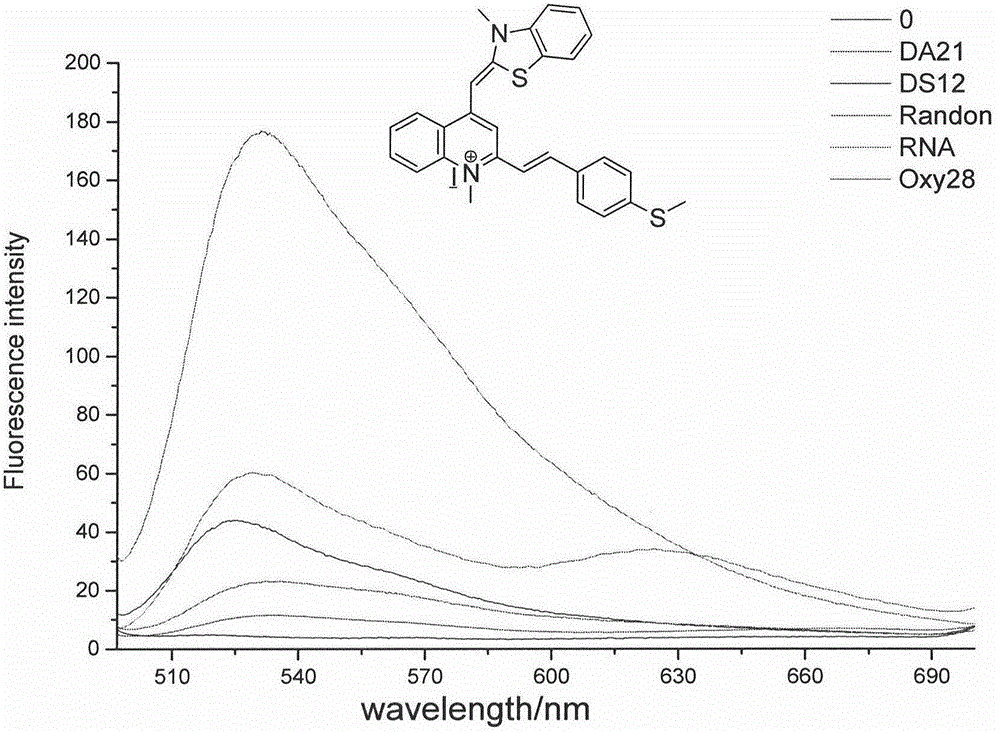
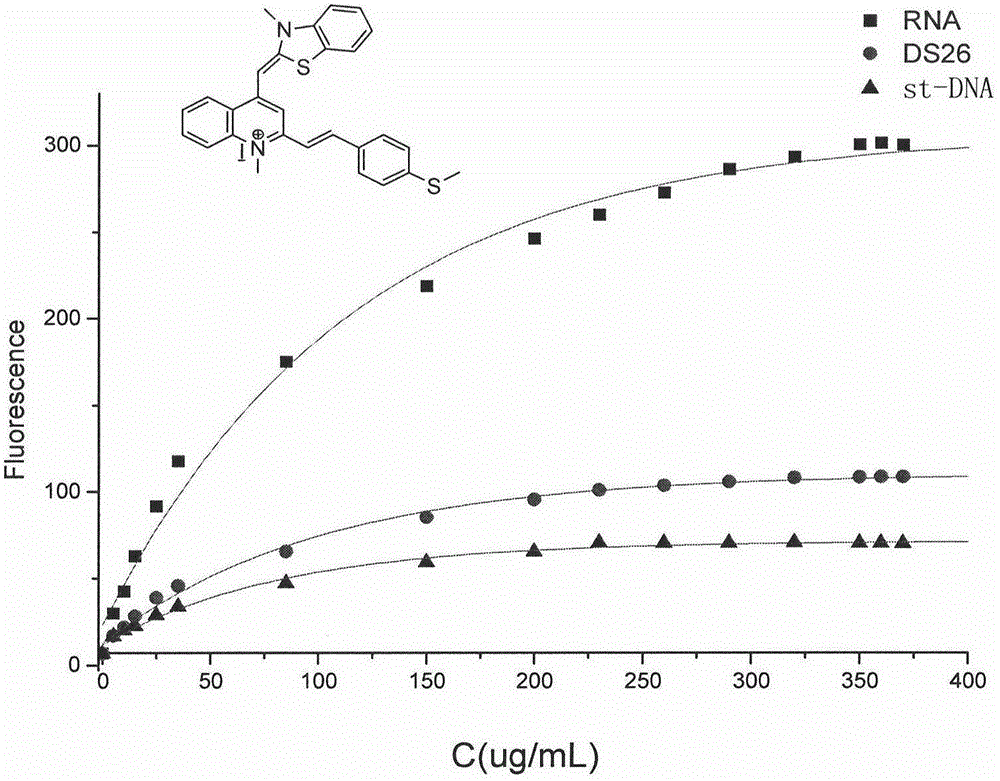
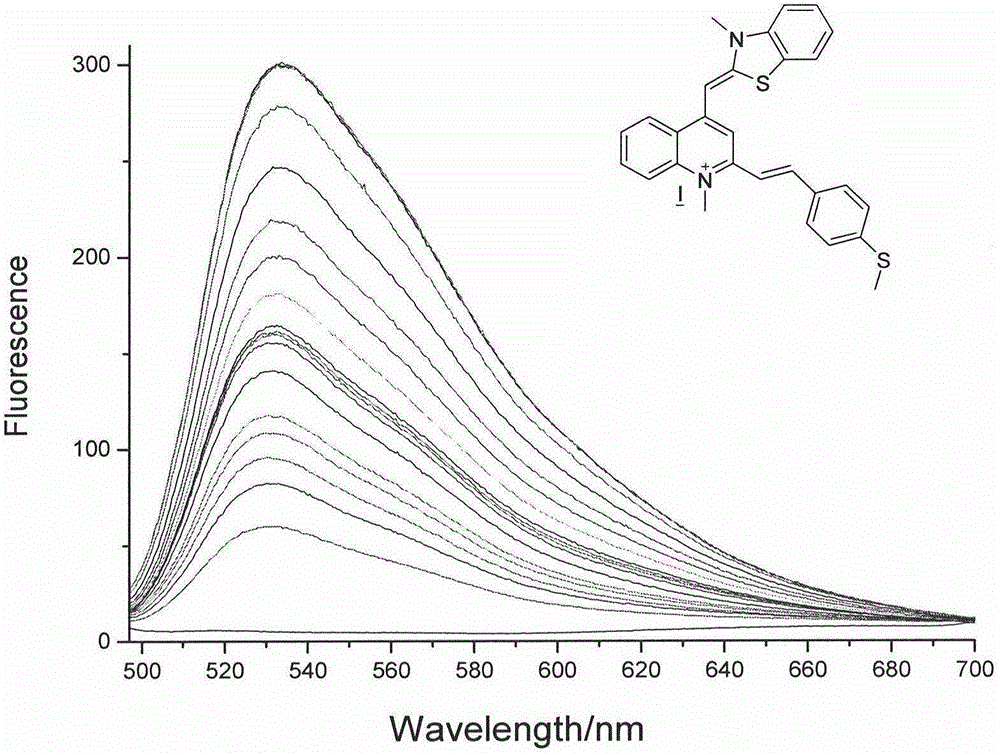
RNA fluorescent probe and manufacturing method and application thereof
A fluorescent probe and reaction technology, which is applied in the detection of RNA and nucleolus imaging in cells, can solve the problems affecting the application value, slow response speed, and high phototoxicity, and achieve low biological toxicity, good membrane permeability, and light The effect of strong stability
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0027] Embodiment one: the synthesis of compound 2
[0028] Weigh 0.2g (1.1236mmol) of 4-chloro-quinaldine into a 25ml round-bottomed flask, add about 0.96g of iodomethane and 1.5ml of sulfolane in 6 times the molar amount, heat the mixture to 50°C, and react for 18 hours , cooled, shaken after adding anhydrous diethyl ether, suction filtered, the solid was washed with anhydrous diethyl ether, weighed after vacuum drying to obtain 0.345g of compound 2 with a yield of 95.8%: 1 H NMR (400MHz, DMSO) δ8.56(d, J=8.4Hz, 1H), 8.46(d, J=8.3Hz, 1H), 8.22(t, J=8.1Hz, 1H), 8.01(t, J =7.9Hz, 1H), 7.55(s, J=7.4Hz, 1H), 4.20(s, 3H), 3.74(s, 1H), 2.68(s, 3H).
Embodiment 2
[0029] Embodiment two: the synthesis of compound 4
