Zwitter-ion hydrogel capable of collecting type II collagen and preparation method and application thereof
A zwitterionic and hydrogel technology, which is applied in the field of zwitterionic hydrogel and its preparation, to recruit type II collagen secreted by bone marrow mesenchymal stem cells or chondrocytes, can solve the problems of inability to recruit type II collagen, and achieve easy Mass production and application promotion, broad application prospects, and the effect of improving the restoration effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
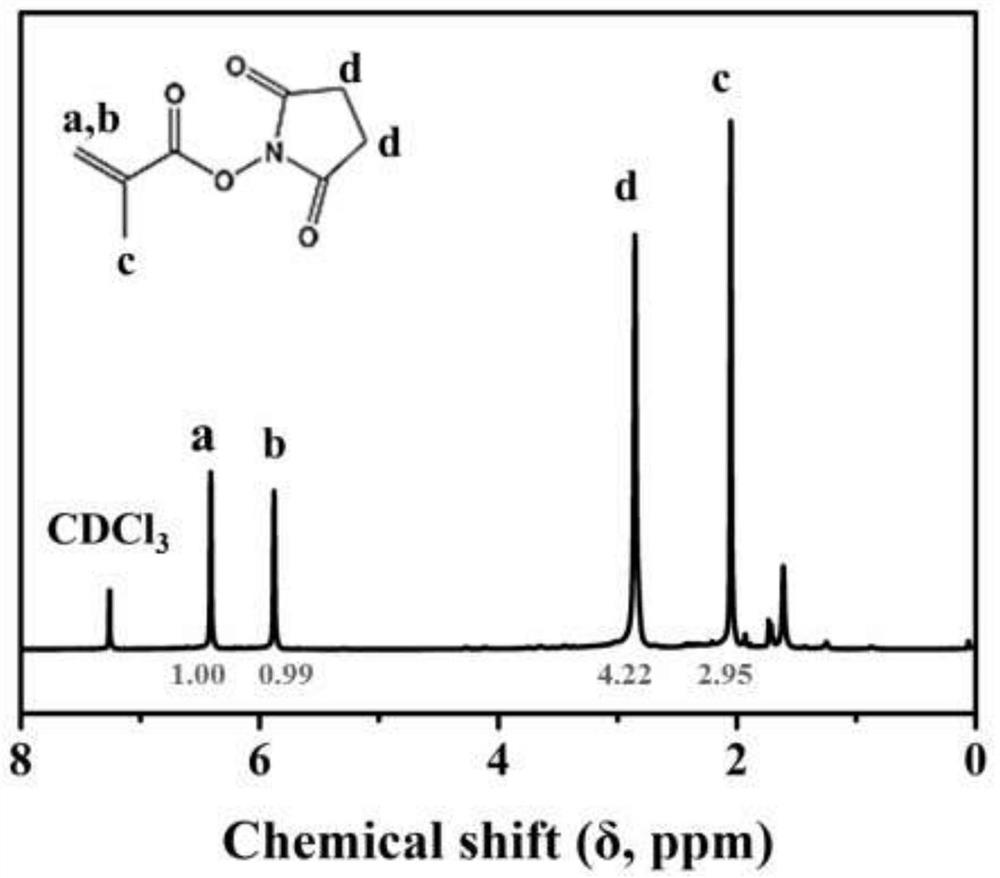
[0037] In this embodiment, the preparation method of active ester functional monomer is provided, and the steps are as follows:
[0038] Dissolve N-hydroxysuccinimide (NHS, 1.1736g, 12mmol) and triethylamine (1,650μL, 12mmol) in 5mL of chromatographic grade dichloromethane, and stir for 30 minutes under ice-bath conditions. At the same time, methyl Acryloyl chloride (1 mL, 11 mmol) was dissolved in 5 mL of chromatographic grade dichloromethane and added dropwise to the previously prepared solution at a rate of 1 drop per 10 seconds. After the solution was added dropwise, the reaction system was gradually returned to room temperature and the reaction was continued for 12 h. After the reaction, wash with distilled water 3 times, 5 mL each time; then dry with anhydrous sodium sulfate, concentrate the obtained organic solution by rotary evaporation, and dissolve in ethyl acetate / n-hexane (1 / 3) mixed solution Precipitation gave a white product. Its reaction equation is shown in f...
