Hydrogel eye drop carrier containing polymer micelle and preparation method of hydrogel eye drop carrier
A polymer glue and polymer technology, which can be used in pharmaceutical formulations, medical preparations with non-active ingredients, and non-active ingredients of polymer compounds. The effect of oxygen permeability, great social benefit and economic benefit
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
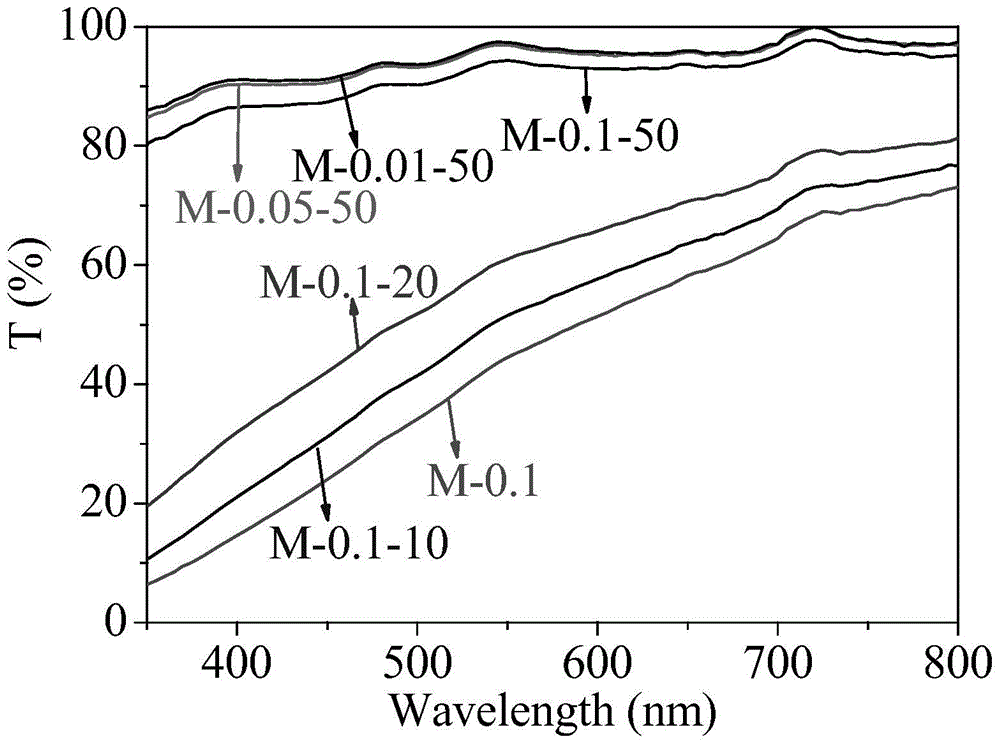
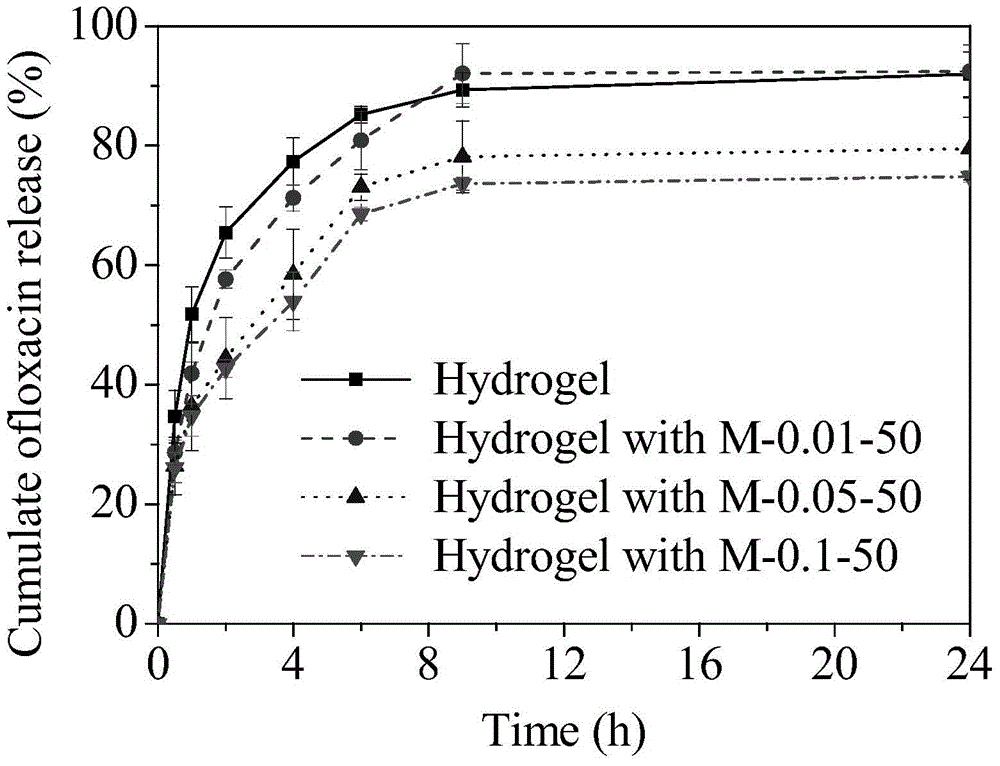
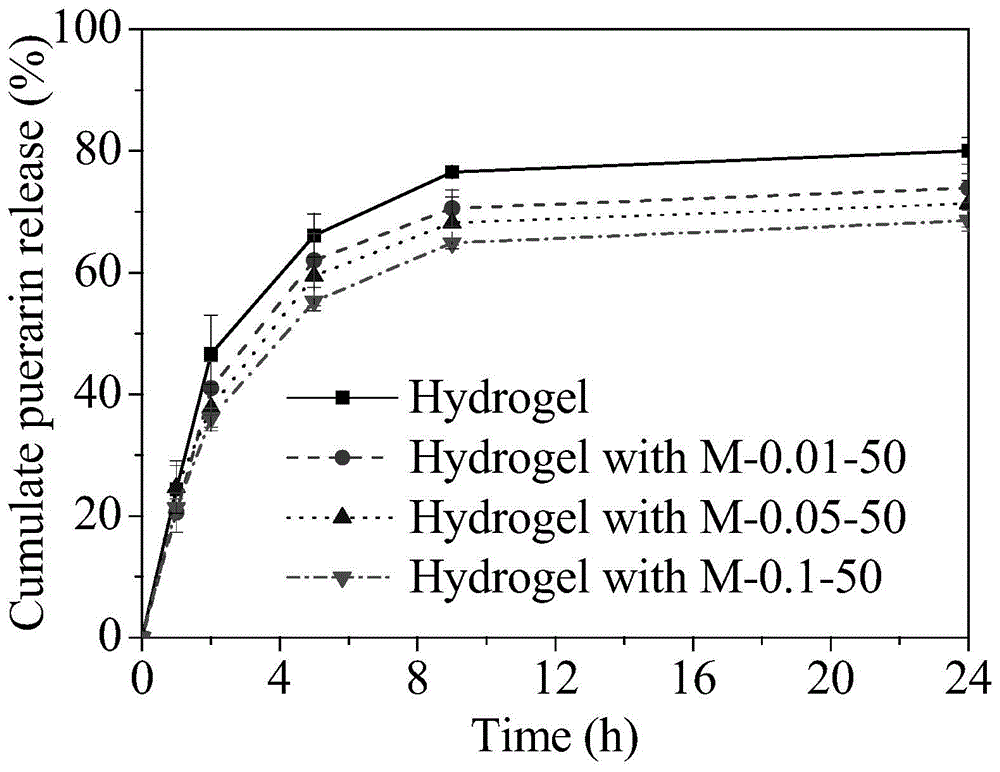
[0063] Dissolve 57.6mg of β-CD in 10 ml of ultrapure water, then add 10ml of 0.72% W / V PAA (number average molecular weight 100000, the same below) aqueous solution and 10ml 0.44% W / V PEG (number average molecular weight of 6000, below Same) aqueous solution. The resulting mixture was stirred at room temperature for 24 hours, then allowed to stand overnight. The solution containing the polymer micelles was dialyzed for 3 days with a dialysis bag with a molecular weight cut-off of 3000, and the water was changed every 4 hours to remove free β-CD in the mixed solution to obtain a polymer micelles solution. The light transmission of polymer micelles is greater than 90%, such as figure 1 M-0.1-50. The particle size is 64nm, the ζ potential is -20.1±0.8, the loading capacity of ofloxacin in the polymer micelle is 3.26 μg / ml, and the loading capacity of puerarin in the polymer micelle is 6.17 μg / ml. HEMA monomer (with a volume ratio of 2:1 to water in the total system) and 2wt% M...
Embodiment 2
[0065] Dissolve 5.8 mg of β-CD in 10 ml of ultrapure water, then add 10 ml of 0.072% W / V PAA aqueous solution and 10 ml of 0.044% W / V PEG aqueous solution. The resulting mixture was stirred at room temperature for 24 hours, then allowed to stand overnight. The solution containing the polymer micelles was dialyzed for 