Preparation method of quaternary ammonium salt type trisiloxane gemini surfactant
A technology of trisiloxane and gemini surface, which is applied in the preparation of quaternary ammonium salt type trisiloxane gemini surfactant and the field of quaternary ammonium salt type trisiloxane gemini surfactant, which can solve the limitation of research and industrial production , harsh experimental conditions, long reaction time and other problems, to achieve the effect of easy control, simple steps and simple reaction conditions
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
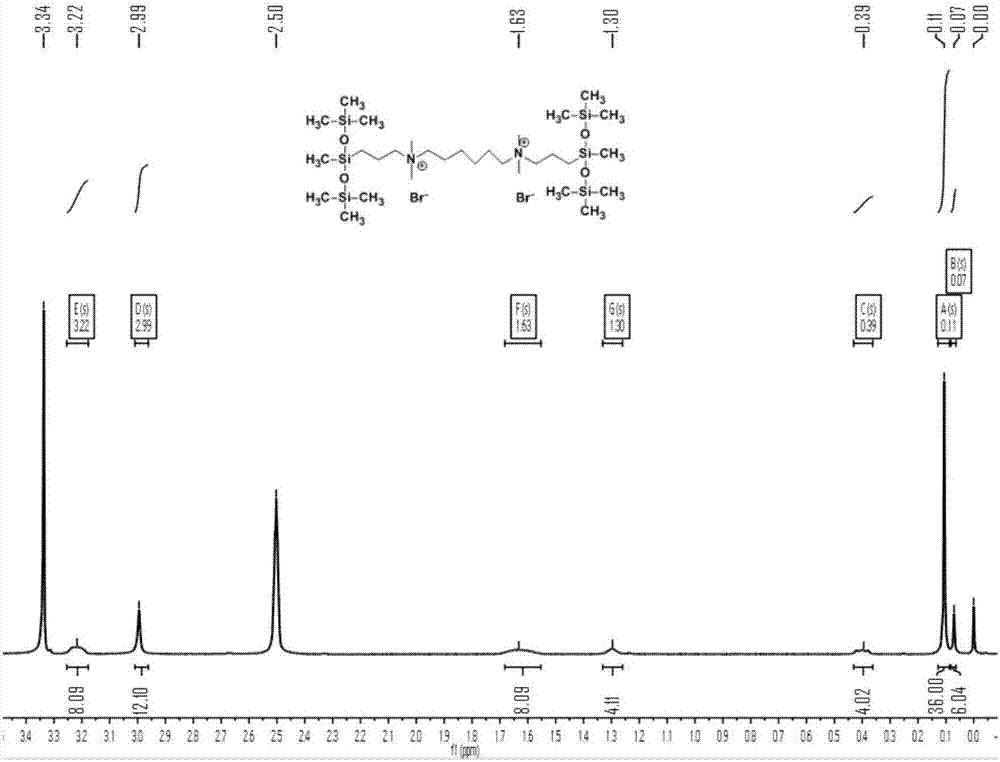
[0042] Example 1: Quaternary ammonium trisiloxane gemini surfactant MR 6 Preparation of M
[0043] (1) Synthesis of tertiary amines containing silyl groups (hydrosilylation reaction)
[0044] Add N,N-dimethylallylamine (20.6mmol) and heptamethyltrisiloxane (20.6mmol) into a 100mL three-necked flask with a magnetic stirrer, stir and heat up to 75°C under nitrogen protection, Stir for 30 minutes, add chloroplatinic acid in isopropanol solution (75ppm of heptamethyltrisiloxane) as a catalyst, raise the temperature to 100°C, and reflux for 3 hours. The obtained product was rotary evaporated at 40°C under reduced pressure to remove isopropanol to obtain a colorless transparent liquid.
[0045] (2)MR 6 Synthesis of M (quaternization reaction)
[0046] Take 20mL of ethanol and place it in a 100mL three-necked flask, add the silyl-containing tertiary amine (12mmol) and 1,6-dibromohexane (5mmol) obtained in the above step 1, raise the temperature to 80°C, and reflux for 48h. After...
