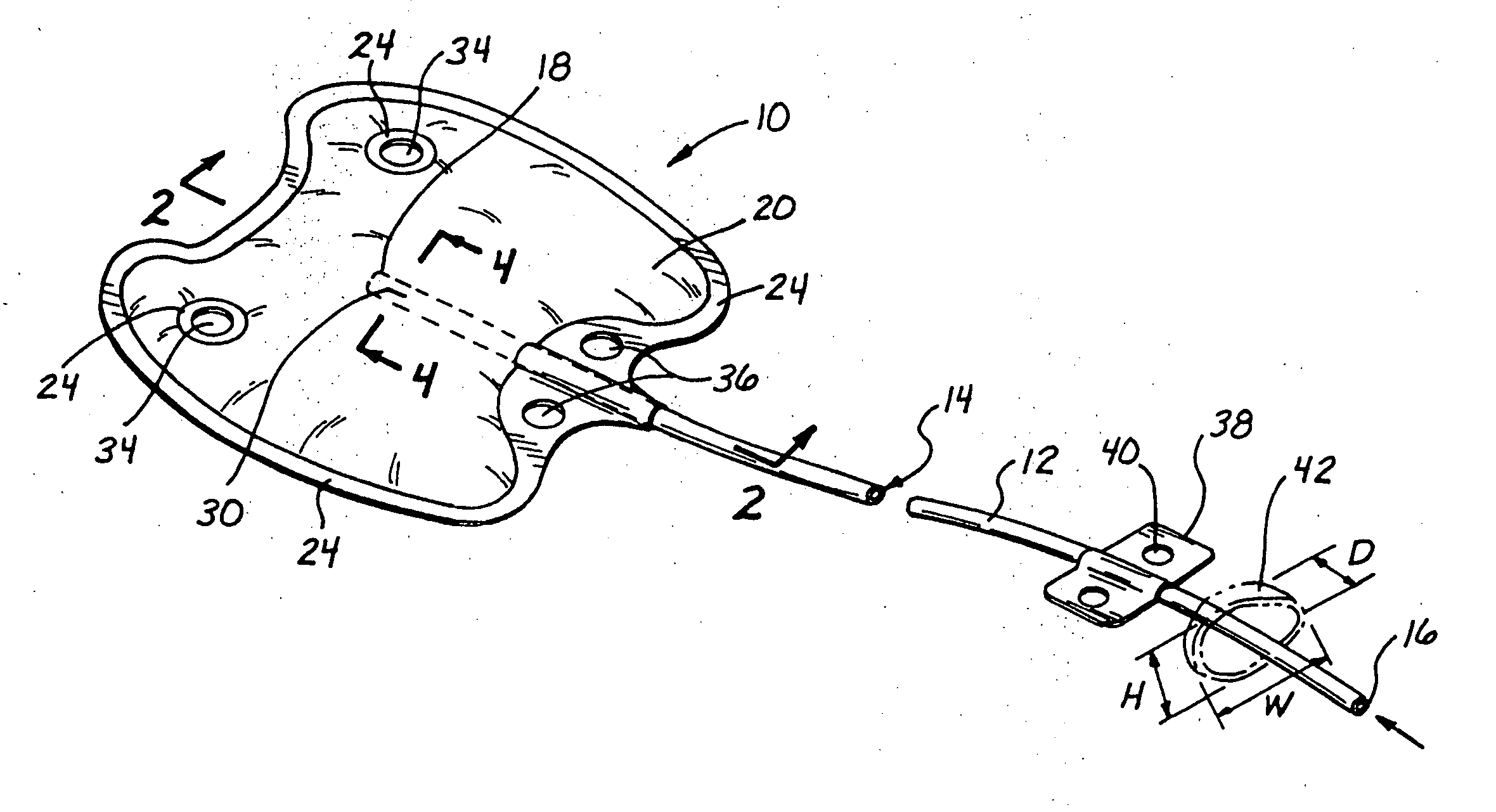
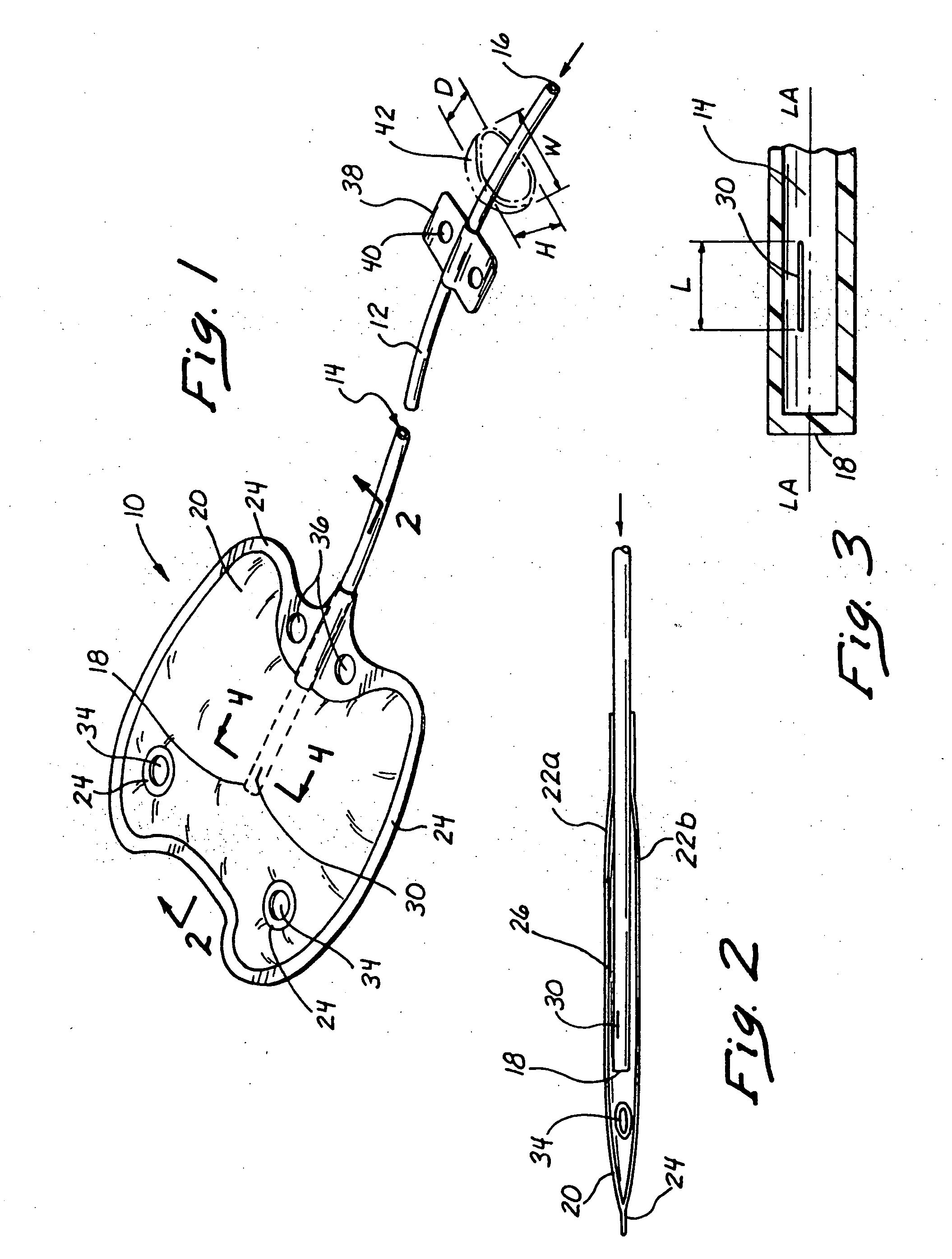
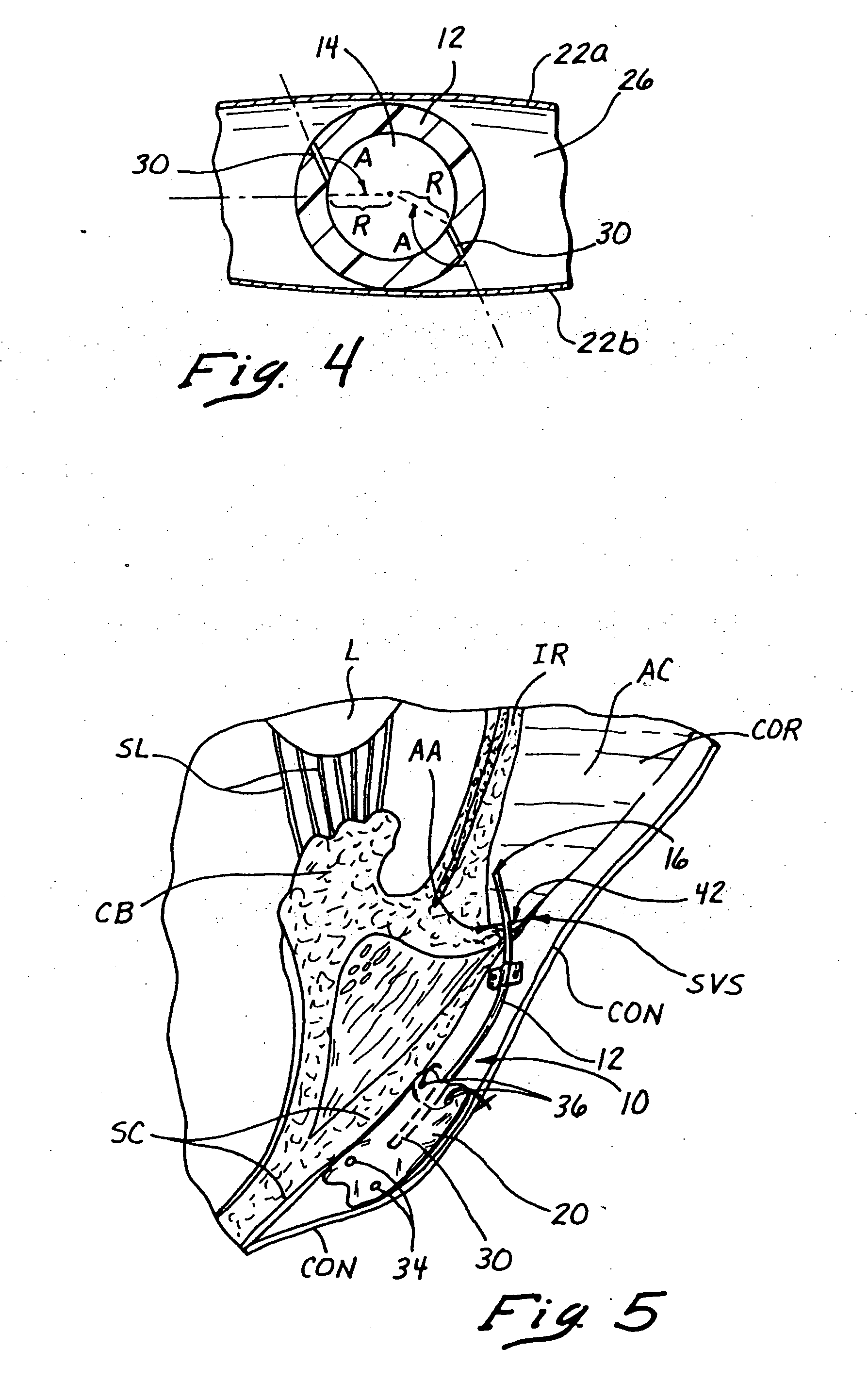
In general,
glaucoma results from a defect in the functional drainage
system, whereby naturally occurring endogenous fluid (e.g.,
aqueous humor) is drained from the interior of the eye.
“chronic simple
glaucoma”“simple
glaucoma”, “wide-angle glaucoma) the angle of the anterior chamber remains normal, but the drainage of
aqueous humor from the anterior chamber is impeded or blocked by other means, such as
edema or swelling of the trabecular spaces, abnormal
pigment dispersion, or non-perforating injury to the eye resulting in vascular congestion.
However, these various
drug therapies for glaucoma are sometimes associated with significant untoward effects, including headache,
blurred vision, allergic reactions,
retinal detachment, phacodinesis, histological changes within the eye and potential interactions with other drugs.
The major problems associated with these surgical
filtration procedures stem from the size of the opening or hole made into the anterior chamber.
Such proliferation of
connective tissue tends to close off the surgically-formed opening into the anterior chamber, thereby deterring or preventing the desired
filtration of
aqueous humor into the subconjunctival space.
The major
disadvantage associated with the use of implantable shunts for treatment of glaucoma is that, in the immediate
