PA (pseudomonas aeruginosa) recombinant protein Vac33, as well as preparation method and application thereof
A Pseudomonas aeruginosa, recombinant protein technology, applied in biochemical equipment and methods, chemical instruments and methods, recombinant DNA technology, etc., to achieve mild purification conditions, good immune protection effect, maintain spatial conformation and immunogenicity. Effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
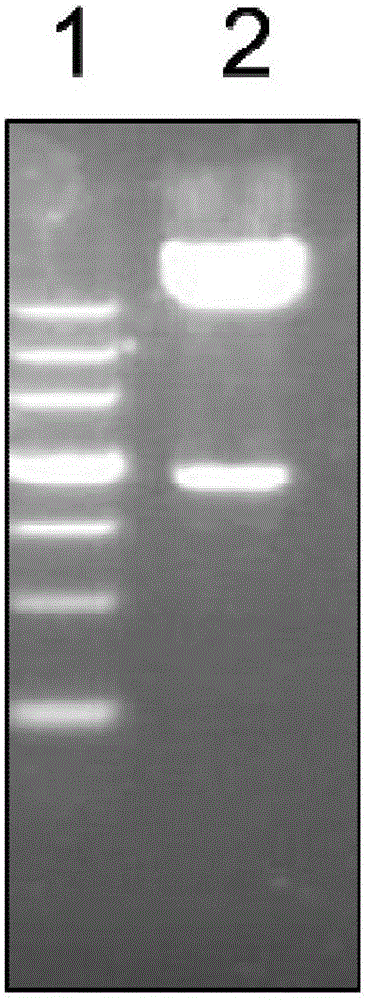
[0033] Example 1 Construction and Identification of Recombinant Plasmids
[0034] 1. The synthesis of the DNA sequence of Vac33 and the connection of the sequence and pGEX-6p-2 were synthesized by Shanghai Sangon Bioengineering Co., Ltd.
[0035] 2. Transformation of recombinant plasmids
[0036] Take 3 tubes of Escherichia coli XL1blue competent cells (Shanghai Chaoyan Biotechnology Co., Ltd.) from the -80°C refrigerator, add pGEX-6P-2 plasmid (GE Healthcare Life Sciences) to the first tube as a positive control; add 1ulVac33 synthetic plasmid to the second tube; No exogenous DNA was added to the third tube as a negative control. Ice bath for 50min, heat shock in 42℃ metal bath for 90s, rapid ice bath for 2min. Add 600 μl LB blank medium, mix well, and place in a shaker at 37°C at 220rp for 1h.
[0037] Each tube was centrifuged at 5000 rpm for 3 min at room temperature, discarded 300 μl of supernatant, and then resuspended the bacteria, took 200 μl and spread it on an A...
Embodiment 2
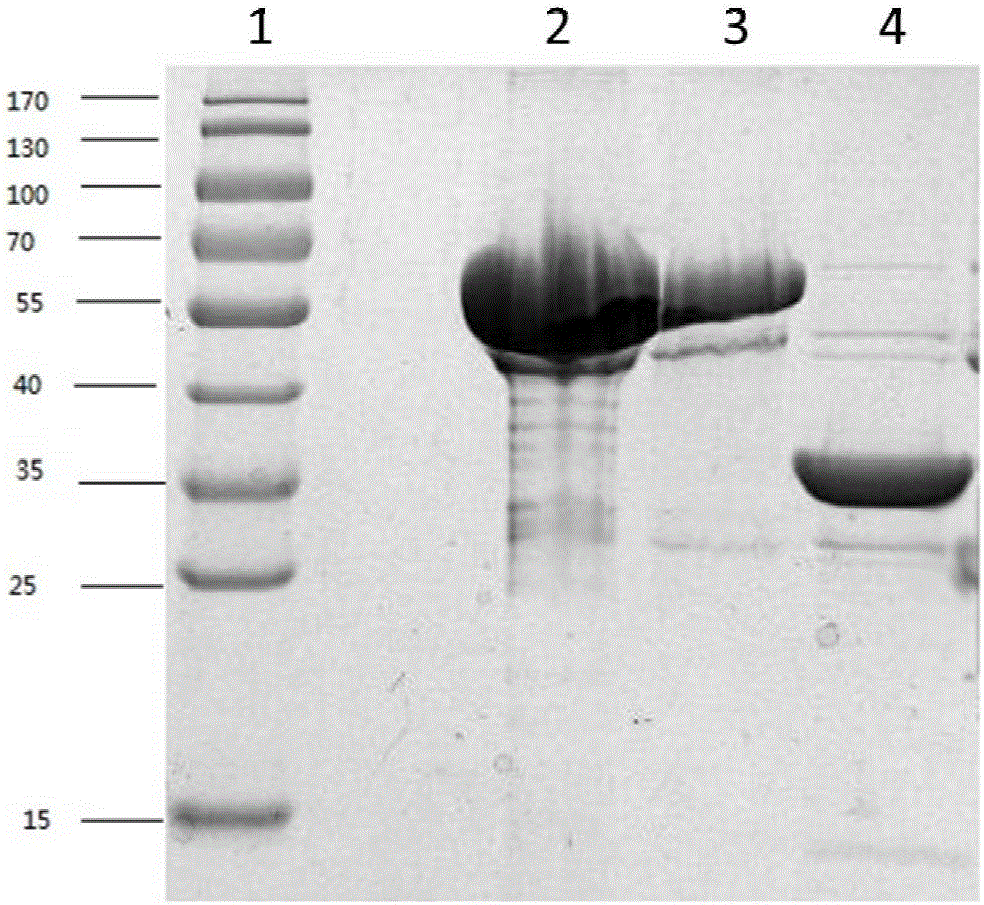
[0043] Example 2 Induced expression, purification and identification of expression form of recombinant fusion protein PcrV-OprI in prokaryotic expression system-Escherichia coli
[0044] 1. Vac33 induced expression
