A kind of helicobacter pylori tetravalent adhesion factor multi-epitope vaccine and preparation method thereof
A Helicobacter pylori and adhesion factor technology, applied in the field of biomedicine, can solve problems such as vaccine difficulties, poor patient compliance, and enhanced Hp drug resistance, to prevent immunopathological damage, avoid biological toxicity, and high immune specificity Effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
example 1
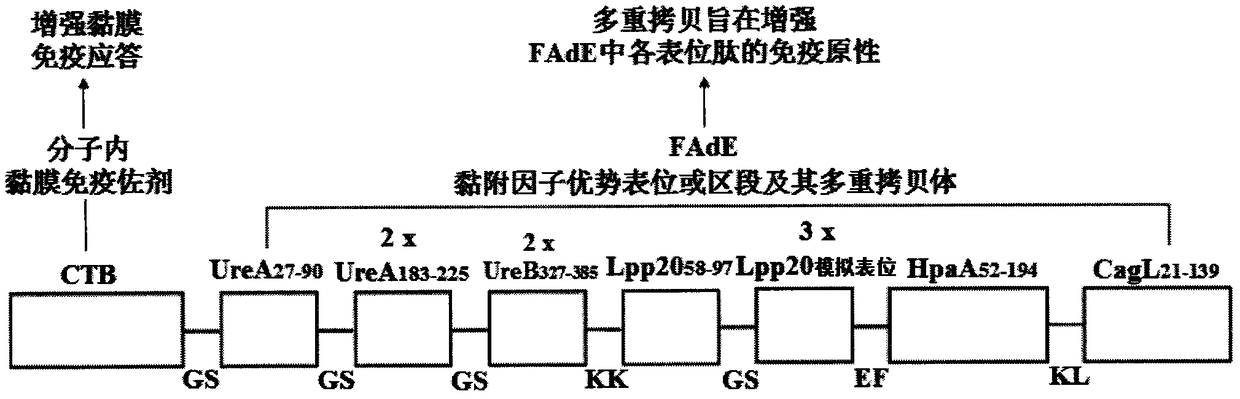
[0053] Example 1: Molecular structure design of Helicobacter pylori tetravalent adhesion factor multi-epitope vaccine CFAdE
[0054] According to "the body's immune protection mechanism against Hp" and "the immunological properties of key adhesion factor epitopes or segments such as Hp urease A and B double subunits, outer membrane protein Lpp20, HpaA and CagL", through biological information UreA 27-90 、UreA 183-225 , UreB 327-385 , Lpp20 58-97 , Lpp20 mimotope (SWPLYSDASGLG), HpaA 52-194 , CagL 21-139 The isoantigen epitopes or segments and the intramolecular mucosal immune adjuvant CTB were used in the construction of the Helicobacter pylori tetravalent adhesion factor multi-epitope vaccine CFAdE. Then, through the construction theory of the epitope vaccine and the analysis of bioinformatics, the connection sequence, spacer sequence and antigen epitope copy number of the selected antigenic epitope or segment are analyzed and determined, and finally a scientifically rea...
example 2
[0056] Example 2: Construction of recombinant expression vector pCzn1-CFAdE (containing fusion gene CFAdE)
[0057] (1) Gene synthesis of adhesion factor polyepitope peptide FAdE nucleotide sequence
[0058] The amino acid sequence of the previously designed adhesion factor multi-epitope peptide FAdE was transformed into the corresponding nucleotide sequence according to the codon preference principle of Escherichia coli, and Zhongding Biotechnology Company was entrusted to carry out gene synthesis.
[0059] (3) Connection of adhesion factor multi-epitope peptide FAdE gene and pCzn1-C expression vector
[0060] The synthesized adhesion factor multi-epitope peptide FAdE gene and pCzn1-C (containing CTB gene) were double digested with BamH I / Xbal respectively, and the reaction system was as follows:
[0061]
[0062] 37°C enzyme digestion reaction for 2h, then electrophoresis with 1% agarose gel, and observe the electrophoresis results. The adhesion factor multi-epitope pep...
