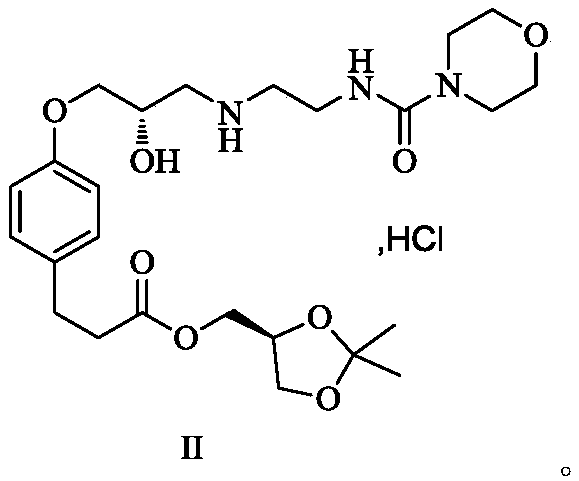
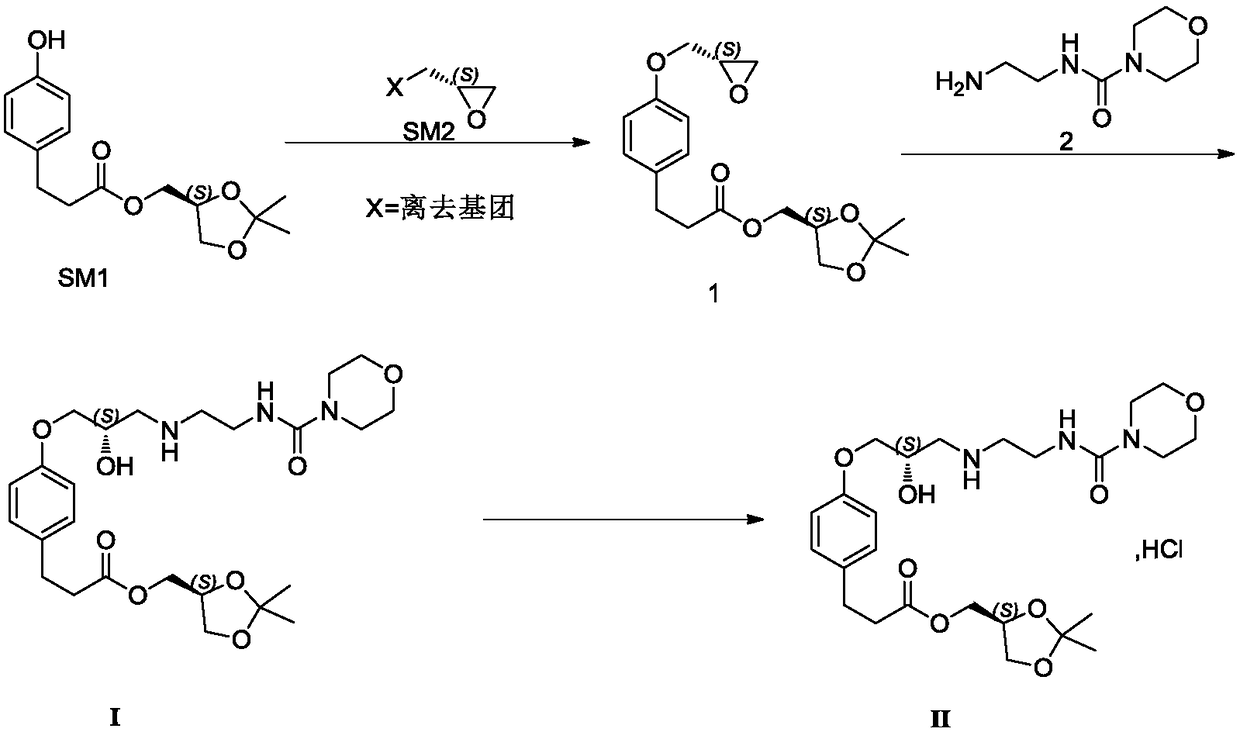
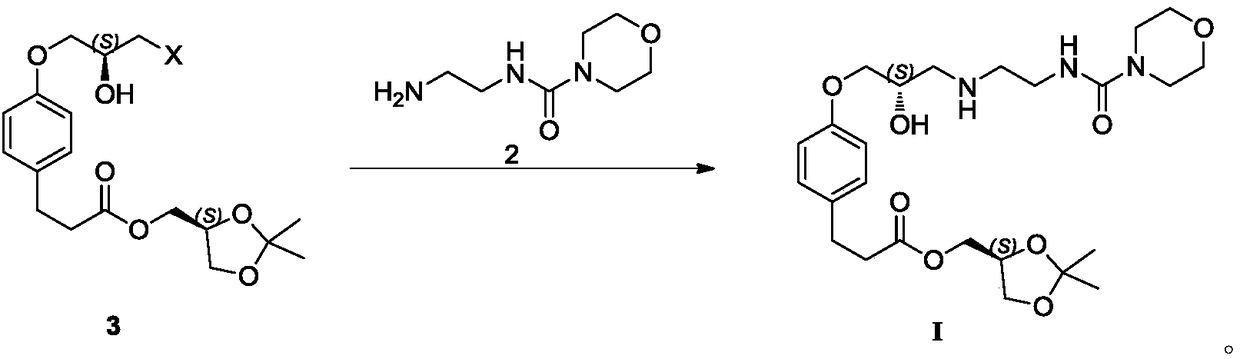
Preparation method of landiolol hydrochloride
A technology for landiolol hydrochloride and salt formation, applied in the field of preparation of landiolol hydrochloride, can solve problems such as affecting product quality and yield, malodorous thioether, insufficient alkali stability and the like
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
specific Embodiment approach
[0049] The following examples are used to further describe the present invention, but these examples do not limit the scope of the present invention.
[0050] Test equipment used in the experiment:
[0051] Chromatographic column: Octadecylsilane bonded silica gel as filler
[0052] Detection wavelength: 223nm
[0053] Flow rate: 0.8ml / min
[0054] Mobile phase A: Phosphate solution (take 0.5g sodium heptanesulfonate and 3.9g sodium dihydrogen phosphate, add water 1000mL to dissolve)-methanol (60:40)
[0055] Mobile Phase B: Methanol
[0056] Carry out gradient elution according to the following table:
[0057]
[0058] The experimental methods not indicating specific conditions in the examples of the present invention are generally in accordance with conventional conditions, or in accordance with the conditions suggested by raw material or commodity manufacturers. Reagents without specific sources indicated are conventional reagents purchased in the market.
