Elemiethanol glycoside buccal tablet and its preparation method and application
A technology of phenethyl glycoside and fenugreek, which is applied in the field of phenethyloside buccal tablets and their preparation, can solve the problems of low utilization rate of fenugreek, and achieve the effects of improving filling efficiency, assisting memory improvement, and preventing water absorption
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
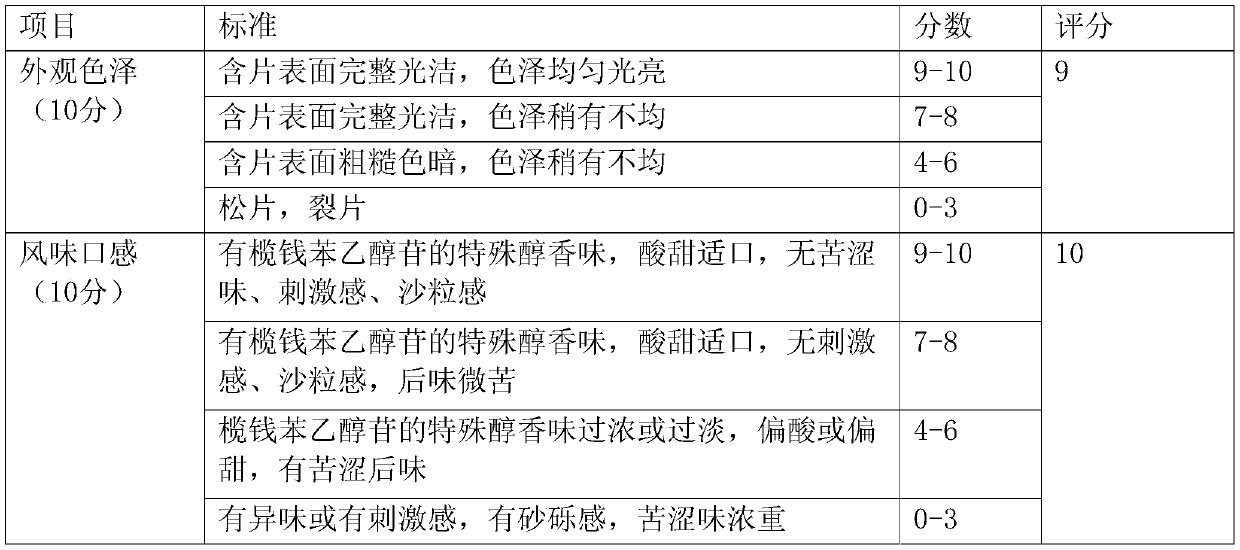
Examples
Embodiment 1
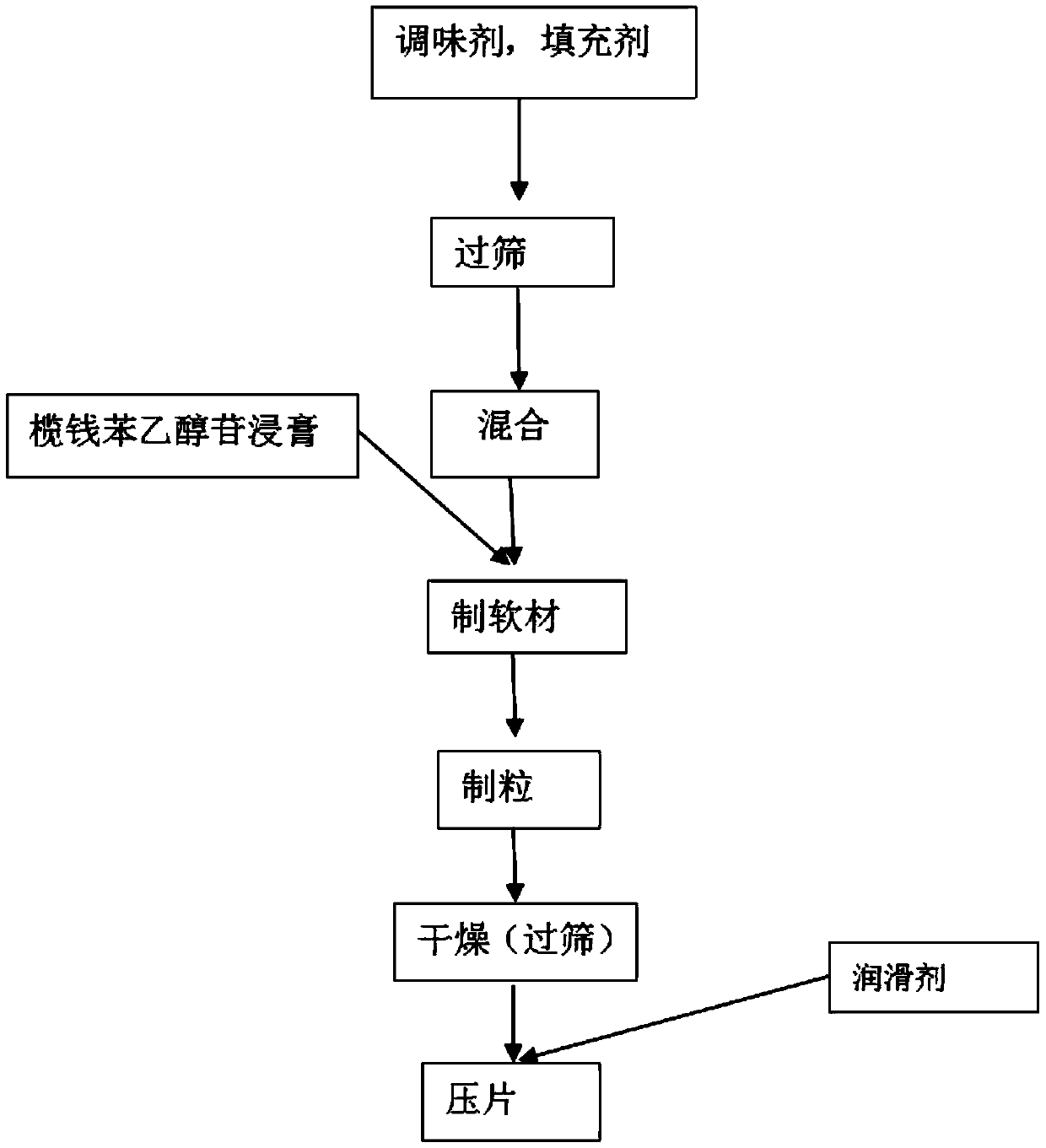
[0033] The elemiethanol buccal tablet is made of the following components in parts by weight: 15 parts of elemiethanol extract, 80 parts of filler, 1.5 parts of lubricant, 1.5 parts of disintegrant, and 2 parts of flavoring agent;
[0034] The filler is 20 parts of pregelatinized starch, 20 parts of lactose, 20 parts of mannitol and 20 parts of xylitol;
[0035] Described lubricant is magnesium stearate;
[0036] Described disintegrant is sodium carboxymethyl starch;
[0037] The flavoring agent is 1 part of citric acid and 1 part of vitamin C.
[0038] The preparation method of described elemiethanol glycoside extract, comprises the steps:
[0039] S1, preparation of Lavender powder: freeze-dry the fresh Lavender fruit, the temperature of the freeze-drying cold trap is 40°C, the vacuum degree is 25Pa, and the final drying is controlled until the water loss rate is 80%. After the money fruit is crushed, the powder of the money is obtained;
[0040] S2. Preparation of elemi...
Embodiment 2
[0061] The elenoside buccal tablet is made of the following components in parts by weight: 10 parts of elemiethanol extract, 70 parts of filler, 1 part of lubricant, 1 part of disintegrant, and 1.5 parts of flavoring agent;