Embodiment 2
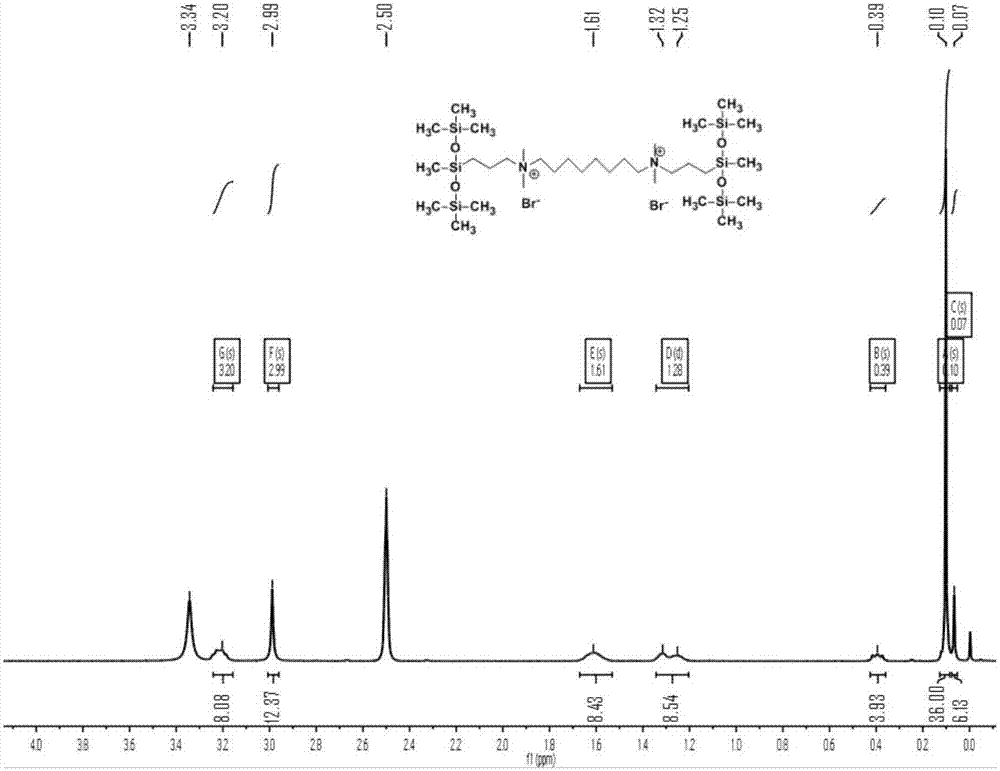
[0049] Example 2: Quaternary ammonium trisiloxane gemini surfactant MR 8 Preparation of M
[0050] (1) Synthesis of tertiary amines containing silyl groups (hydrosilylation reaction)
[0051] Add N,N-dimethylallylamine (20.8mmol) and heptamethyltrisiloxane (20mmol) into a 100mL three-neck flask with a magnetic stirrer, stir under nitrogen protection and raise the temperature to 60°C, and stir for 30min , add chloroplatinic acid in isopropanol solution (the amount used is 30ppm of heptamethyltrisiloxane) as a catalyst, raise the temperature to 120°C, and reflux for 3h. The obtained product was rotary evaporated at 60°C under reduced pressure to remove isopropanol to obtain a colorless transparent liquid.
[0052] (2)MR 8 Synthesis of M (quaternization reaction)
[0053] Take 20mL of n-propanol and put it in a 100mL three-necked flask, add the silane-containing tertiary amine (10.5mmol) and 1,8-dibromooctane (5mmol) obtained in the above step 1, raise the temperature to 80°C...
Embodiment 3
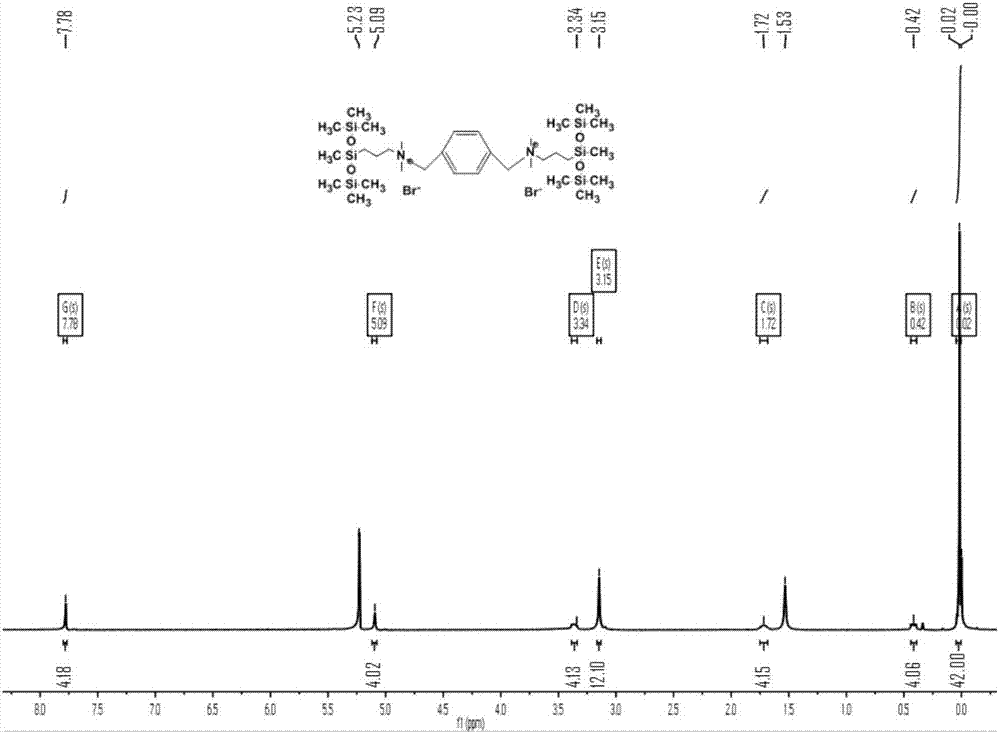
[0056] Embodiment 3: Preparation of quaternary ammonium salt type trisiloxane gemini surfactant MDM
[0057] (1) Synthesis of tertiary amines containing silyl groups (hydrosilylation reaction)
[0058] Add N,N-dimethylallylamine (26mmol) and heptamethyltrisiloxane (20mmol) into a 100mL three-necked flask with a magnetic stirrer, stir and raise the temperature to 80°C under nitrogen protection, and stir for 30min. Add a solution of chloroplatinic acid in isopropanol (50 ppm of heptamethyltrisiloxane) as a catalyst, raise the temperature to 90° C., and reflux for 5 hours. The obtained product was rotary evaporated at 70°C under reduced pressure to remove isopropanol to obtain a colorless transparent liquid.
[0059] (2) Synthesis of MDM (quaternization reaction)
[0060] Take 20mL of ethyl acetate and place it in a 100mL three-necked flask, add the above intermediate (14mmol) and 1,4-bis(bromomethyl)benzene (5mmol), heat up to 100°C, and reflux for 96h. After the reaction was...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - Generate Ideas
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com