Embodiment 1、4
[0059] Example 1, 4-[(2S)-3-cyclopropoxy]phenylpropanoic acid [(4S)-2,2-dimethyl-1,3-dioxolan-4-yl]methyl ester ( The preparation of formula 1)
[0060]
[0061] Add acetonitrile (9.0kg) in reactor, 600g 4-hydroxyphenylpropionic acid [(4S)-2,2-dimethyl-1,3-dioxolan-4-yl] methyl ester and 566g (S )-glycidyl m-nitrobenzenesulfonate, then add potassium carbonate (1.18kg). Then the reaction system was raised to 62-70°C for reaction. After the reaction, the reaction system was cooled down to room temperature. After filtration, the filtrate was concentrated to dryness to obtain an oil, which was directly used in the next reaction.
Embodiment 2
[0062] Embodiment 2, the preparation of N-(2-aminoethyl)-4-morpholine formamide (formula 2 compound)
[0063]
[0064] After adding 30 kg of dichloromethane and 3.0 kg of N-(2-aminoethyl)-4-morpholine formamide oxalate into the reaction kettle, 5.0 kg of anhydrous sodium sulfate and 1.14 kg of sodium hydroxide were added. React at 30°C for 15-18h. After filtration, the filtrate was concentrated to dryness under reduced pressure to obtain about 1.97 kg of light yellow oil, with a yield of 99.5%.
PUM
| Property | Measurement | Unit |
|---|---|---|
| Wavelength | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More