Heat-clearing eight-ingredient pill detection method
A detection method and a technology of a determination method, which are applied in the quality detection of Mongolian medicine and the quality detection field of Mongolian medicine product Qingre Bawei Pills, to achieve the effect of product quality safety and sensitivity improvement
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0076] The identification of embodiment 1 Coptis chinensis:
[0077] Preparation of the test solution: take 3g of this product, grind it finely, add 15ml of 10% sulfuric acid solution, heat and reflux for 15min, let it cool, extract 2 times with chloroform, combine chloroform, concentrate to 2ml, and use as Test solution.
[0078] Preparation of the reference drug solution: take 1 g of the reference drug of Coptis chinensis, add 15 ml of 10% sulfuric acid solution, heat to reflux for 15 minutes, let it cool, extract twice with chloroform, combine the chloroform, concentrate to 2 ml, and use it as the reference drug solution.
[0079] Preparation of reference substance solution: take appropriate amount of vanillic acid reference substance and cinnamic acid reference substance, add chloroform respectively to make a solution containing 2mg per 1ml, as the reference substance solution.
[0080] Identification: According to the thin-layer chromatography test, draw 10 μl of the abo...
Embodiment 2
[0081] The identification of embodiment 2 Kudiding:
[0082] Preparation of the test solution: Take 3g of this product, grind it finely, add 1ml of concentrated ammonia water to infiltrate, add 20ml of chloroform, let it stand for 10h, filter, evaporate the filtrate to dryness, add 1ml of methanol to the residue to dissolve, and use it as the test sample solution.
[0083] Preparation of the reference medicinal material solution: Take 1 g of Kudidin reference medicinal material, add 1 ml of concentrated ammonia water to infiltrate, add 20 ml of chloroform, let stand for 10 h, filter, evaporate the filtrate to dryness, add 1 ml of methanol to dissolve the residue, and use it as the reference medicinal material solution.
[0084] Identification: According to the TLC test, draw 10 μl each of the above-mentioned test solution and the control medicinal material solution, respectively spot on the same silica gel G thin-layer plate, and develop with 7:2:1 cyclohexane-chloroform-metha...
Embodiment 3
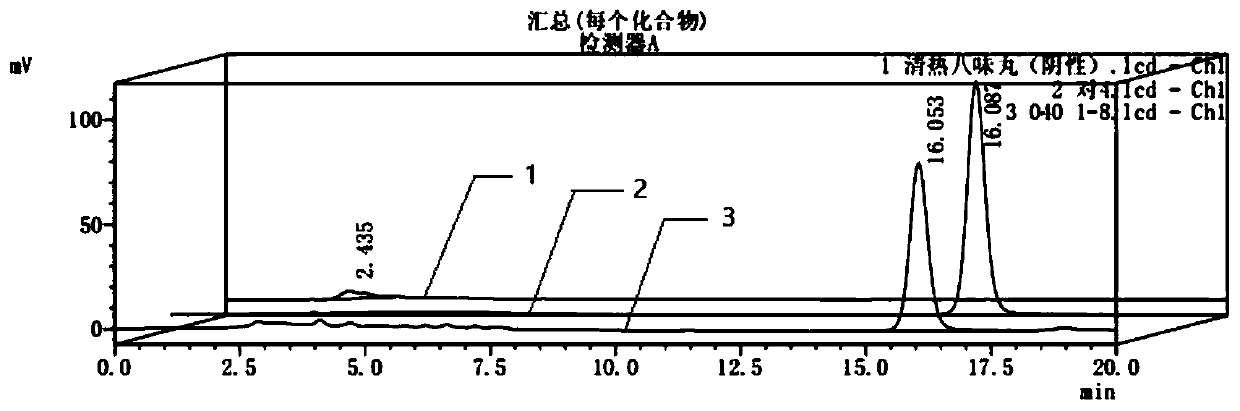
[0085] Embodiment 3 hydroxy safflower yellow A content determination
[0086] HPLC conditions: octadecylsilane bonded silica gel as filler; methanol-acetonitrile-0.7% phosphoric acid in 9:1:40 as mobile phase; detection wavelength 403nm; flow rate 1ml / min; column temperature 35 ℃, the injection volume is 10 μl.
[0087] Preparation of the test solution: take an appropriate amount of this product, grind it finely, take 2g, accurately weigh, put in a 50ml volumetric flask, add an appropriate amount of 25% methanol solution, ultrasonic power 300W, frequency 50kHz for 40min, let cool to room temperature, add Dilute 25% methanol solution to the mark, shake well, pass through a 0.45 μm microporous membrane, and take the subsequent filtrate to obtain the product.
[0088] Preparation of reference substance solution: Take an appropriate amount of hydroxysafflower yellow A reference substance, weigh it accurately, add 25% methanol aqueous solution to make a solution containing 0.13mg ...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More