Urine protein marker for asthma and application thereof
A protein, asthma technology, applied in the field of clinical medicine
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0042]Example 1. Establishment of asthma model
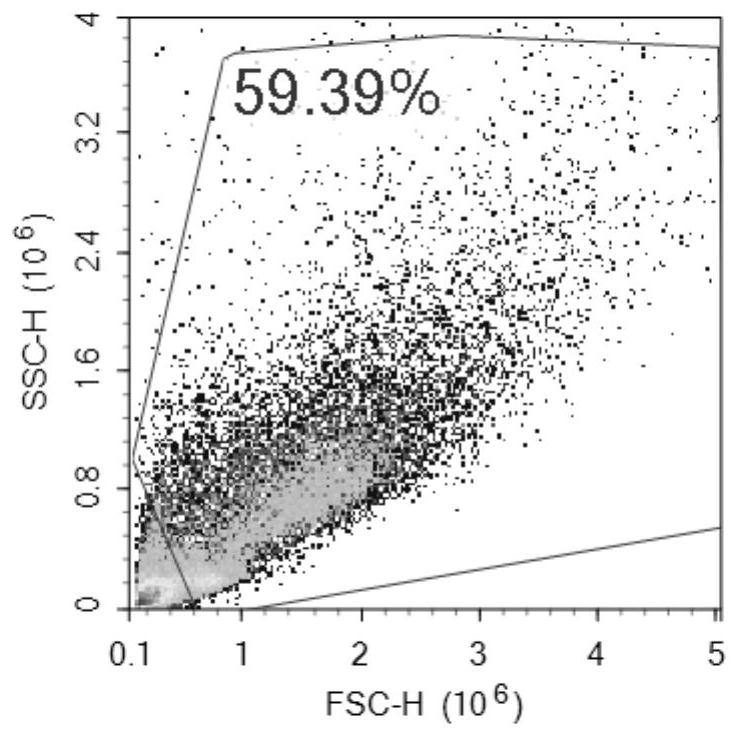
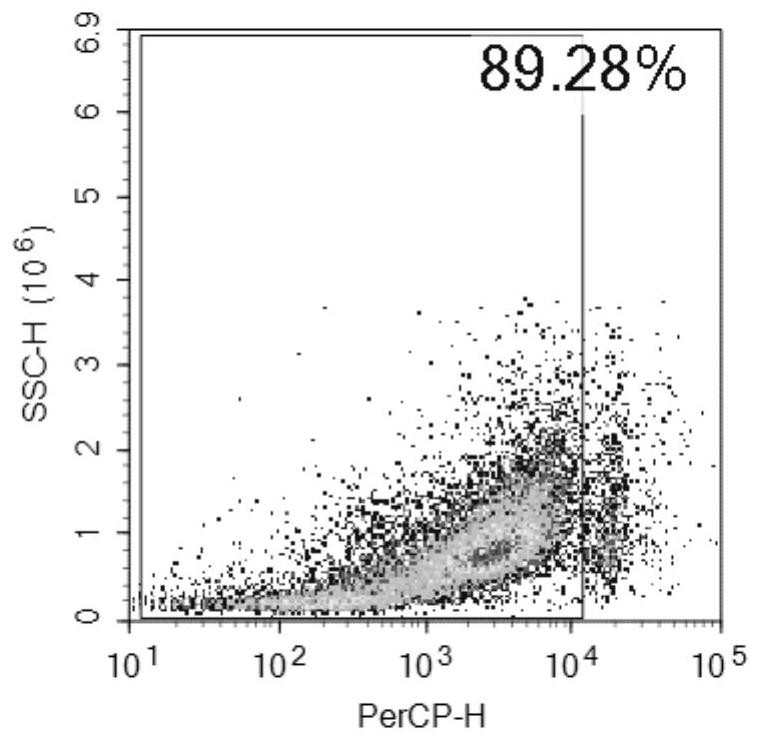
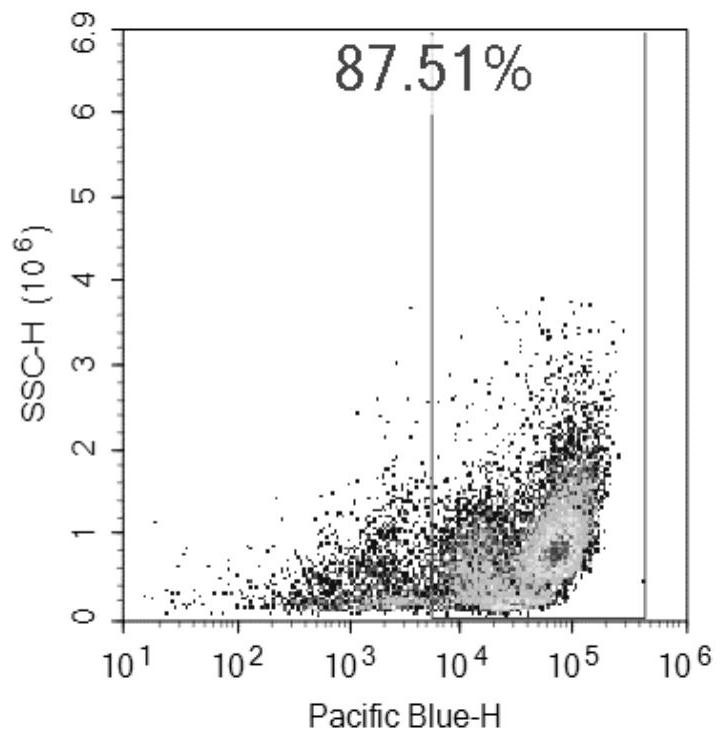
[0043]Take 6-8 weeks old male C56BL / 6 mice, 3 mice in the experimental group and 6 mice in the control group. On day 0 and day 7, intraperitoneal injection of 200uL emulsion (containing OVA: 50ug; Alum: 2mg) was sensitized ; After sensitization, drip the nose from the 14th day to the 16th day, anesthetize the mice with ether and then drip 50ul (including OVA: 50ug). In this example, the inventors injected ovalbumin (OVA) and nasal drops into mice to create a model to simulate asthma. Urine was collected for analysis in the early, early and late stages of asthma.
[0044]1. Materials and reagents
[0045]1) Instrument:
[0046]Orbitrap Fusion Lumos Tribird mass spectrometer was purchased from Thermo Scientific; mouse metabolic cage: purchased from Beijing Jiayuan Xingye Technology Co., Ltd.
[0047]2) Main reagents:
[0048]Chromatography mass spectrometry grade water, acetonitrile, formic acid and methanol were purchased from Fisher; Iodoacetylammonium...
Embodiment 2
[0054]Example 2. Urine sample collection and protein identification
[0055]1. Urine sample collection:
[0056]After modeling, the experimental group and control group mice were placed in metabolic cages to collect urine on the 2, 7, 10, 14, and 17 days.
[0057]2. Extraction of protein in urine by ethanol precipitation
[0058]A total of 15 urine samples from 3 mice on days 2, 7, 14, 17 and 3 control mice were taken to extract protein for subsequent analysis. The method of ethanol precipitation of urine protein refers to the method of Sun W et al. (see Sun W et al. Proteomics, 2005, 5(18):4994-5001).
[0059]The specific steps are:
[0060]1) Take the mouse urine stored in the refrigerator at -80℃, thaw it at 37℃, centrifuge the urine after thawing: take 0.4mL, 12000g, 4℃, centrifuge for 10min; save the supernatant.
[0061]2) Precipitating protein: Add pre-cooled ethanol (1:4) to the retained supernatant, the total volume is about 2 mL.
[0062]3) In the refrigerator at 4°C overnight (-20°C, 2h).
[0063]4...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More