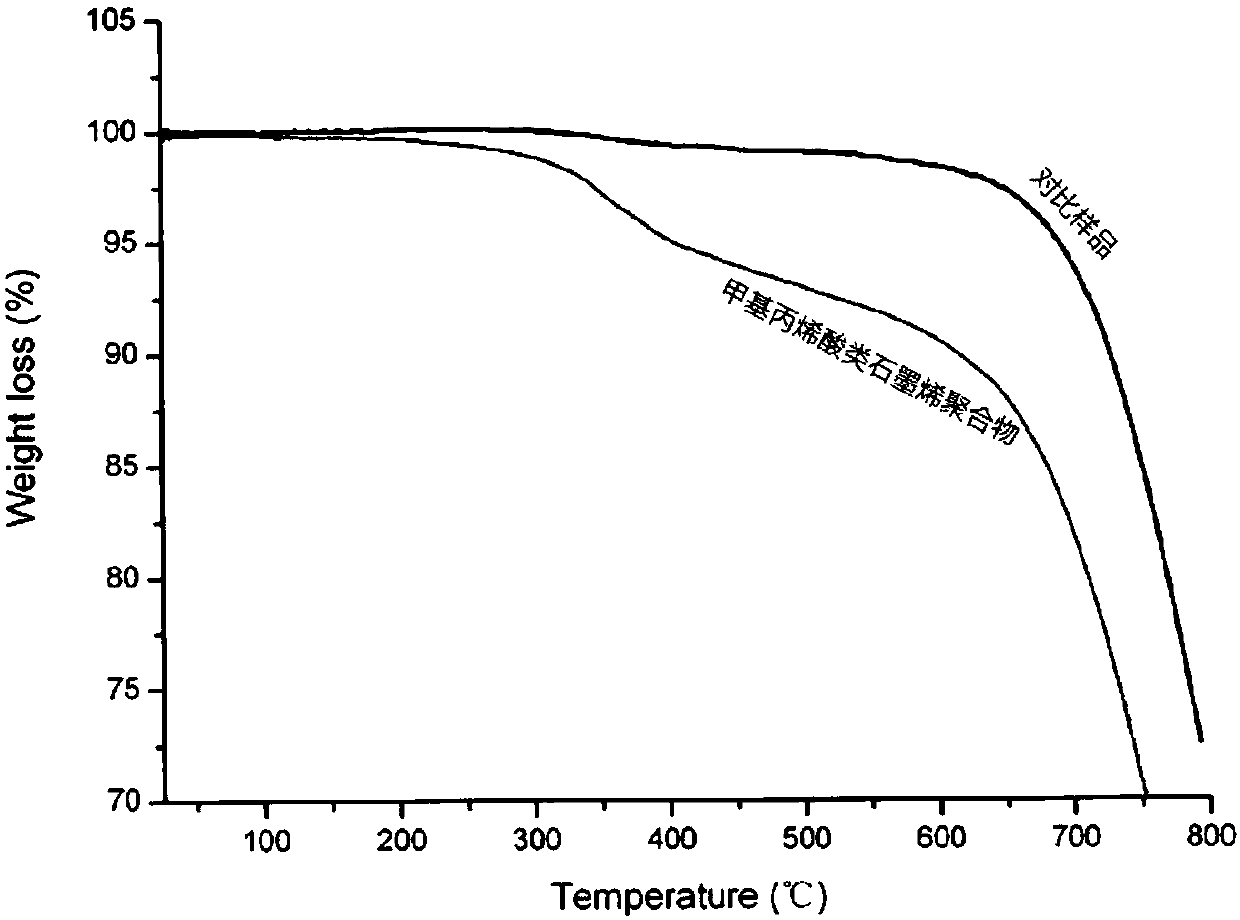
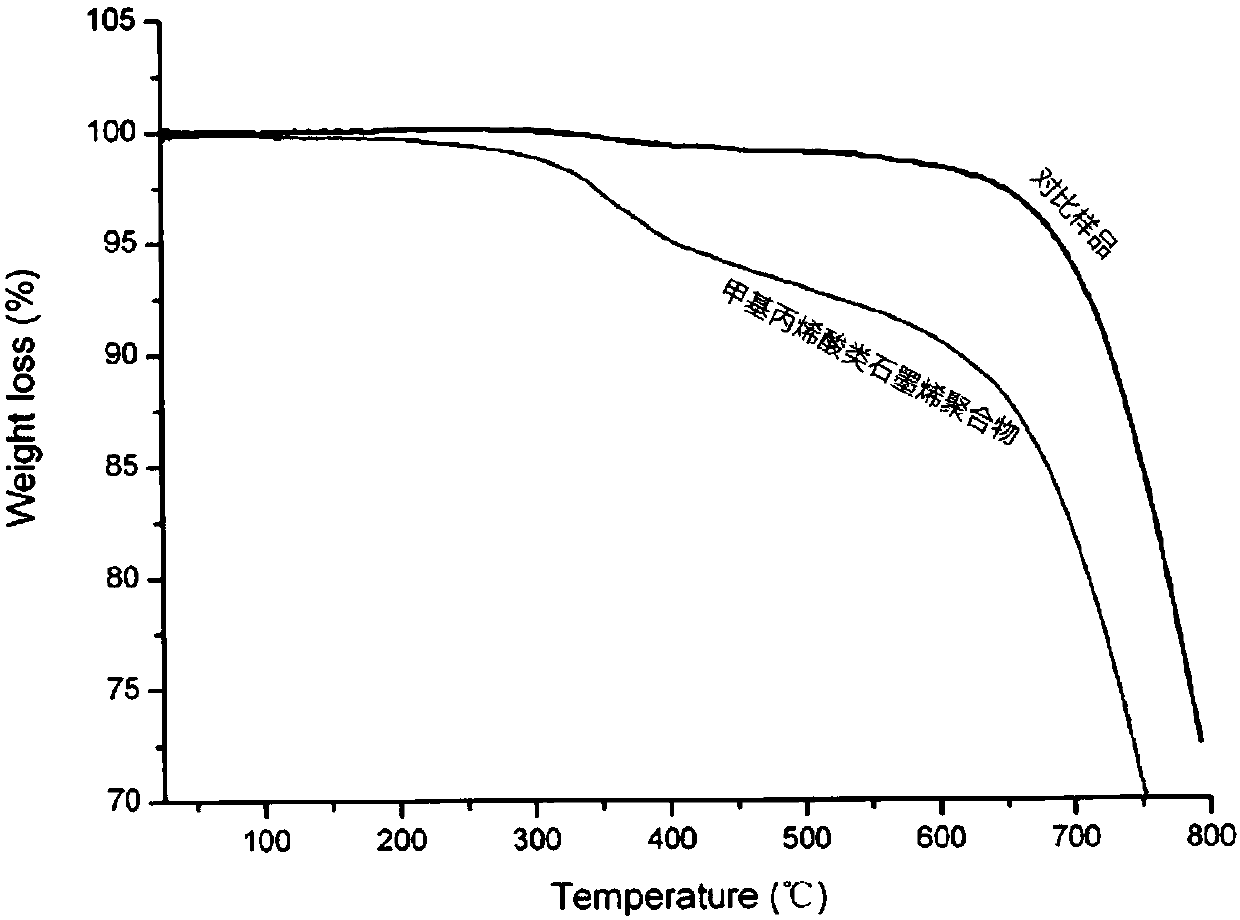
Methacrylic acid graphene polymers and preparation method thereof
A technology of methacrylic acid and graphene, applied in the field of graphene, can solve problems such as poor performance, and achieve the effect of improving overall performance, less defects, and more modification space
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0019] The preparation of embodiment 1 original graphene
[0020] Add 100g of graphite powder into 100mL of NMP solvent, ultrasonicate for a period of time, centrifuge, and keep the supernatant to obtain the graphene dispersion.
Embodiment 2
[0021] The preparation of embodiment 2 enediynes
[0022] A: When the R group is a benzene ring:
[0023] (1) Put o-bromoiodobenzene (7.06mmol, 2g), cuprous iodide (0.228mmol, 0.044g) and bis(triphenylphosphine)palladium dichloride (0.238mmol, 0.166g) into a 100ml In the Schlenk reaction flask, redestilled triethylamine (20ml) was added after rapid vacuum exchange for nitrogen, and phenylacetylene (18.2mmol, 2ml) was added under nitrogen protection after vacuum exchange for nitrogen. Under the protection of nitrogen, the mixture was reacted in an oil bath at 80°C for 48 hours, and the mixed product was separated and purified by column chromatography to obtain product 1.
[0024] (2) As above, add product 1 (3.53mmol, 0.9g), bis(triphenylphosphine) palladium dichloride (0.119mmol, 0.083g), cuprous iodide (0.114mmol, 0.022g) into the reaction flask , triethylamine (10ml), after vacuuming and changing nitrogen, add propynyl alcohol (9.10mmol, 0.6ml), react in an oil bath at 80°...
Embodiment 3
[0034] The preparation of embodiment 3 graphene polymer
[0035] The NMP dispersion (1mg / mL) of prepared 100mL graphene is heated to reflux, slowly adds the NMP solution (100mL) of enediyne (100mg) with the speed of 1r / min with peristaltic pump, cool down after 12 hours of reaction , passing the reaction solution through a microporous membrane, filtering the obtained solid and washing it with THF, and finally dispersing the polymer into the THF solution to obtain a methacrylic graphene polymer solution.
[0036]
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More