Embodiment 2
[0042] In this embodiment, a method for preparing a zwitterionic hydrogel capable of recruiting type II collagen is provided, and the steps are as follows:
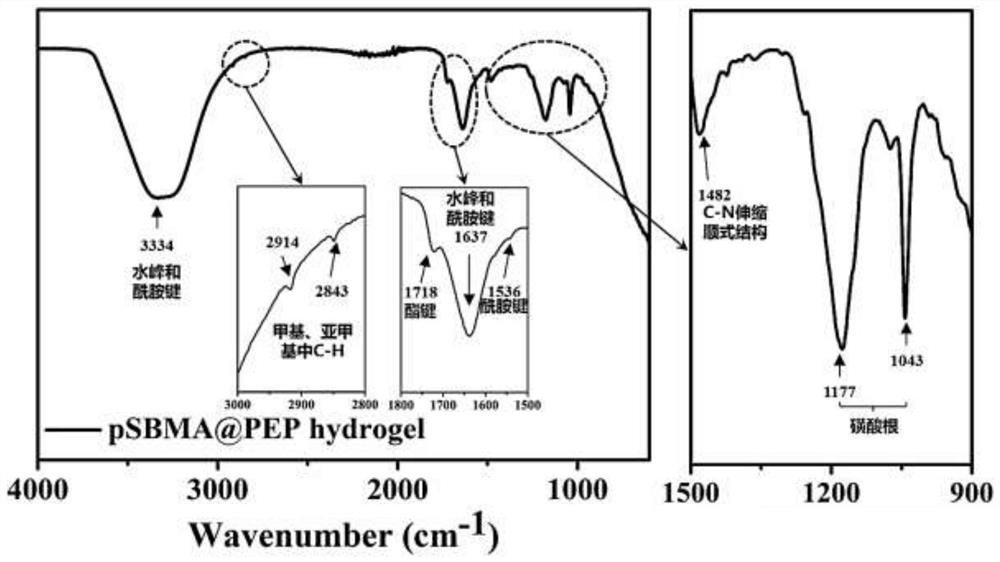
[0043] (1) Preparation of functionalized sulfobetaine hydrogel
[0044] Dissolve sulfobetaine monomer, active ester monomer, cross-linking agent MBA and photoinitiator I2959 in 15 mL of ultrapure water, and ultrasonically dissolve all components fully. Add the solution to a 24-well plate with 1 mL of liquid per well. Finally, it is cross-linked into a gel under ultraviolet light. Wherein, the chemical structure of the zwitterionic monomer used as the main structure of the hydrogel is shown in formula (II):
[0045]
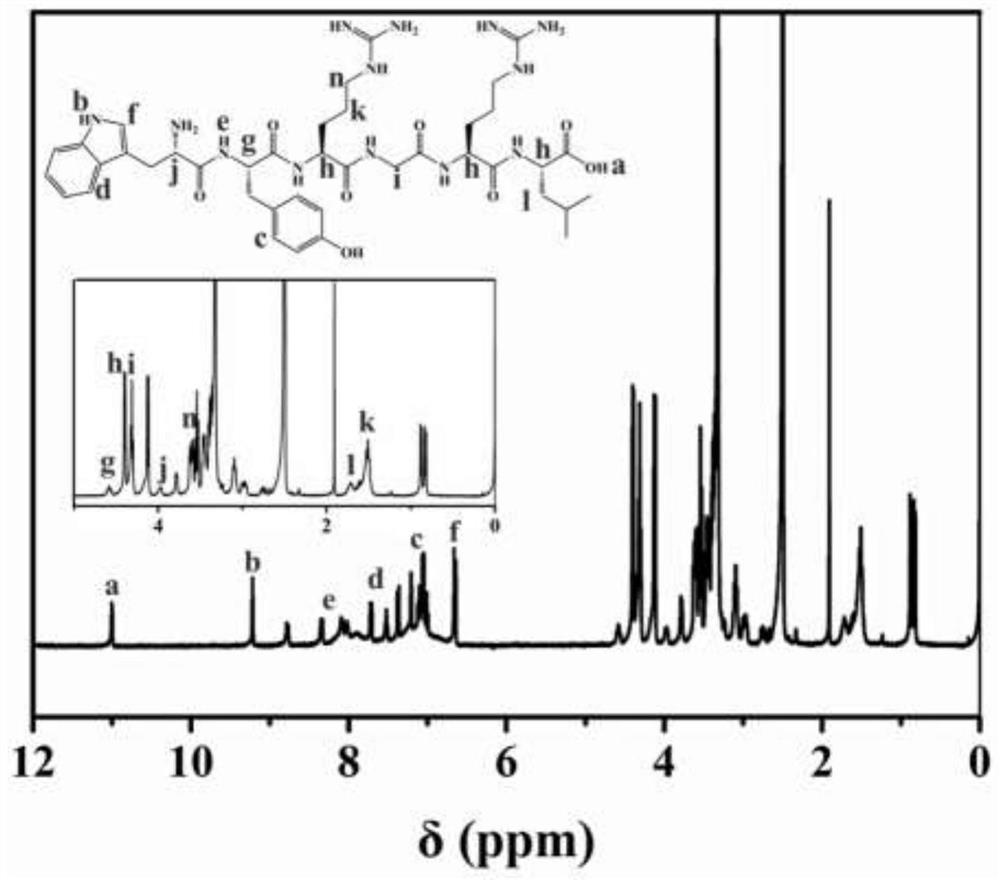
[0046] (2) Preparation of type II collagen-binding peptide modified sulfobetaine hydrogel
[0047] Soak the functionalized sulfobetaine hydrogel prepared in step 1 in PBS buffer, and change the water every 6 hours until the unreacted monomers are completely removed. Meanwhile, the WYRGRL polypeptide wa...
Embodiment 3
[0050]In this example, the synthesis scheme of the active ester monomer and the preparation of the hydrogel are basically the same as in Example 1 and Example 2, the difference is that the sulfobetaine monomer in Example 2 is replaced by phosphorylcholine Monomer, to prepare a zwitterionic hydrogel with the ability to recruit type II collagen, denoted as pMPC@PEP. Wherein, the chemical structure of the zwitterionic monomer used as the main structure of the hydrogel is shown in formula (III):
[0051]
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More