Antibodies for ubiquitinated proteins
a technology of ubiquitinated proteins and antibodies, which is applied in the field of antibodies for ubiquitinated proteins, can solve the problems of insufficient information on how to manipulate ubiquitination and modulate some processes involving ubiquitination, and the difficulty of elucidating the proteins targeted by ubiquitination. and other problems, to achieve the effect of expanding our understanding and facilitating the identification of ubiquitinated proteins
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
EXAMPLE 1
Materials and Methods
[0116]The Example illustrates some of the materials and methods used in developing the subject matter of the claims.
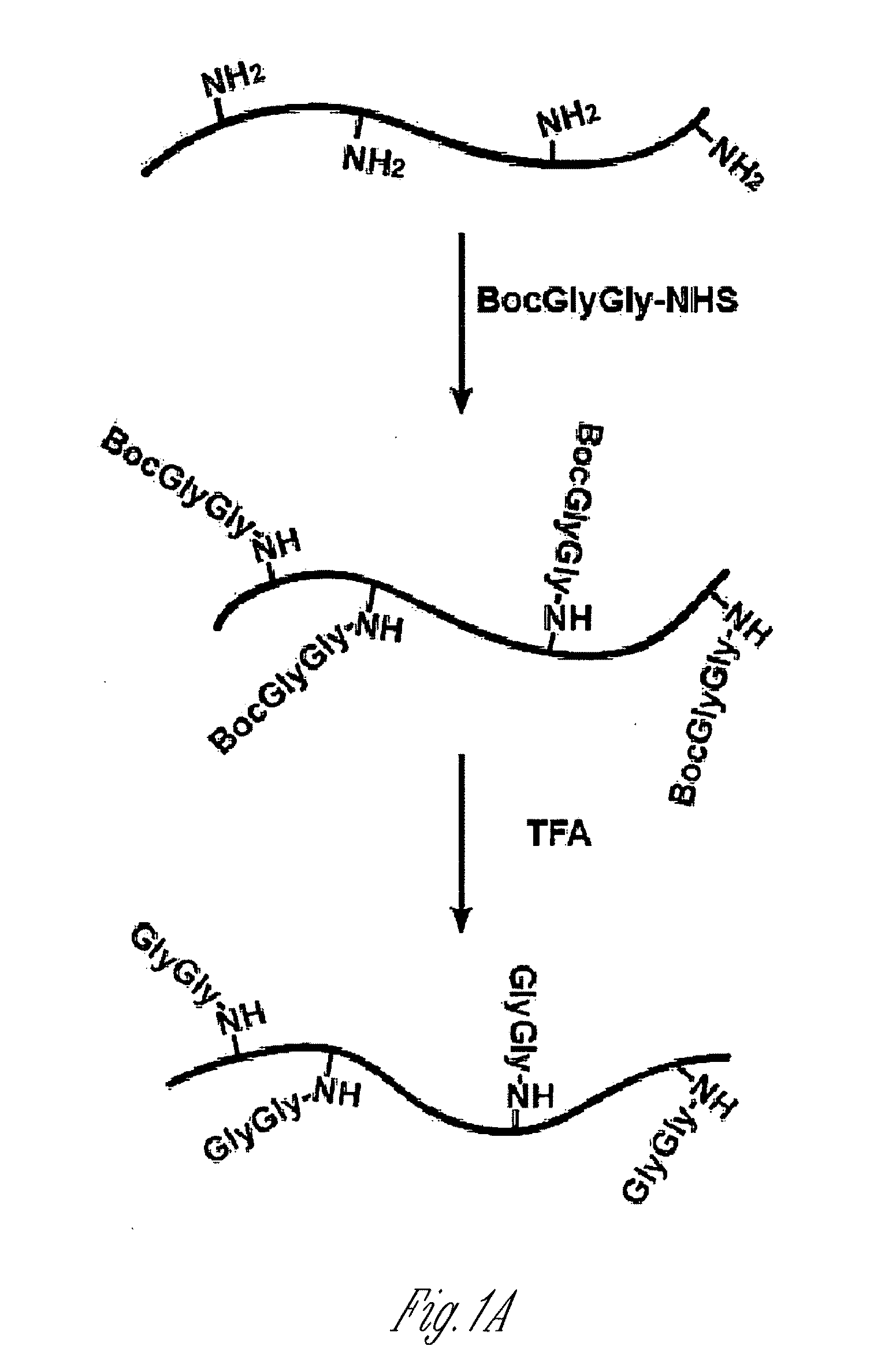
[0117]Antigen design and antibody purification. Lysine-rich histone (10 mg, sample A) from calf thymus (type III-S, Sigma) was dissolved in 100 mM NaHCO3 buffer (10 ml) at pH 10. 50 mM t-butyloxycarbonyl-Gly-Gly-N-hydroxysuccinimide (500 μl, Boc-Gly-Gly-NHS; Derrien D., et al., Glycoconj J. 1989, 6, 241-55) in DMSO was added to histone solution and the reaction was carried out at room temperature for 1 h by shaking on a plate rotator. This step was repeated three additional times and sample B was obtained. For deprotection of the Boc group, neat trifluoroacetic acid (6 ml, TFA, Sigma) was added and the solution was shaken for 2 h at room temperature. The reaction was neutralized and stopped by adding 10 M NaOH dropwise on ice (sample C). All of sample C and part of sample A and sample B were dialyzed four times against 20 mM acetic acid f...