[0045] Take 100 μL of the overnight cultured pGEX-6P-2-Vac33 / XL-1blue bacterial solution and add it to 10 mL of Amp+ resistant LB medium, culture overnight at 180 rpm at 37°C, respectively take 400 μL of the overnight cultured bacterial solution and add it to 20 mL of Amp+ resistant LB medium (The rest of the bacterial solution is stored in a refrigerator at 4°C for later use), culture at 37°C for 2-3 hours, rotate at 200 rpm, and reactivate until the OD600 is 0.8-1.0, add 4 μL of IPTG to make the final concentration 200 μM, and place on a shaker Induced expression Induced expression at 30°C for 3h.
[0046] 2) Take out the bacterial solution after induced expression, centrifuge at 12000rpm for 5min, discard the supernatant, add 1mL of lysisbuffer (20mMPB, pH7....
Embodiment 3Va
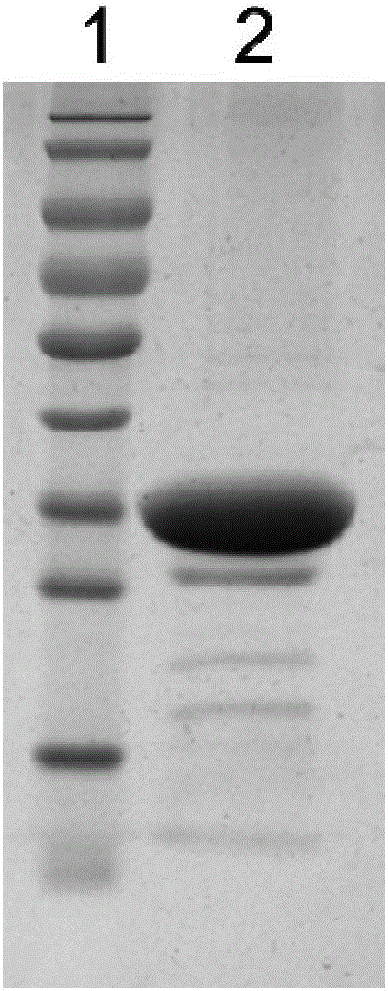
[0053] The preparation of embodiment 3Vac33 antigen
[0054] 1. Amplify culture to obtain protein
[0055] Take 400 μL of the spare pGEX-6P-2-Vac33 / XL-1blue bacterial solution stored in the refrigerator at 4°C and add it to 20mL LB medium containing Amp resistance for one activation. After culturing at 200rpm for 5-6 hours at 37°C, take 8mL once The activated bacterial solution was added to 400mL LB medium containing Amp resistance for secondary activation, cultured at 37°C for 3-4h until the OD600 was 1.0, then added 80μLPTG (final concentration 200μM) and placed in a shaker at 16°C overnight to induce Afterwards, 12000rpm centrifugal 15min collects thalline, adds 20mLlysisbuffer (same as embodiment 2) after resuspending thalline, bacterium liquid is carried out ultrasonic cracking 3min (200V), collect supernatant and GlutathioneSepharose4B (GE Company) gel beads (beads) binding treatment; then SDS-PAGE gel electrophoresis.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com