[0030] Weigh 0.25g (1.68mmol) of 2-methyl-benzothiazole into a 25ml round bottom flask, add about 1g of methyl iodide and 5ml of absolute ethanol in 6 times the molar amount, and react at 80°C for 15 hours. The reacted solution was cooled to room temperature, then 5ml of absolute ethanol and chloroform were added, suction filtered after shaking, and the precipitate was washed with a small amount of ethanol and chloroform, and after vacuum drying, Compound 4 was obtained as a white powdery solid 0.448g. The rate is 91.7%: 1 H NMR (400MHz, DMSO) δ8.44(d, J=8.1Hz, 1H), 8.30(d, J=8.4Hz, 1H), 7.90(t, J=7.8Hz, 1H), 7.81(t, J =7.7Hz, 1H), 4.20(s, 3H), 3.54(s, 1H), 3.17(s, 3H).
Embodiment 3
[0031] Embodiment three: the synthesis of compound 5
[0032] Weigh 0.50 g each of compounds 2 and 4, add them into a round bottom flask containing 10 ml of methanol, stir at room temperature for 6 minutes, then add 2 ml of 0.5 mol / L sodium bicarbonate aqueous solution, and stir at room temperature for about 1 hour. Add 4ml of saturated KI solution to the reacted solution, stir for about 15 minutes, then filter with suction, then wash with 10ml of water and 4ml of acetone to finally obtain a brick red solid, and obtain 0.98g of compound 5 after drying, with a yield of 81.7%: 1 H NMR (400MHz, DMSO) δ8.77(d, J=8.3Hz, 1H), 8.18(d, J=8.7Hz, 1H), 8.02-7.96(m, 2H), 7.74(d, J=8.2Hz , 2H), 7.59(t, J=7.7Hz, 1H), 7.39(t, J=7.5Hz, 1H), 7.34(s, 1H), 6.85(s, 1H), 4.07(s, 3H), 3.98 (s,3H), 2.87(s,3H).
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More