example 2
EXAMPLE 2
Identifying Ubiquitinated Proteins and Ubiquitination Sites
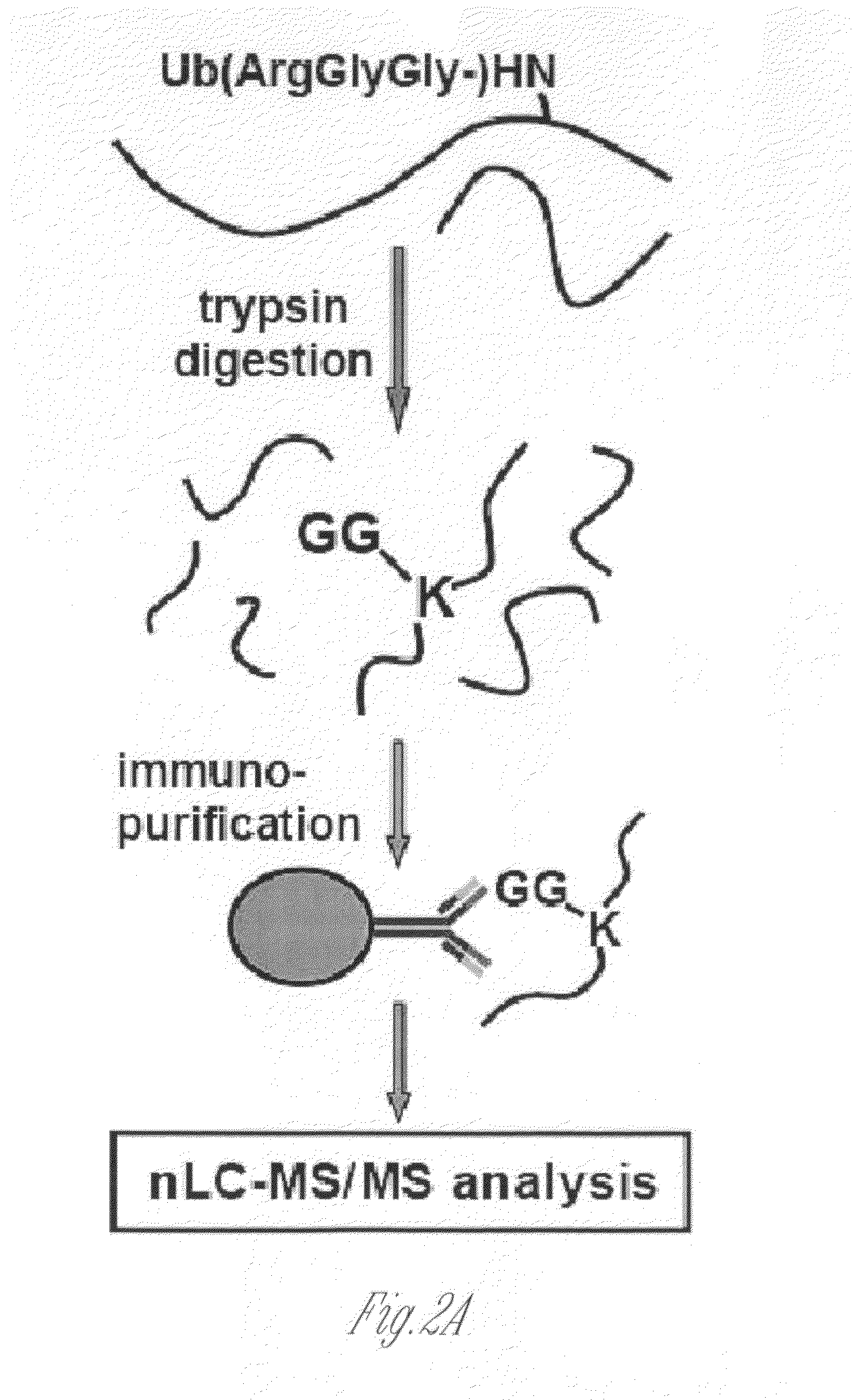
[0132]This Example illustrates novel methods for identifying ubiquitinated proteins and ubiquitination sites using an antibody that selectively binds to the diglycine remnant in peptides generated from tryptic digestion of biological samples. Using this immunoaffinity approach coupled to nano LC-MS / MS, more than 300 ubiquitinated proteins and nearly 400 ubiquitination sites were identified. Of these ubiquitinated proteins, 224 have not previously been known to be ubiquitinated. These experiments illustrate that the immunoaffinity profiling methods described herein have broad utility in characterizing the occurrence and extent of ubiquitination in diverse tissues and disease states.
[0133]To generate an antibody that recognizes peptides containing the ubiquitin remnant, a protein antigen was prepared that contained one or more diglycine adducts the terminal amine of the side chain of lysines naturally present in the ...
example 3
EXAMPLE 3
Anti Diglycyl-Lysine Antibodies can be Used to Identify Ubiquitinated Ubiquitin, and the Specific Ubiquitin-Ubiquitin Linkages
[0151]Ubiquitin is often present as polyubiquitin chains, with multiple ubiquitins attached in a chain or other branched configuration on proteins. The C-terminus of ubiquitin is conjugated to other ubiquitins via one of its seven lysine residues. The antibodies described here can be used to detect the presence or abundance of specific ubiquitin chains, due to the detection of ubiquitin-specific peptides.
[0152]FIG. 12 shows a MS / MS spectrum of a fragment from ubiquitin, containing a ubiquitin remnant diglycine on K48. This peptide can be detected with either the polyclonal or monoclonal anti-diglycyl-lysine antibodies. In this case, the antibody was immobilized to protein A or protein G beads. Cellular proteins were extracted from HEK293 cells, digested by trypsin, and immunoprecipitated by GlyGly-antibody and detected on ion-trap or Q-TOF LC-MS / MS....
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More