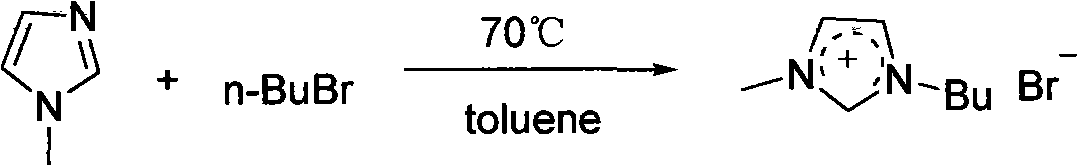
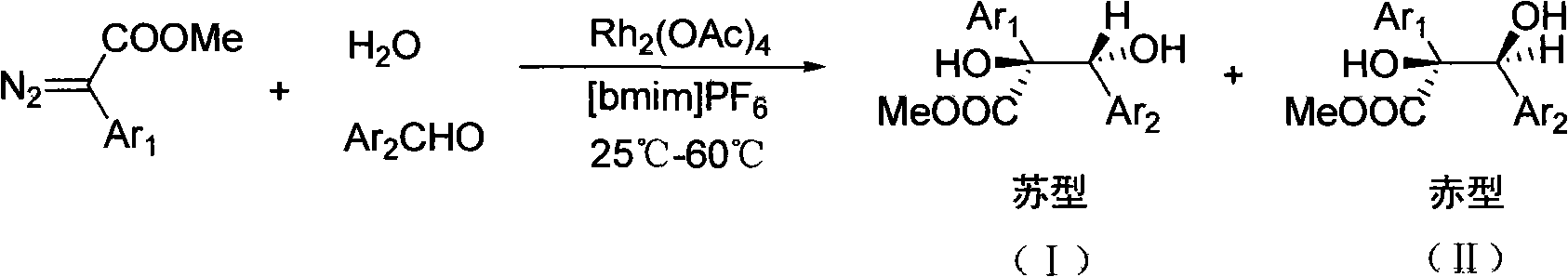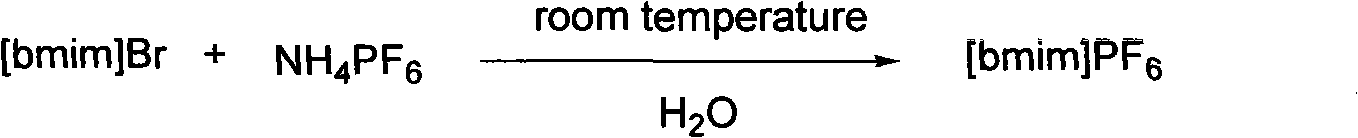Preparation for alpha, beta-di-hydroxyl carboxylic ether compound
A bishydroxycarboxylate and compound technology, applied in the field of organic synthesis, can solve problems such as heavy metal rhodium cannot be recycled, environmental pollution, etc., and achieve the effects of broad prospects for industrial production, short process routes, and mild reaction conditions
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0025] Take a 25mL two-necked bottle, add p-nitrobenzaldehyde 0.1511g (1mmol) and 0.01mmol of Rh 2 (OAc) 4 . Subsequently, 5 mL of the above-prepared ionic liquid [bmim]PF was added 6 , stir well, and after the substrate is completely dissolved, add 3 mL of water. Continue to stir and heat to 60°C. Dissolve 0.352 g (2 mmol) of methyl diazophenylacetate in 3 mL of the above-mentioned ionic liquid, and add it dropwise to the system through a sample injector, and the sample injection is completed in 3 hours. Continue to react for 3h, stop the reaction. Separate the aqueous phase, extract the ionic liquid five times with 50 mL of ether, concentrate the extraction solution, and use ethyl acetate / petroleum ether=1 / 5 as the developing agent to separate the product through a column to obtain the product 2,3-dihydroxy-3-(4 -Nitrophenyl)-2-phenylpropionic acid methyl ester, 0.2379 g of α, β-dihydroxycarboxylate compound, yield 75%.
Embodiment 2
[0027] Take a 25mL two-necked bottle, add m-nitrobenzaldehyde 0.1512g (1mmol) and 0.02mmol of Rh 2 (OAc) 4 . Subsequently, 10 mL of the above-prepared ionic liquid [bmim]PF was added 6 , stir well, and after the substrate is completely dissolved, add 1 mL of water. Continue to stir and react at room temperature at 25°C. Dissolve 0.264 g (1.5 mmol) of methyl diazophenylacetate in 4 mL of the above-mentioned ionic liquid, and add it dropwise to the system through a sample injector, and the sample injection is completed in 3 hours. Continue to react for 2h, stop the reaction. Separate the aqueous phase, extract the ionic liquid five times with 50 mL of ether, concentrate the extraction solution, and use ethyl acetate / petroleum ether=1 / 5 as the developing agent to separate the product through a column to obtain the product 2,3-dihydroxy-3-(3 -Nitrophenyl)-2-phenylpropionic acid methyl ester, 0.2220 g of α, β-dihydroxycarboxylate compound, yield 70%.
Embodiment 3
[0029] Take a 25mL two-necked bottle, add o-nitrobenzaldehyde 0.1512g (1mmol) and 0.05mmol of Rh 2 (OAc) 4 . Subsequently, 5 mL of the above-prepared ionic liquid [bmim]PF was added 6, stir well, and after the substrate is completely dissolved, add 2 mL of water. Continue to stir and heat to 60°C. Dissolve 0.317 g (1.8 mmol) of methyl diazophenylacetate in 3 mL of the above-mentioned ionic liquid, and add it dropwise to the system through a sample injector, and the sample injection is completed in 3 hours. Continue to react for 5h, stop the reaction. Separate the aqueous phase, extract the ionic liquid five times with 50 mL of ether, concentrate the extraction solution, and use ethyl acetate / petroleum ether=1 / 5 as the developing agent to separate the product through a column to obtain the product 2,3-dihydroxy-3-(2 -Nitrophenyl)-2-phenylpropionic acid methyl ester, 0.1681 g of α, β-dihydroxycarboxylate compound, yield 53%.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More