Plant-derived alkaline alpha-galactosidase
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
examples
Materials and Experimental Methods
[0097] Cultivation of Plants and Preparation of Samples:
[0098] Plants of muskmelon (Cucumis melo L.cv C-8) were grown under standard conditions in a greenhouse in Beit Dagan, Israel. Female flowers were hand pollinated and tagged at anthesis and fruit load was limited to 1 fruit per plant after 10 DAA (days after anthesis). For the study of fruit development, primary fruits were harvested from 3 days prior to anthesis, at anthesis and 1, 2, 4, 6, 10, 14, 20, 30 and 45 days after anthesis (DAA). Whole fruits before 6 DAA, and the inner mesocarp of the fruit tissues after 10 DAA, were thinly sliced and immediately frozen in liquid nitrogen prior to storage at −80 C. Chemicals and enzymes, unless specified otherwise, were purchased from Sigma and Boehringer, Mannheim, Germany.
[0099] Germinating barley seeds were prepared by imbibing 340 seeds of barley (H. vulgare var. Himalaya) on moist filter paper at 25° C. for 24 hours. The embryos were carefull...
example i
Purification of Alpha-Galactosidases from Melon Fruit
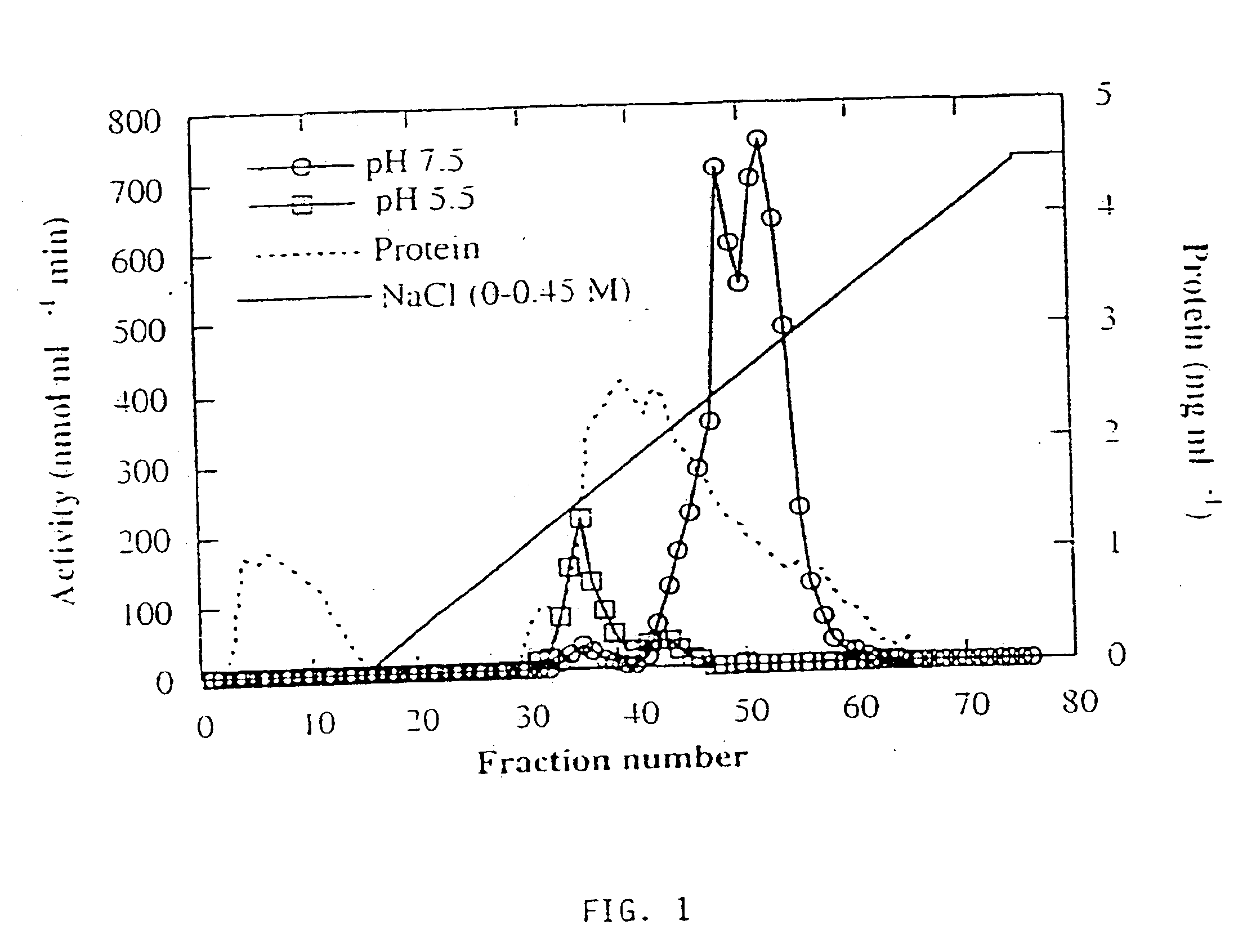
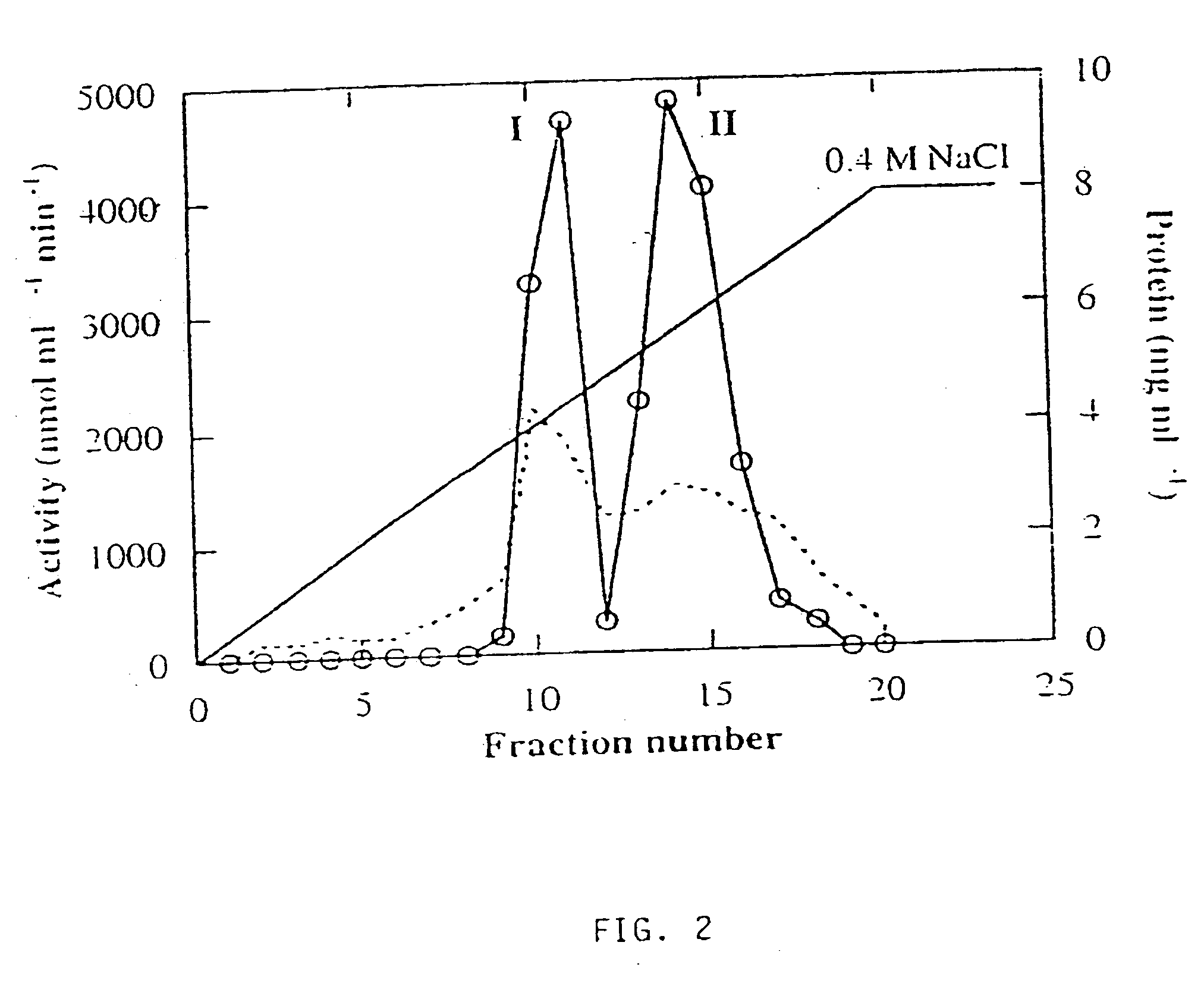
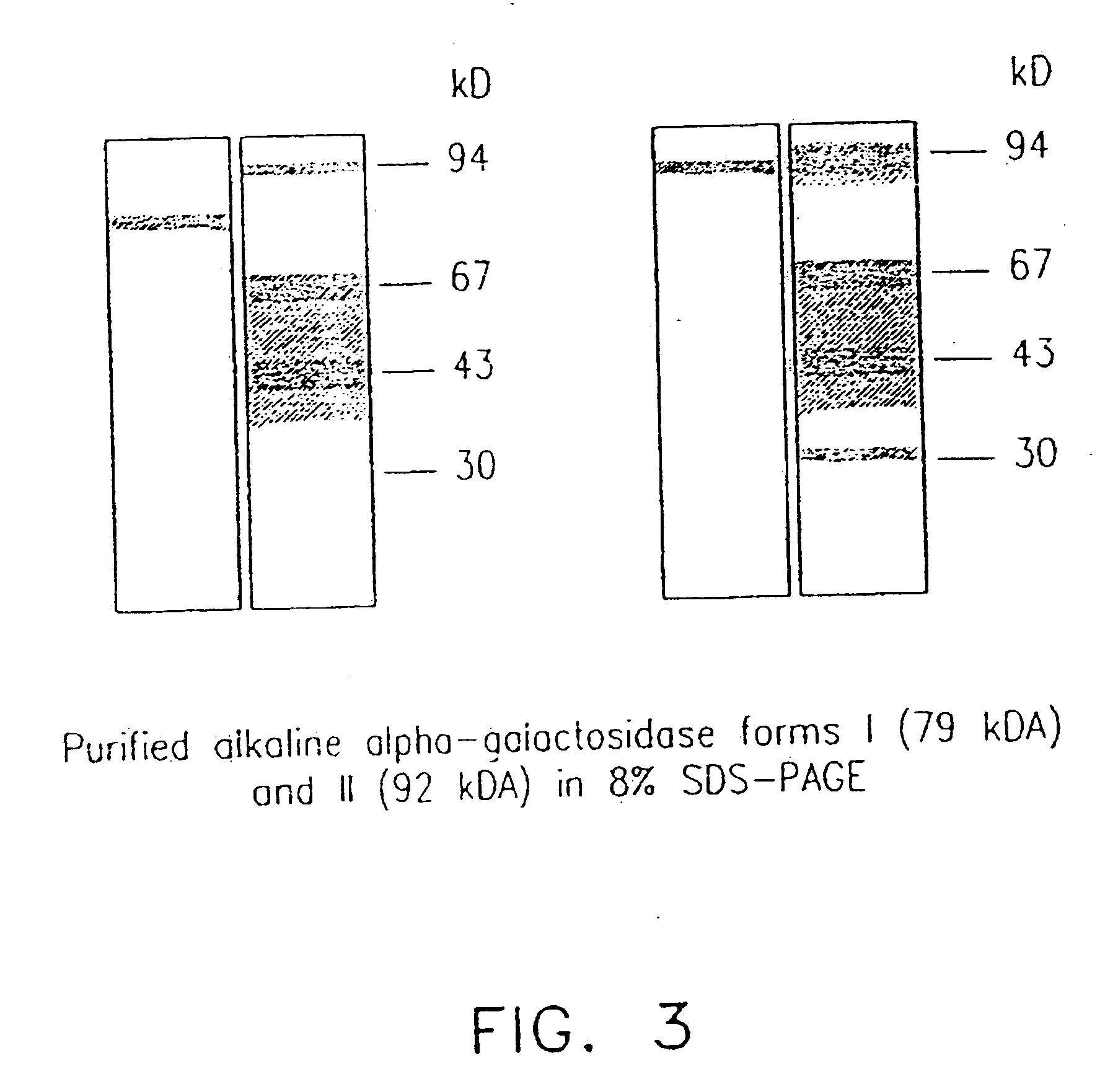
[0126] Three forms of alpha-galactosidase were resolved from young melon fruit mesocarp by DEAE-Sepharose ion exchange chromatography, in conjunction with Mono-Q chromatography, using pNPG as substrate (FIGS. 1 and 2). The first peak showed higher activity at pH 5.5 than at pH 7.5, while the latter two peaks both showed activity at pH 7.5 with little activity at pH 5.5. Accordingly, we referred the first peak as an acid form of alpha-galactosidase and the other two peaks as alkaline alpha-galactosidases Form I and Form II, respectively. The three enzyme forms were partially purified for the purpose of characterization (Table 1). Mono-Q ion exchange successfully resolved the two alkaline forms, and hydrophobic interaction chromatography was useful in the purification of alkaline Form II. After further purification, as described in Table 1, the two alkaline forms were electrophoresed on a denaturing SDS-PAGE gel and a photograph of...
example 2
Changes of Acid and Alkaline Alpha-Galactosidases During Melon Fruit Development
[0138] The substrate preferences (Table 2) and pH profiles (FIG. 6A) from the purified acid and Form I and Form II alkaline alpha-galactosidases allowed us to measure and estimate their activities even in crude extracts of melon fruit, using their natural substrates. Very little overlap in activity occurs between pH 5.5 and pH 7.5 (FIG. 4A) and, at pH 7.5, the activities of alkaline alpha-galactosidase I and II in the crude extracts could be distinguished by their respective activities when using raffinose or stachyose as substrate. The activity with raffinose at pH 7.5 is a good indicator of Form I activity since Form II is relatively specific for stachyose. There should be an overestimation of Form II activity when using stachyose due the hydrolysis of this substrate by Form I which is also present in the crude extract. Nevertheless, distinct developmental patterns of alpha-galactosidase activities ar...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Mass | aaaaa | aaaaa |
| Mass | aaaaa | aaaaa |
| Mass | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More