Expression and assembly of human group c rotavirus-like particles and uses thereof
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
example 1
[0202]Cloning and construction of baculovirus recombinants. Segment 5, encoding VP6, from human group C RV strain S-1 was amplified by RT-PCR using BMJ44 (5′-AGC-CAC-ATA-GTT-CAC-ATT-TC-3′) (SEQ ID NO: 14) and BMJ141 (5′-ATC-TCA-TTC-ACA-ATG-GAT-G-3′) (SEQ ID NO: 15) (28). Segment 8, encoding VP7, from strain S-1 was amplified by RT-PCR using primers BMJ13 (5′-AGC-CAC-ATG-ATC-TTG-TTT-3′) (SEQ ID NO: 20) and BMJ14 (5′-GGC-ATT-TAA-AAA-AGA-AGA-3′) (SEQ ID NO: 21) (13, 28). Segment 2, encoding VP2, from strain ASP88 was amplified by RT-PCR using BMJ197 (5′-TCG-AGG-ACA-AAT-CGT-CCA-AG-3′) (SEQ ID NO: 22) and BMJ180 (5′-AGC-CAC-AGA-GTT-TGA-GGT-C-3′) (SEQ ID NO: 23). Cloning and construction of recombinant baculovirus expressing S-1 VP7 was previously described (14). DNA fragments of segment 2 and 5 were cloned into vector pVL1393 and transfections were performed with the Bac-N-Blue transfection kit (Gibco, Grand Island, N.Y.). Baculovirus constructs were amplified in Spodoptera frugiperda 9 ...
example 2
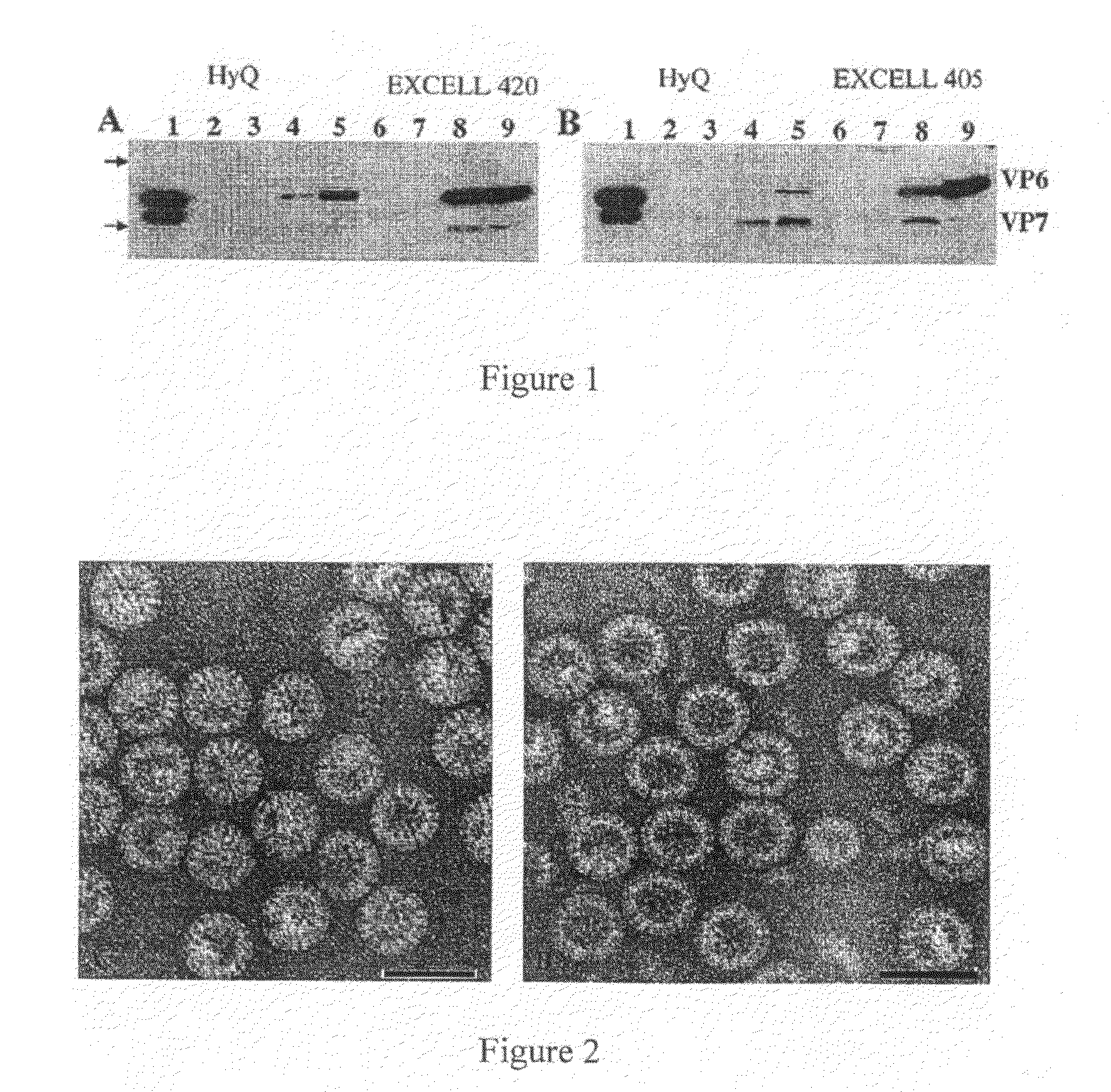
[0208]Cells and superinfections. Sf9 or High Five (Hi5) insect cells were grown and maintained in EX-CELL 420 or 405 media (Sigma, Lenexa, Kans.) or HyQ SFX-INSECT media in shaker flasks at 27° C. Sf9 and Hi5 cells were subcultured every 3 or 4 days at a concentration of 1×106 cells / ml and 5×105 cells / ml, respectively. Stationary superinfections were performed by seeding Sf9 cells in HyQ or EX-CELL 420 and Hi5 cells in HyQ or EX-CELL 405 into a T150 flask at a concentration of 3×105 cells / ml. Baculovirus constructs (rVP2, rVP6 and rVP7) were added at a multiplicity of infection (MOI) of 1 each. Infections were carried out without proteinase inhibitors and infected cultures were harvested at day 5. Large scale VLP production was performed in suspension culture by seeding Sf9 cells in EX-CELL 420 into fernbach flasks at a concentration of 1×106 cells / ml. Baculovirus recombinants were added one day later at an MOI of 1.4 each and harvested on day 4.
example 3
[0209]Western blot. Sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) was carried out with 12% separating and 5% stacking gels using the Laemmli discontinuous buffer system (16). Samples were heated at 97° C. for 5 min with 10% β-mecaptoethanol prior to loading and then electrophoresed. Proteins were transferred to a PVDF membrane in transfer buffer (25 mM Tris, 192 mM glycine, 10% methanol). After blocking with 10% (for unpurified GpC RV proteins) blotto in PBS-T for 1-2 hrs at room temperature or 15% (for purified GpC RV proteins) blotto in PBS-T overnight at 4° C., membranes were incubated with porcine hyperimmune serum (1:2,000) to Cowden in 5% blotto in PBS-T overnight at 4° C. or rabbit hyperimmune serum (1:20,000) to human GpC VLPs in 10% blotto in PBS-T for 1 h. Membranes were washed in PBS-T, incubated with horseradish peroxidase (HRP) goat anti-pig (1:142,000) (KPL, Gaithersburg, Md.) in 5% blotto or HRP-goat anti rabbit (1:20,000) (Pierce, Rockford, Ill...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Fraction | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More