Novel long-acting antipyretic analgesic ibuprofen composition and preparation method thereof
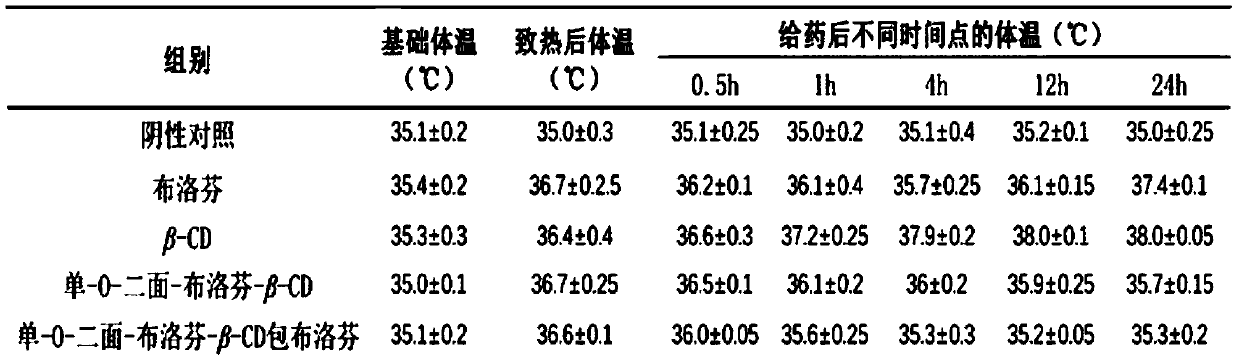
An antipyretic and analgesic composition technology, which is applied in the field of pharmaceutical chemical synthesis, can solve the problems of reducing the antipyretic and analgesic time of ibuprofen, the large fluctuation of ibuprofen blood drug concentration, and the single type of ibuprofen. Avoid the peaks and valleys of the drug effect, improve the effect of the drug effect, and reduce the effect of blood concentration fluctuations
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
preparation example Construction
[0021] A preparation method of a novel long-acting antipyretic and analgesic ibuprofen composition, characterized in that it comprises the following steps:
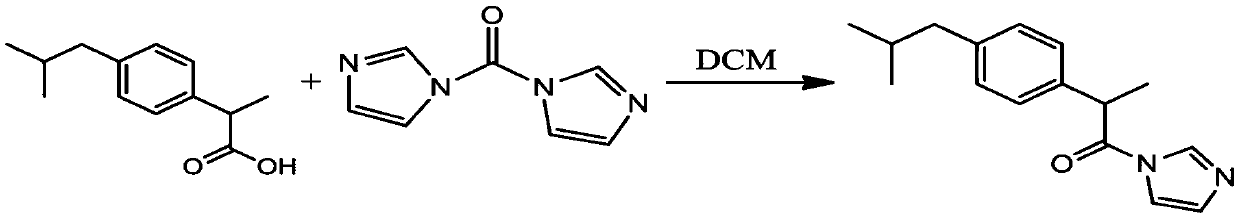
[0022] Step a), the synthesis of ibuprofen imidazolate, ibuprofen is dissolved in dichloromethane, N, N'-carbonyldiimidazole (CDI) is dissolved in dichloromethane, and then the dichloromethane of ibuprofen The methane solution is added dropwise into the dichloromethane solution of CDI through the dropping funnel, stirred evenly until the reaction is completed, and dried after acid-base extraction and n-hexane precipitation to obtain imidazolate ibuprofen;
[0023] The synthetic route of ibuprofen imidazolate is:
[0024]
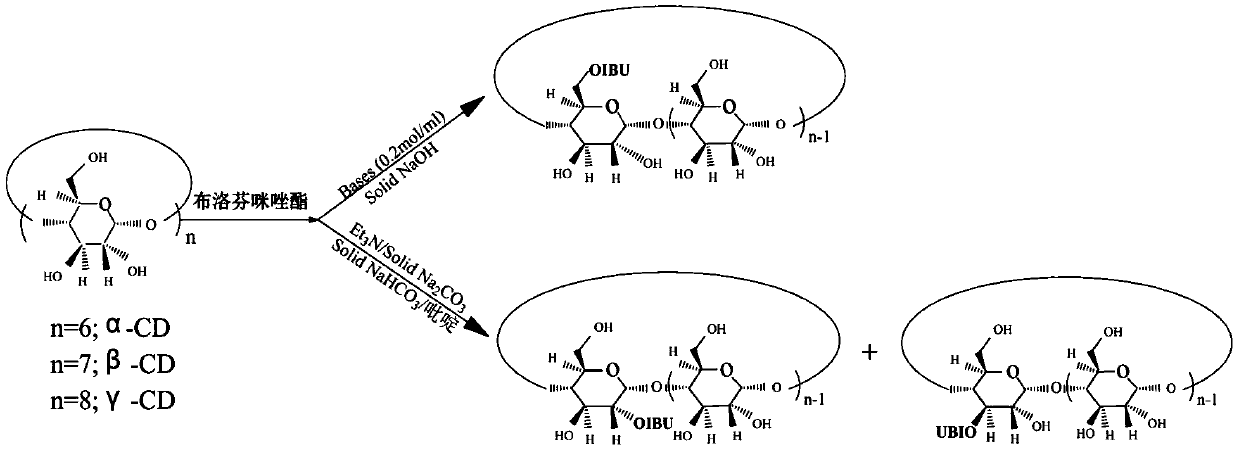
[0025] Step b), the synthesis of ibuprofen-beta-cyclodextrin second side derivatives, the ibuprofen imidazolate prepared in step a) is dissolved in N, N-dimethylformamide (DMF), forming The DMF solution of ibuprofen imidazolate, add β-cyclodextrin in the DMF solution of ibuprofen imidazolate, stir e...
Embodiment 1
