Preparation method of poly-substituted oxazole derivative
A derivative and multi-substitution technology, which is applied in the field of preparation of multi-substituted oxazole derivatives, can solve the problems of serious pollution, use of heavy metals, poor atom economy, etc., and achieve simple synthesis method, easy purification, and scientific synthesis method reasonable effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
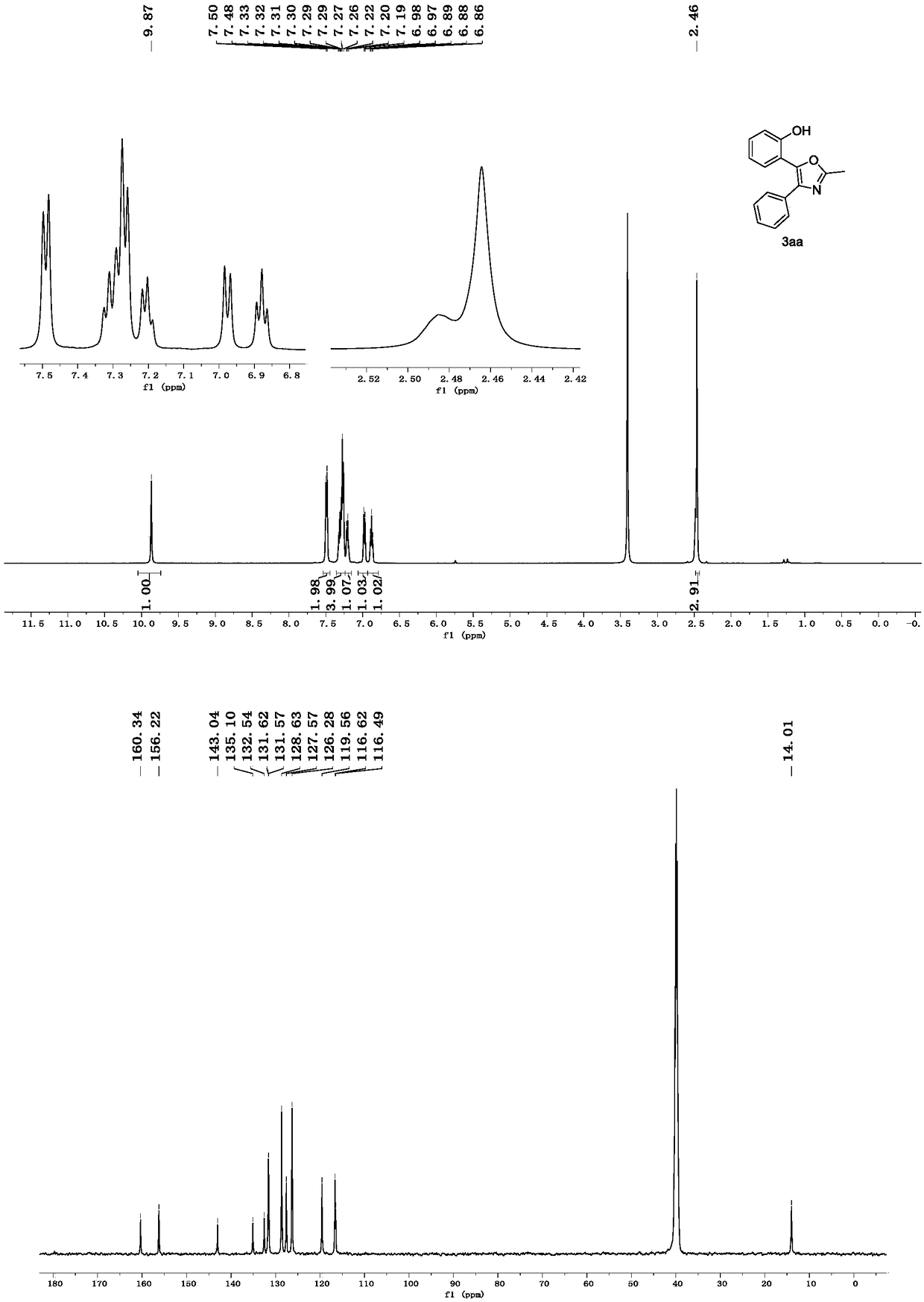
[0023] 1) Preparation of oxazole derivative 3aa
[0024]
[0025] To a 10 ml round bottom flask was added N-phenoxyacetamide 1a (0.36 mmol, 54.4 mg), 2a (0.3 mmol, 104.4 mg) and K 2 CO 3 (0.6mmol, 82.9mg). Add 1,2-dichloroethane (2 mL), stir in a water bath at 20° C., and react for 4 hours. After the reaction is complete, use a rotary evaporator to remove the solvent to obtain a crude product, which is separated by column chromatography (200-300 mesh silica gel) (petroleum ether / ethyl acetate=4 / 1), and uses a rotary evaporator to remove the solvent to obtain the target The yield of the multi-substituted oxazole derivative 3aa was 90%.
[0026] Spectrum analysis data 3aa:
[0027] 1 H NMR (500MHz, DMSO-d 6 )δ9.87(s,1H),7.49(d,J=7.5Hz,2H),7.35–7.24(m,4H),7.20(t,J=7.3Hz,1H),6.98(d,J=8.2 Hz,1H),6.88(t,J=7.5Hz,1H),2.46(s,3H); 13 C NMR(125MHz,DMSO)δ160.34,156.22,143.04,135.10,132.54,131.62,131.57,128.63,127.57,126.28,119.56,116.62,116.49,40.36,40.20,40.04,39.87,39.71,39.5...
Embodiment 2
[0029] Replace 1a in Example 1 with 1b, and other conditions are the same as Example 1. The experimental results are shown in Table 1.
[0030]
[0031] Spectrum analysis data 3ba:
[0032] 1 H NMR (500MHz, DMSO-d 6 )δ9.89(s, 1H), 7.47(d, J=7.5Hz, 2H), 7.30(t, J=7.7Hz, 2H), 7.23(t, J=7.4Hz, 1H), 7.15(ddt, J=12.5,9.0,4.3Hz,2H),6.95(dd,J=9.0,4.7Hz,1H),2.47(s,3H); 13 C NMR (125MHz, DMSO-d 6 )δ160.66,156.28,154.41,152.57,141.64,135.68,132.29,128.68,127.80,126.41,118.16,117.98,117.69,117.40,117.21,13.99; HRMS(ESI)m / z C calcd 16 h 13 NO 2 F+[M+H]+270.0930, found 270.0932.
Embodiment 3
[0034] Replace 1a in Example 1 with 1c, and other conditions are the same as Example 1. The experimental results are shown in Table 1.
[0035]
[0036] Spectrum analysis data 3ca:
[0037] 1 H NMR (500MHz, DMSO-d 6 )δ10.17(s,1H),7.45(d,J=7.4Hz,2H),7.38–7.26(m,4H),7.22(t,J=7.3Hz,1H),6.96(d,J=8.9 Hz,1H),2.46(s,3H); 13 C NMR (125MHz, DMSO-d 6 )δ160.74,155.12,141.37,135.82,132.29,131.15,130.64,128.69,127.83,126.42,122.83,118.32,118.19,40.41,40.24,40.08,39.90,39.74,39.57,39.41,13.97;HRMS(ESI)m / z calcd for C 16 h 13 NO 2 Cl + [M+H] + 286.0635, found 286.0638.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com