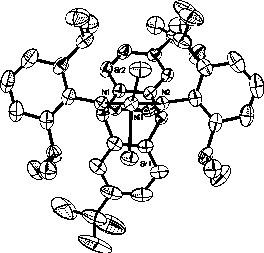
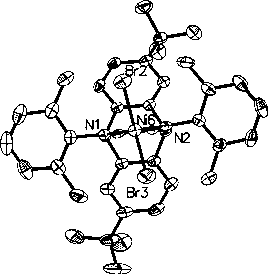
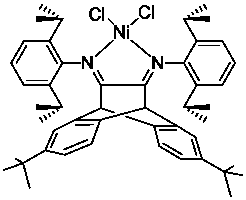
A kind of nickel base complex and its preparation method and application
A complex and nickel-based technology, which is applied in the direction of nickel organic compounds, can solve the problems of failing to meet the requirements of industrialization, poor thermal stability of catalysts, and rapid deactivation, etc., to achieve inhibition of aniline aromatic ring rotation, good temperature resistance, and increase in temperature. The effect of catalyst activity
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0058] Example 1 Synthesis of Ligand L1
[0059] Under nitrogen atmosphere and at room temperature, 100 mL of toluene, 2,6-di-tert-butyl-9,10-dihydro-9,10-ethylideneanthracene-11, 0.83 g (2.39 mmol) of 12-diketone, 0.975 g (0.96 mL, 5.5 mmol) of 2,6-diisopropylaniline, and a catalytic amount of p-toluenesulfonic acid were added, and heated at 110 °C to separate water and reflux for 24 hours . The reaction system gradually changed from light yellow to dark red, and released a lot of heat. The reacted mixture was rotary evaporated to remove the solvent, and the solid was recrystallized from ethanol to obtain a yellow solid powder with a yield of 75%. 1 H NMR (CDCl 3 , 300 MHz), δ (ppm): 1.03 (m, 12H), 1.5 (m, 12H), 1.23 (s, 18H), 2.53 (m, 4H), 4.91 (s, 2H), 7.07−7.25 (m , 12H). 13 C NMR (CDCl 3 , 400MHz), 22.30, 23.16, 28.28, 31.21, 34.79, 122.80, 123.91, 124.80, 135.31, 136.42, 138.46, 145.61, 150.29, 158.76.
Embodiment 2
[0060] Example 2 Synthesis of Ligand L2
[0061] According to the synthesis method of ligand L1 in Example 1, 2,6-dimethylaniline was used instead of 2,6-diisopropylaniline, and other operating conditions were the same. A tan solid powder was obtained, yield: 80%. 1 H NMR (CDCl 3 , 300 MHz), δ (ppm): 1.25(s, 18H), 1.82 (s, 6H), 1.91 (s, 6H), 3.49 (s, 2H), 6.99−7.23 (m, 12H).
Embodiment 3
[0062] Example 3 Synthesis of Ligand L3
[0063] According to the synthesis method of ligand L1 in Example 1, aniline was used instead of 2,6-diisopropylaniline, and other operating conditions were the same. A tan solid powder was obtained, yield: 85%. 1 H NMR (300MHz, CDC1 3 ), δ (ppm): 1.24 (s, 18H), 3.59 (s, 2H), 6.89−7.33 (m, 16H).
[0064] 2. Preparation of nickel-based complexes
PUM
| Property | Measurement | Unit |
|---|---|---|
| polydispersity index | aaaaa | aaaaa |
| polydispersity index | aaaaa | aaaaa |
| polydispersity index | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com