A kind of stable fasudil hydrochloride injection and preparation method thereof
A technology of fasudil hydrochloride and injection, which is applied in the direction of medical preparations with non-active ingredients, medical preparations containing active ingredients, and pharmaceutical formulas, which can solve the problems of increasing the content of insoluble particles, reduce the dosage, reduce the Potential safety hazards and the effect of improving drug safety
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
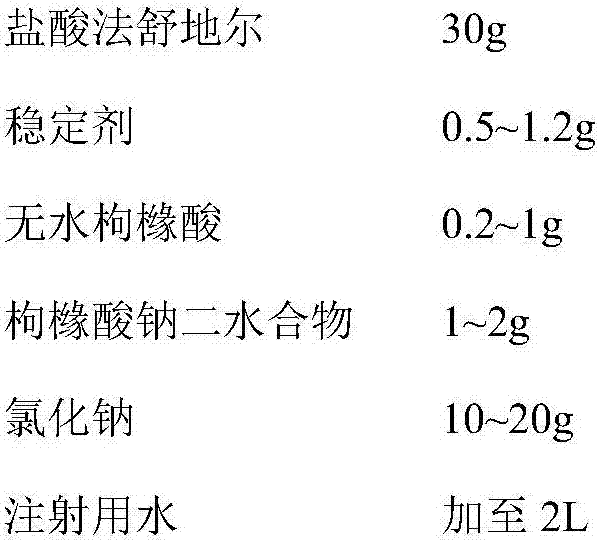
[0041] Every 1000 pieces of described fasudil hydrochloride injection, its formula consists of:
[0042]
[0043] The stabilizer is composed of N-carboxymethyl chitosan and reduced glutathione in a mass ratio of 1:0.25.
[0044] Preparation:
[0045](1) get the water for injection of recipe quantity and use the polyvinyl chloride exchange column that contains diethylaminoethyl dextran gel to carry out pyrogen removal treatment, then feed carbon dioxide to saturation, for subsequent use;
[0046] (2) get the recipe quantity of fasudil hydrochloride, N-carboxymethyl chitosan, reduced glutathione, anhydrous citric acid, sodium citrate dihydrate and sodium chloride and mix, add The water for injection of 90% of the recipe quantity treated in step (1) was stirred until dissolved, and the pH was adjusted to 5.0 with hydrochloric acid or sodium hydroxide, then 3 g of medicinal charcoal for freezing treatment in a -10°C refrigerator was added, and the stirring was carried out for ...
Embodiment 2
[0049] Every 1000 pieces of described fasudil hydrochloride injection, its formula consists of:
[0050]
[0051] The stabilizer is composed of N-carboxymethyl chitosan and reduced glutathione in a mass ratio of 1:0.25.
[0052] The preparation method is the same as in Example 1.
Embodiment 3
[0054] Every 1000 pieces of described fasudil hydrochloride injection, its formula consists of:
[0055]
[0056] The stabilizer is composed of N-carboxymethyl chitosan and reduced glutathione in a mass ratio of 1:0.25.
[0057] The preparation method is the same as in Example 1.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More