post operative period, the shunt may facilitate excessive fluid drainage which results in hypotony within the anterior chamber, flattening of the anterior chamber and potential choroidal detachment and / or phthisis bulbi.
Such excessive post-operative fluid outflow may also result in expansion of the fibrous
capsule located beneath the rectus muscles of the eye.
Such expansion of the fibrous
capsule can stretch and tighten the rectus muscles, thereby inducing heterotropia and impairing the
motility of the eye in the quadrant wherein the
implant is located.
Additionally, due to the size of some of these shunt devices, the bulky presence of the device itself within the subconjunctival space can cause scleral
erosion, changes in the natural curvature of the eye, or damage to adjacent vasculature and tissue.
Other problems associated with the use of implantable shunt devices for the treatment of glaucoma involve friction and wear imparted by the implanted
shunt device,
irritation of the iris
endothelium caused by
insertion of the
shunt device into the anterior chamber, and migration of microbes, cells, proteins or other matter through the lumen of the
shunt device and into the anterior chamber of the eye.
Also, the
surgical procedures used to
implant the prior art fluid
shunting devices have typically been laborious in nature and have typically required that suturing of the fluid shunting device to the surrounding tissue of the host, to hold the fluid shunting device at its desired location within the eye.
The installation of sutures to anchor the implanted fluid shunting device is
time consuming and, in cases where such sutures are not properly placed, can result in undesirable tugging, traction or stress on the surrounding tissue and / or disconfiguration of the
implanted device.
Also, the installation of such sutures can result in unintentional, iatrogenic perforation of the anterior or posterior chabber of the eye, with
resultant leakage of aqueous or vitreous humor and / or
resultant cellular ingrowth and opacification of the aqueous and / or vitreous humor.
One complication associated with the use of implantable shunt devices to drain fluid from body cavities is that proteins, cellular matter, or other debris may block the lumen of the shunt tube thereby interfering with the drainage of fluid through the tube.
Also, proliferation of tissue or blebs may compress, collapse, or block the shunt tube.
Moreover,
pathogenic microorganism or irritating proteins or other matter may migrate through the lumen of the shunt tube into the affected body cavity in a manner which can lead to iatrogenic infection,
irritation or
inflammation of the affected body cavity.
Given the above-summarized limitations and drawbacks associated with the implantable fluid-shunting devices of the prior art, it is apparent that no single fluid-shunting device has proven to be optimal for all applications.
 Login to View More
Login to View More  Login to View More
Login to View More