[0062] The filler is 15 parts of pregelatinized starch, 20 parts of lactose, 20 parts of mannitol and 15 parts of xylitol;
[0063] Described lubricant is magnesium stearate;
[0064] Described disintegrant is sodium carboxymethyl starch;
[0065] The flavoring agent is 0.8 part of citric acid and 0.7 part of vitamin C;
[0066] The preparation method of described elemiethanol glycoside extract comprises the steps:
[0067] S1, preparation of Lavender powder: freeze-dry the fresh Lavender fruit, the temperature of the freeze-drying cold trap is -35°C, the vacuum degree is 20Pa, and the final drying is controlled until the water loss rate is 75%, and the freeze-dried After the Lacaria fruit is crushed, the Lacaria powder is obtained;
[0068] S2. Prepa...
Embodiment 3
[0071] The elenoside buccal tablet is characterized in that it is made of the following components by weight: 20 parts of elemiethanol extract, 90 parts of filler, 2 parts of lubricant, 2 parts of disintegrant, flavoring 2.5 parts of agent;
[0072] The filler is 20 parts of pregelatinized starch, 25 parts of lactose, 25 parts of mannitol and 20 parts of xylitol;
[0073] Described lubricant is magnesium stearate;
[0074] Described disintegrant is sodium carboxymethyl starch;
[0075] The flavoring agent is 1.3 parts of citric acid and 1.2 parts of vitamin C;
[0076] The preparation method of described elemiethanol glycoside extract comprises the steps:
[0077] S1, preparation of Lavender powder: freeze-dry the fresh Lavender fruit, the temperature of the freeze-drying cold trap is -45°C, the vacuum degree is 30Pa, and the final drying is controlled until the water loss rate is 85%, and the freeze-dried After the Lacaria fruit is crushed, the Lacaria powder is obtained;...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More