example 3
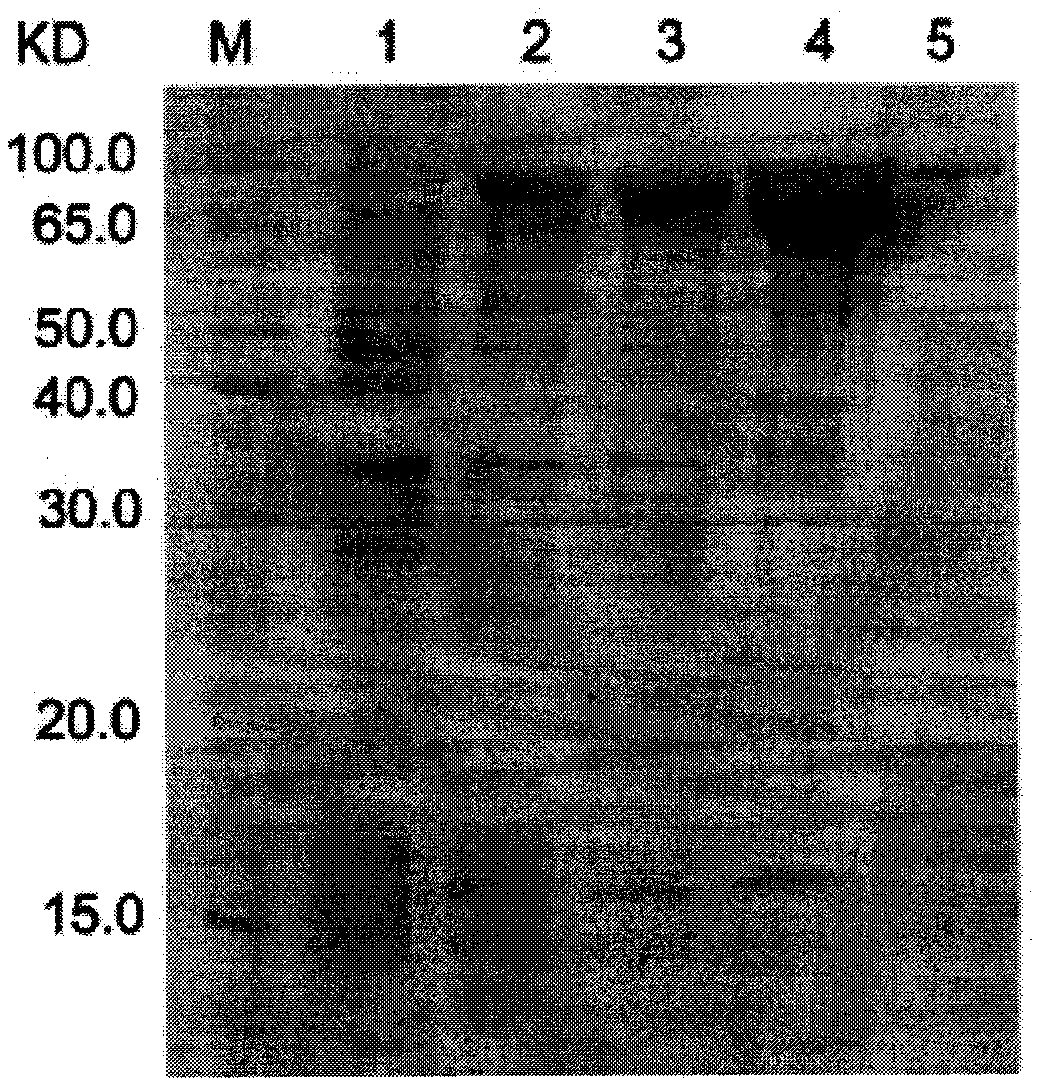
[0066] Example 3: Prokaryotic expression of recombinant protein CFAdE
[0067] The correct recombinant expression plasmid pCzn1-CFAdE was verified to be transformed into the Arctic Express strain. On the pre-prepared LB plate containing 50 μg / mL Amp, inoculate the loop-streaked genetically engineered strain Arctic Express / pCzn1-CFAdE, place it upside down in a 37°C incubator, and culture it overnight for 12-16 hours. Pick a single colony and inoculate it in In a test tube containing 50 μg / ml AMP in 3 ml of LB culture solution, shake overnight at 37°C at 220 rpm; the next day, inoculate at 1:100 in 30 ml of LB culture solution with 50 μg / ml AMP, shake at 37°C at 220 rpm until the cell OD600 is 0.6- 0.8 (about 2h). Take out 1ml of the culture, centrifuge at 10000g for 2min at room temperature, discard the supernatant, and resuspend the bacterial pellet with 100μl 1× loading buffer. Add IPTG to the remaining culture to a final concentration of 0.5 mM, shake at 220 rpm at 37°C f...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More