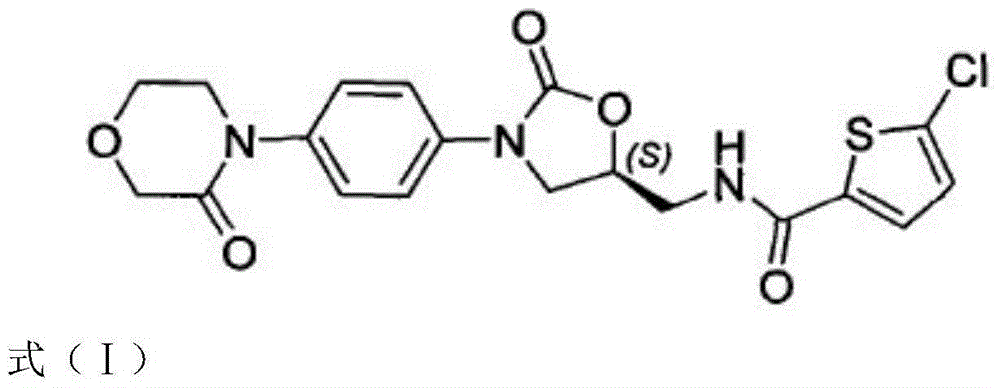
Rivaroxaban-containing composition and preparation method thereof
A technology of rivaroxaban and composition, which is applied in the field of compositions containing rivaroxaban, can solve the problems of cumbersome process, high energy consumption, and drug degradation reaction, and achieve the effect of wide application and increased dissolution
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0028] Measurement comparison implementation 1 disintegration: about 6.5 minutes;
[0029] Dissolution: Dissolution conditions: USP paddle method, 900ml sodium acetate buffer solution with pH 4.5 containing 0.5% sodium lauryl sulfate, rotating speed 75 rpm). Samples were taken at 15, 30, 45 and 60 minutes respectively, filtered, and the filtrate was taken for HPLC determination, and the cumulative dissolution percentage was calculated.
[0030] Dissolution results
[0031]
Embodiment 1
[0051] Composition of rivaroxaban / per tablet:
[0052]
[0053] Among them, rivaroxaban uses micronized crystal form I, and the particle size distribution is X 50 =3.5μm, X 90 =7.6μm
[0054] Preparation Process:
[0055] Using methods known in the pharmaceutical industry, mix rivaroxaban with β-cyclodextrin, microcrystalline cellulose, croscarmellose sodium, sodium lauryl sulfate and micronized silica gel into the hopper of a dry granulator In the process, the medicine-containing flakes are rolled and pressed at a pressure of 5-7 Mpa, and then pulverized with a 30-mesh screen to prepare granules. The obtained granules are mixed with magnesium stearate and compressed into tablets, the diameter of the tablet is 6mm, and the hardness is 60-90N. The obtained tablet is coated with a coating material comprising iron oxide, hydroxypropylmethylcellulose and polyethylene glycol.
Embodiment 2
[0057] Composition of rivaroxaban / per capsule:
[0058]
[0059] Among them, rivaroxaban uses micronized crystal form I, and the particle size distribution is X 50 =4.8μm, X 90 =9.2μm
[0060] Preparation Process:
[0061] Using methods known in the pharmaceutical industry, mix rivaroxaban with α-cyclodextrin, micronized silica gel and sodium lauryl sulfate, and put them into No. 1 capsules with a capsule filling machine.
PUM
| Property | Measurement | Unit |
|---|---|---|
| Diameter | aaaaa | aaaaa |
| Diameter | aaaaa | aaaaa |
| Diameter | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - Generate Ideas
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com