[0044] The preparation of ibuprofen imidazolate: ibuprofen 0.01mol (2.06g) is dissolved in 30ml dichloromethane; CDI0.015mol (2.43g) is dissolved in 60ml dichloromethane; The dichloromethane solution of ibuprofen is passed through The dropping funnel was added dropwise into the dichloromethane solution of CDI, reacted for 12-24 hours, extracted with acid and alkali, precipitated with n-hexane and dried to obtain imidazolate ibuprofen (yield 80%).
[0045] The ibuprofen imidazolate that this embodiment makes, nuclear magnetic data is as follows:
[0046] 1 H NMR (400MHz, DMSO-d 6 ):8.49,7.71,7.32,7.30,7.13,7.11,7.01, 4.76,4.74,2.51,2.39,2.37,1.82-1.72,1.49,1.47,0.82,0.80; 13 C NMR (400MHz, DMSO-d 6 ): 171.8, 140.88, 137.63, 130.75, 130.07, 127.53, 117.24, 44.61, 44.36, 40.60-39.44, 29.99, 22.59, 19.59.
[0047] Preparation of ibuprofen-β-cyclodextrin second derivatives: Ibuprofen imidazolate 0.015mol (3.84g) was dissolved in 600mL N,N-dimethylformamide (DMF), and β was adde...
Embodiment 2
[0053] The preparation of ibuprofen imidazolate: ibuprofen 0.01mol (2.06g) is dissolved in 30ml dichloromethane; CDI0.015mol (2.43g) is dissolved in 60ml dichloromethane; The dichloromethane solution of ibuprofen is passed through The dropping funnel was added dropwise into the dichloromethane solution of CDI, reacted for 5-10 hours, extracted with acid and alkali, precipitated with n-hexane and dried to obtain imidazolate ibuprofen (yield 68%).
[0054] Preparation of ibuprofen-β-cyclodextrin second derivatives: Ibuprofen imidazolate 0.015mol (3.84g) was dissolved in 600mL N,N-dimethylformamide (DMF), and β was added under stirring at room temperature - CD 0.0225mol (25.5g), while adding triethylamine (65ml). Continue to stir the reaction at room temperature for 12-24h, and evaporate the solvent under reduced pressure. The residue was chromatographed with excess acetone and filtered to obtain a crude product. The crude product was purified on a C18 reverse-phase column and ...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More