Purified hepatitis C virus envelope proteins for diagnostic and therapeutic use
a technology of hepatitis c virus and envelope proteins, which is applied in the direction of viruses, peptides/protein ingredients, fungi, etc., can solve the problems of much more expensive manufacturing of hbsag from mammalian cells compared with yeast-derived hepatitis b vaccines
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
example 2
Construction of HCV Recombinant Plasmids
[0306] 2.1. Constructs Encoding Different Forms of the E1 Protein
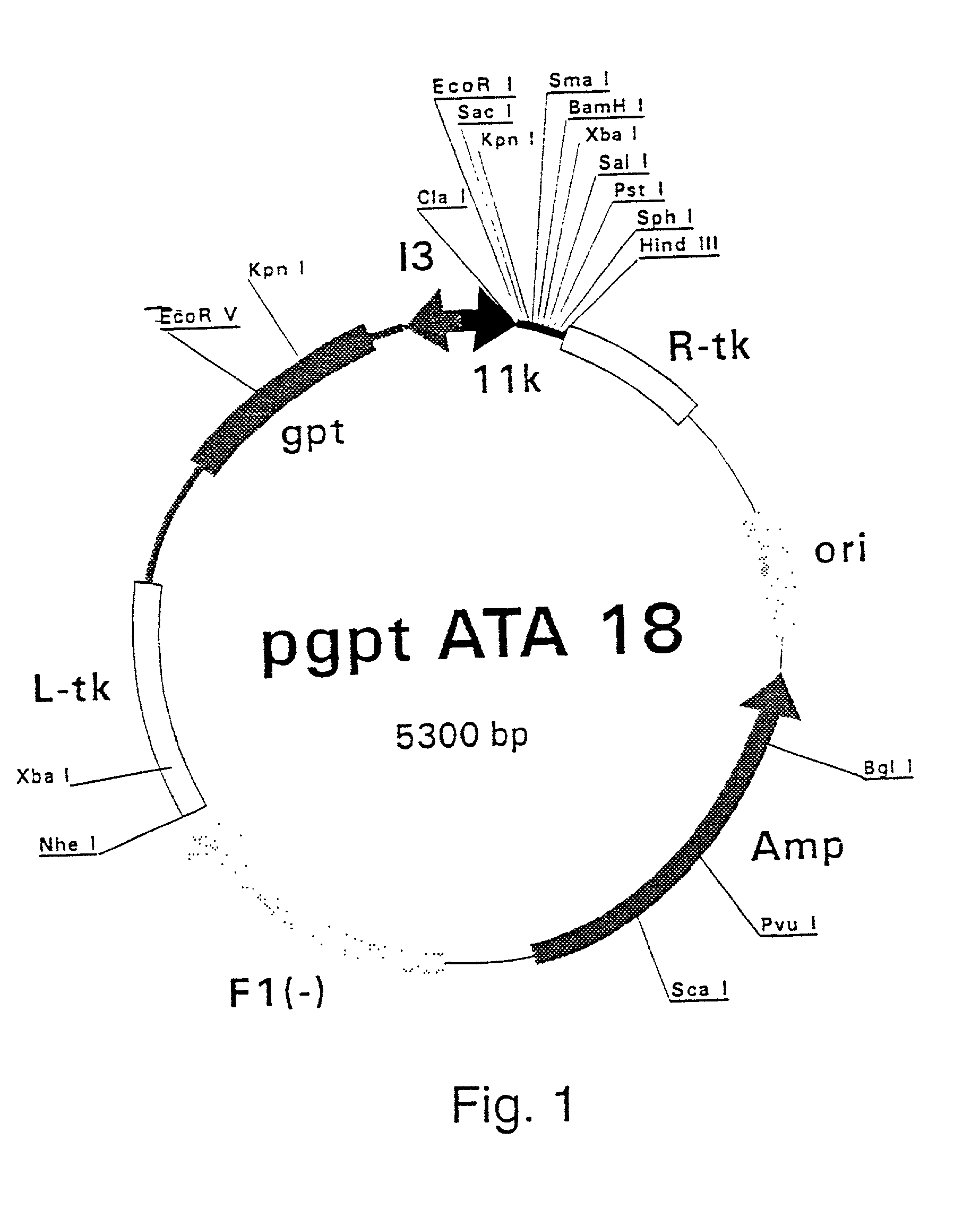
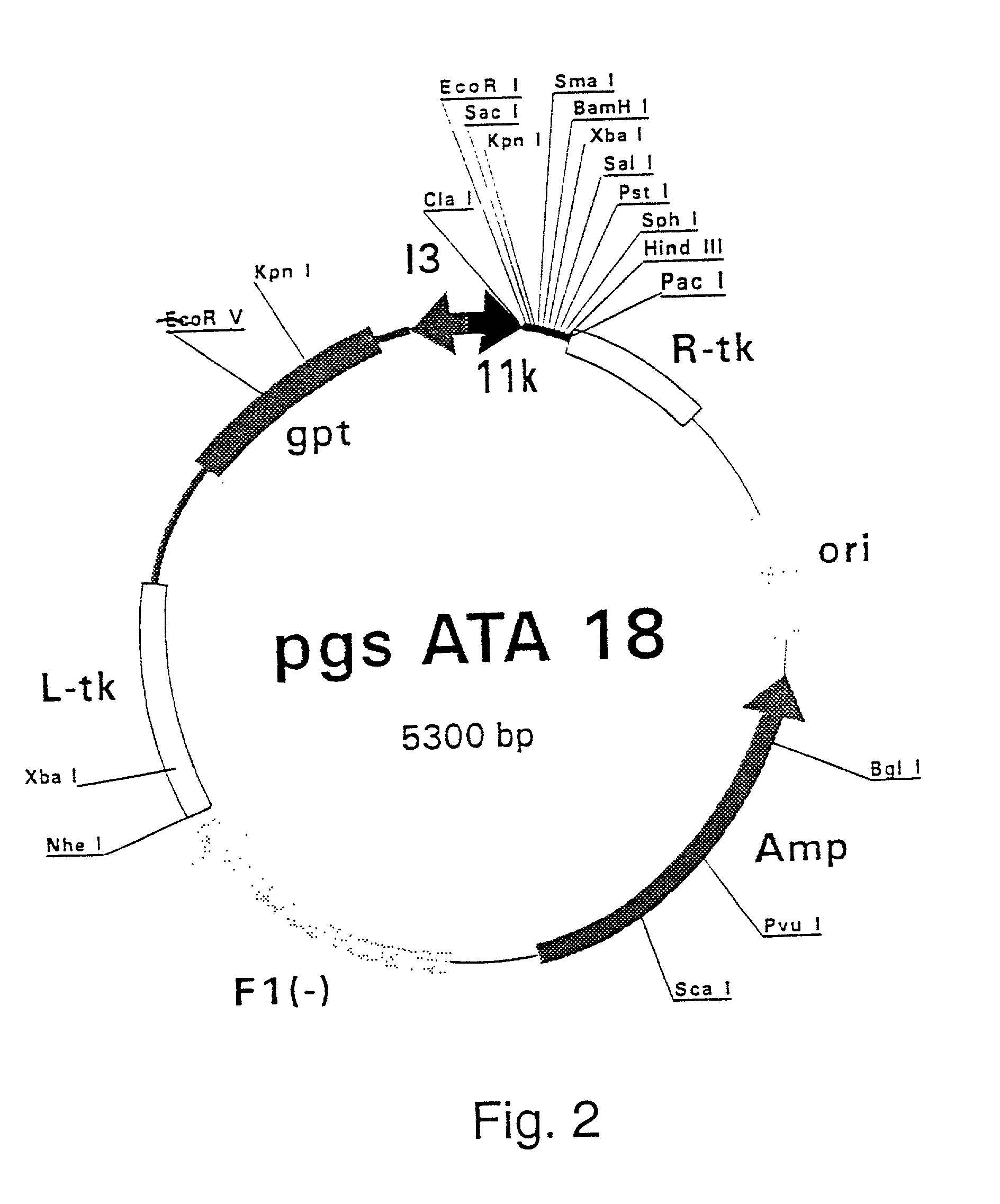
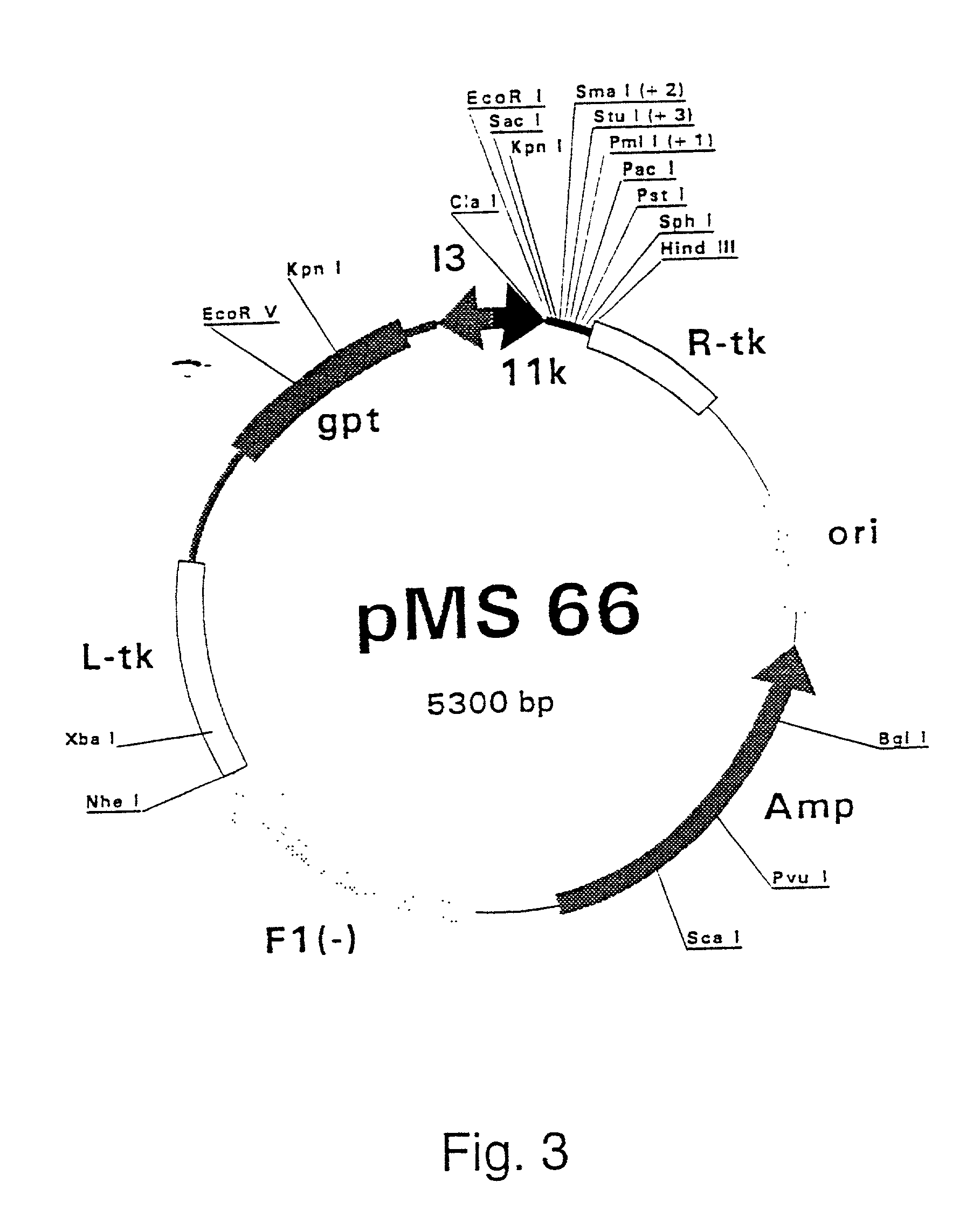
[0307] Polymerase Chain Reaction (PCR) products were derived from the serum samples by RNA preparation and subsequent reverse-transcription and PCR as described previously (Stuyver et al., 1993b). Table 1 shows the features of the respective clones and the primers used for amplification. The PCR fragments were cloned into the Sma I-cut pSP72 (Promega) plasmids. The following clones were selected for insertion into vaccinia recombination vectors: HCCl9A (SEQ ID NO 3), HCCl10A (SEQ ID NO 5), HCCl11A (SEQ ID NO 7), HCCl12A (SE: ID NO 9), HCCl13A (SEQ ID NO 11) and HCCl17A (SEQ ID NO 13) as depicted in FIG. 21. cDNA fragments containing the E1-coding regions were cleaved by EcoRI and HindIII restriction from the respective pSP72 plasmids and inserted into the EcoRI / HindIII-cut pgptA-18 vaccinia recombination vector (described in example 1), downstream of the 11K vaccinia virus late p...
example 3
Infection of Cells with Recombinant Vaccinia Viruses
[0317] A confluent monolayer of RK13 calls was infected at a m.o.i. of 3 with the recombinant HCV-vaccinia viruses as described in example 2. For infection, the cell monolayer was washed twice with phosphate-buffered saline pH 7.4 (PBS) and the recombinant vaccinia virus stock was diluted in MEM medium. Two hundred .mu.l of the virus solution was added per 10.sup.6 cells such that the m.o.i. was 3, and incubated for 45 min at 24.degree. C. The virus solution was aspirated and 2 ml of complete growth medium (see example 2) was added per 10.sup.6 cells. The cells were incubated for 24 hr at 37.degree. C. during which expression of the HCV proteins took place.
example 4
Analysis of Recombinant Proteins by Means of Western Blotting
[0318] The infected cells were washed two times with PBS, directly lysed with lysis buffer (50 mM Tris.HCl pH 7.5, 150 mM NaCl, 1% Triton X-100, 5 mM MgCl.sub.2, 1 .mu.g / ml aprotinin (Sigma, Bornem, Belgium)) or detached from the flasks by incubation in 50 mM Tris.HCL pH 7.5 / 10 mM EDTA / 150 mM NaCl for 5 min, and collected by centrifugation (5 min at 1000 g). The cell pellet was then resuspended in 200 .mu.l lysis buffer (50 mM Tris.HCL pH 8.0, 2 mM EDTA, 150 mM NaCl, 5 mM MgCl.sub.2 aprotinin, 1% Triton X-100) per 10.sup.5 cells. The call lysates were cleared for 5 min at 14.000 rpm in an Eppendorf centrifuge to remove the insoluble debris. Proteins of 20 .mu.l lysate were separated by means of sodium dodecyl sulphate-polyacrylamide gel electrophoresis (SDS-PAGE). The proteins were then electro-transferred from the gel to a nitrocellulose street (Amersham) using a Hoefer HSI transfer unit cooled to 4.degree. C. for 2 hr at...
PUM
| Property | Measurement | Unit |
|---|---|---|
| molecular weight | aaaaa | aaaaa |
| molecular weight | aaaaa | aaaaa |
| pH | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More