3 days with a dialysis bag with a molecular weight cut-off of 3000, and the water was changed every 4 hours to remove free β-CD in the mixed solution to obtain a polymer micelles solution. The light transmission of polymer micelles is greater than 95%, such as figure 1 M-0.01-50. The particle size is 39nm, the zeta potential is -29.7±0.9, and the loading capacity of puerarin in the polymer micelle is 1.15μg / ml. HEMA monomer (volume ratio of water in the total system is 2:1) and 2% MPC monomer (0.5% of the total mass of micelles liquid and monomer) were mixed, and then 35% aqueous solution of polymer micelles was added (by mixing After the total mass of the mono...
Embodiment 3
[0067] 28.8 mg of β-CD was dissolved in 10 ml of ultrapure water, and then 10 ml of 0.36% W / V PAA aqueous solution and 10 ml of 0.22% W / V PEG aqueous solution were added. The resulting mixture was stirred at room temperature for 24 hours, then allowed to stand overnight. The solution containing the polymer micelles was dialyzed for 3 days with a dialysis bag with a molecular weight cut-off of 3000, and the water was changed every 4 hours to remove free β-CD in the mixed solution to obtain a polymer micelles solution. The light transmission of polymer micelles is greater than 95%, such as figure 1M-0.05-50. The particle size is 53nm, the ζ potential is -23.3±0.9, the loading capacity of ofloxacin in the polymer micelle is 1.94 μg / ml, and the loading capacity of puerarin in the polymer micelle is 3.85 μg / ml. HEMA monomer (volume ratio of water in the total system is 2:1) and 2% MPC monomer (0.5% of the total mass of micelles liquid and monomer) were mixed, and then 35% aqueous...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Particle size | aaaaa | aaaaa |
| Particle size | aaaaa | aaaaa |
| Particle size | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com