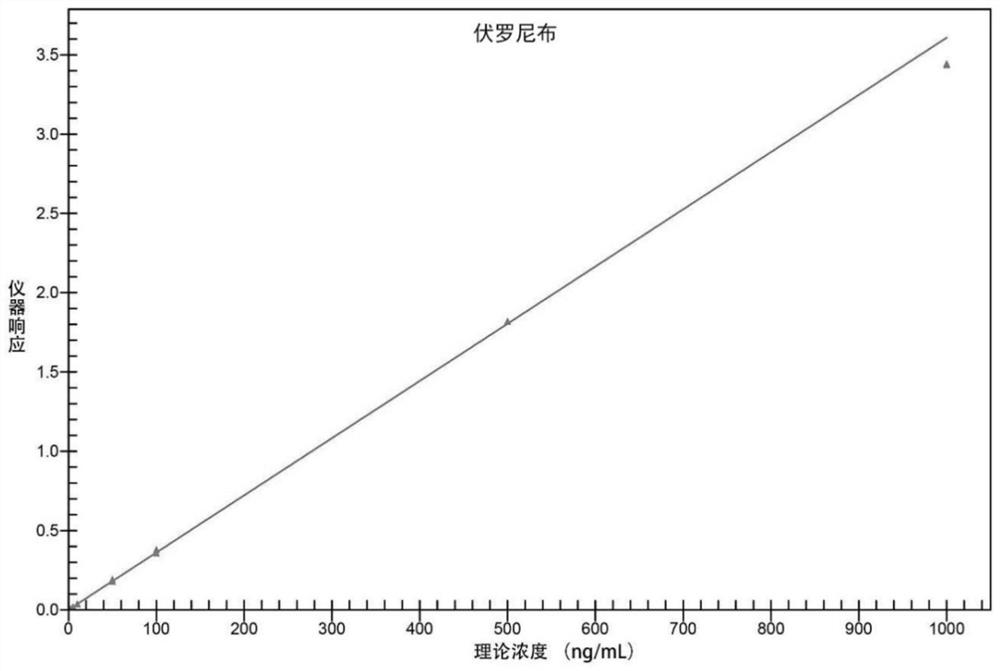
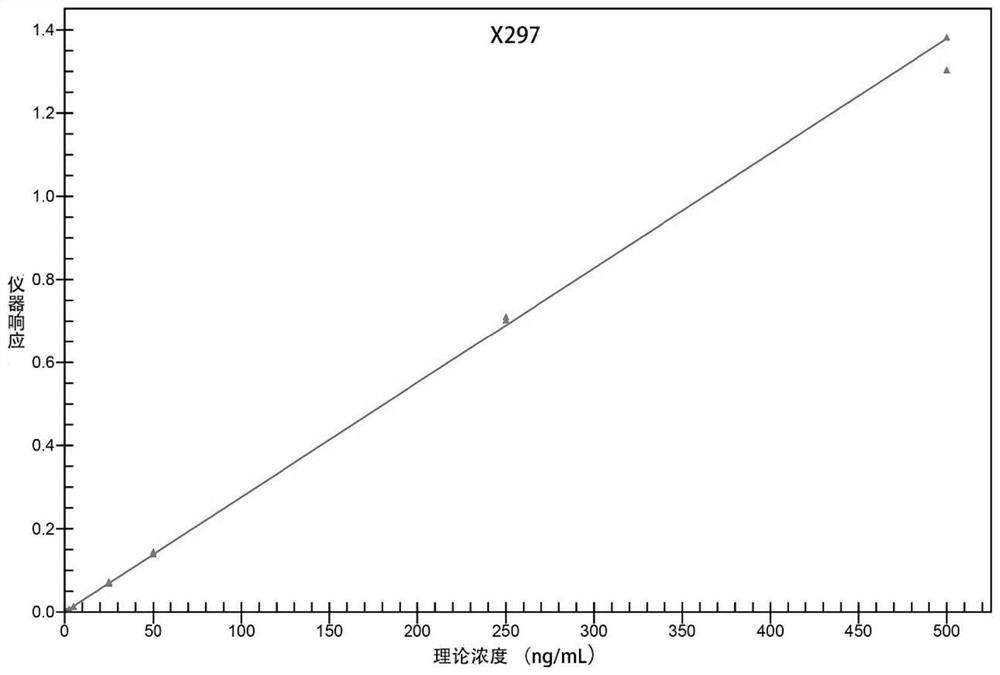
[0078] Adopt the analysis method provided by the invention, use internal standard method to carry out quantitative analysis. The method was validated for specificity, standard curve, lower limit of quantitation, precision and accuracy, matrix effects and recovery.
[0079] A. Specificity: Individual plasma samples from 6 different volunteers were used to measure u...
Embodiment 3
[0087] Embodiment three: Pharmacokinetic study
[0088] This example illustrates the pharmacokinetic study of the original drug and its metabolites after oral administration of voronib in tumor patients.
[0089] Blood samples at different stages were collected from patients with advanced malignant solid tumors after oral administration of voronib 200mg and 400mg once a day respectively:
[0090] a. Dose escalation phase: collect blood samples from subjects 0.5 hours before administration and 0.5, 1, 2, 3, 4, 6, 8, 12, 24, 36, 48 hours after administration on day 1; On the 8th, 15th, and 22nd days of continuous administration, the patients were followed up 0.5 hours before the administration, and 0.5 hours before the administration on the 28th day, 0.5, 1, 2, 3, 4, 6, 8, 12, 0.5, 1, 2, 3, 4, 6, 8, 12, 24-hour blood sample;
[0091] b. Expansion phase: Collect subjects’ blood samples at 0.5 hour before administration, 0.5, 1, 2, 3, 4, 6, 8, 12, 24 hours after administration o...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Collision energy | aaaaa | aaaaa |
| Collision energy | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More