Crystal form G of ibrutinib and preparation method
A technology of ibrutinib and its crystal form, which is applied in the field of medicinal chemistry, can solve problems such as difficulty in implementing industrialization of stirring, low absorption bioavailability, and poor control of the production process, achieving good controllability and reproducibility, The effect of high bioavailability and cheap solvent
Inactive Publication Date: 2016-06-08
孙霖
View PDF7 Cites 24 Cited by
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
The operation of this method is cumbersome, there are repeated heating and cooling in the operation, and the production process is not easy to control
[0006] Suzhou Jingyun CN104327085A discloses another crystal form A (hereinafter referred to as crystal form A'), and its reported preparation method has three kinds: Method 1, dissolving the crude product of ibrutinib in a mixed solution of isopropanol and n-heptane In the method, crystal form A' was obtained by stirring at a speed of 750 revolutions per minute at room temperature. It is very difficult to industrialize such high-speed stirring; method 2, dissolving the crude ibrutinib in a mixed solvent of isopropanol and n-heptane , and at a cooling rate of 0.1 °C / min from 50 °C to 5 °C to obtain crystal form A, such a precise cooling rate is difficult to control; method 3, dissolve the crude ibrutinib in acetone, and then slowly add n-heptyl alkane, and stirred at a speed of 1000 rpm for 1 day to obtain Form A. This method requires a higher speed than Method 1, and it is more difficult to achieve industrialization.
And it has been reported that when these methods are repeated, the reproducibility is poor, and the crystal form A disclosed in WO2013 / 184572 will be obtained if the control is not good.
In addition, the existing dosage forms have low water solubility and low oral absorption bioavailability
Method used
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
View moreImage
Smart Image Click on the blue labels to locate them in the text.
Smart ImageViewing Examples
Examples
Experimental program
Comparison scheme
Effect test
Embodiment 1
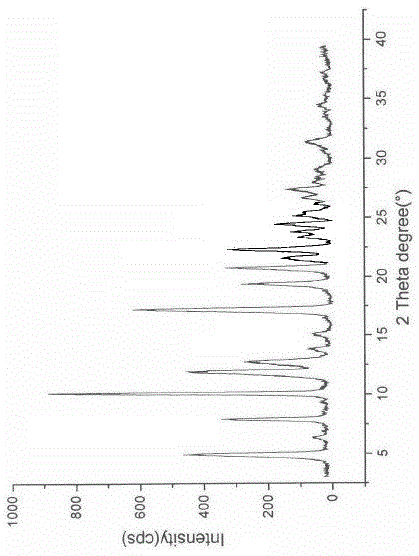
[0019] Add 10 g of ibrutinib amorphous substance into a mixed solution of 100 ml of ethyl acetate and methanol (the volume ratio of isopropanol and methanol is 2:1), heat to reflux to dissolve, stir for 10 hours after dissolving, add 100 ml of water, and stir for 2 Hours, filtered, heated and dried while grinding to obtain crystal G, using Cu-Kα radiation, the X-ray powder diffraction represented by 2θ angle is as follows figure 1 As shown, the HPLC purity was 99.92%.
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More PUM
 Login to View More
Login to View More Abstract
The invention discloses a crystal form G of ibrutinib. The crystal form G is characterized in that X-ray powder diffraction (X-RPD) which adopts Cu-Kalpha radiation and is represented with a 2theta angle has diffraction peaks in positions at angles of 5.0 degrees plus or minus 0.2 degrees, 7.3 degrees plus or minus 0.2 degrees, 10.1 degrees plus or minus 0.2 degrees, 12.0 degrees plus or minus 0.2 degrees, 13.2 degrees plus or minus 0.2 degrees, 17.1 degrees plus or minus 0.2 degrees, 19.5 degrees plus or minus 0.2 degrees, 20.8 degrees plus or minus 0.2 degrees, 22.3 degrees plus or minus 0.2 degrees, 24.3 degrees plus or minus 0.2 degrees, 27.4 degrees plus or minus 0.2 degrees and 31.2 degrees plus or minus 0.2 degrees. Related solvents in a preparation process of the crystal form G are cheap, the conditions are mild, the operation is simple, good controllability and reproducibility are realized, further, the prepared crystal form has great stability, the HPLC (high performance liquid chromatography) purity is higher than 99%, and the phenomenon of crystal transformation can be avoided; besides, the solubility is high, the dissolubility is good, and the bioavailability is high.
Description
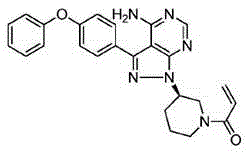
technical field [0001] The invention relates to the field of medicinal chemistry, in particular, the invention relates to a crystal form of ibrutinib, and a preparation method and use of the crystal form. Background technique [0002] Ibrutinib (Ibrutinib) is a first-in-class oral Bruton's tyrosine kinase (BTK) inhibitor, jointly developed by American Pharmacyclics and Johnson & Johnson, the trade name is Imbruvica, and its chemical name is: 1- [(3R)-3-[4-Amino-3-(4-phenoxyphenyl)-1H-pyrazolo[3,4-d]pyrimidin-1-yl]-1-piperidinyl]- 2-propen-1-one, the structural formula is as follows: [0003] [0004] Ibrutinib irreversibly inhibits BTK by selectively covalently binding to the cysteine residue (Cys-481) in the active site of the target protein Btk, thereby effectively preventing tumors from migrating from B cells to adapting to tumor growth. Environmental lymphoid tissue. In November 2013, the US Food and Drug Administration (FDA) approved its listing for the treatmen...
Claims
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More Application Information
Patent Timeline
 Login to View More
Login to View More IPC IPC(8): C07D487/04A61K31/519A61P35/02
CPCC07D487/04C07B2200/13
Inventor 孙霖
Owner 孙霖