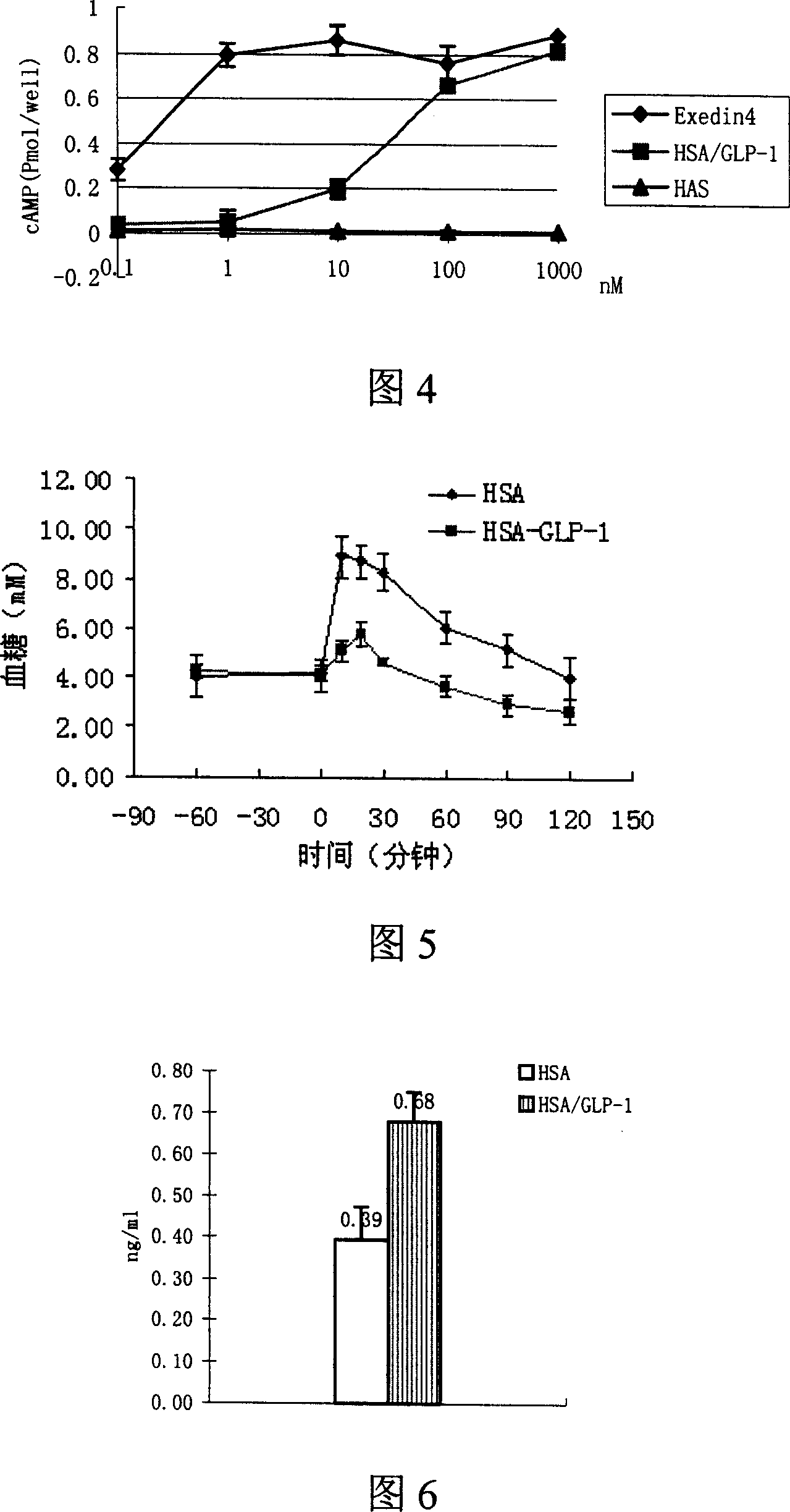
GLP-1 infusion proteins, their preparation and use
A technology of fusion protein and hyperglycemia, applied in the field of medicine, can solve the problems of unknown function of human serum albumin and the like
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
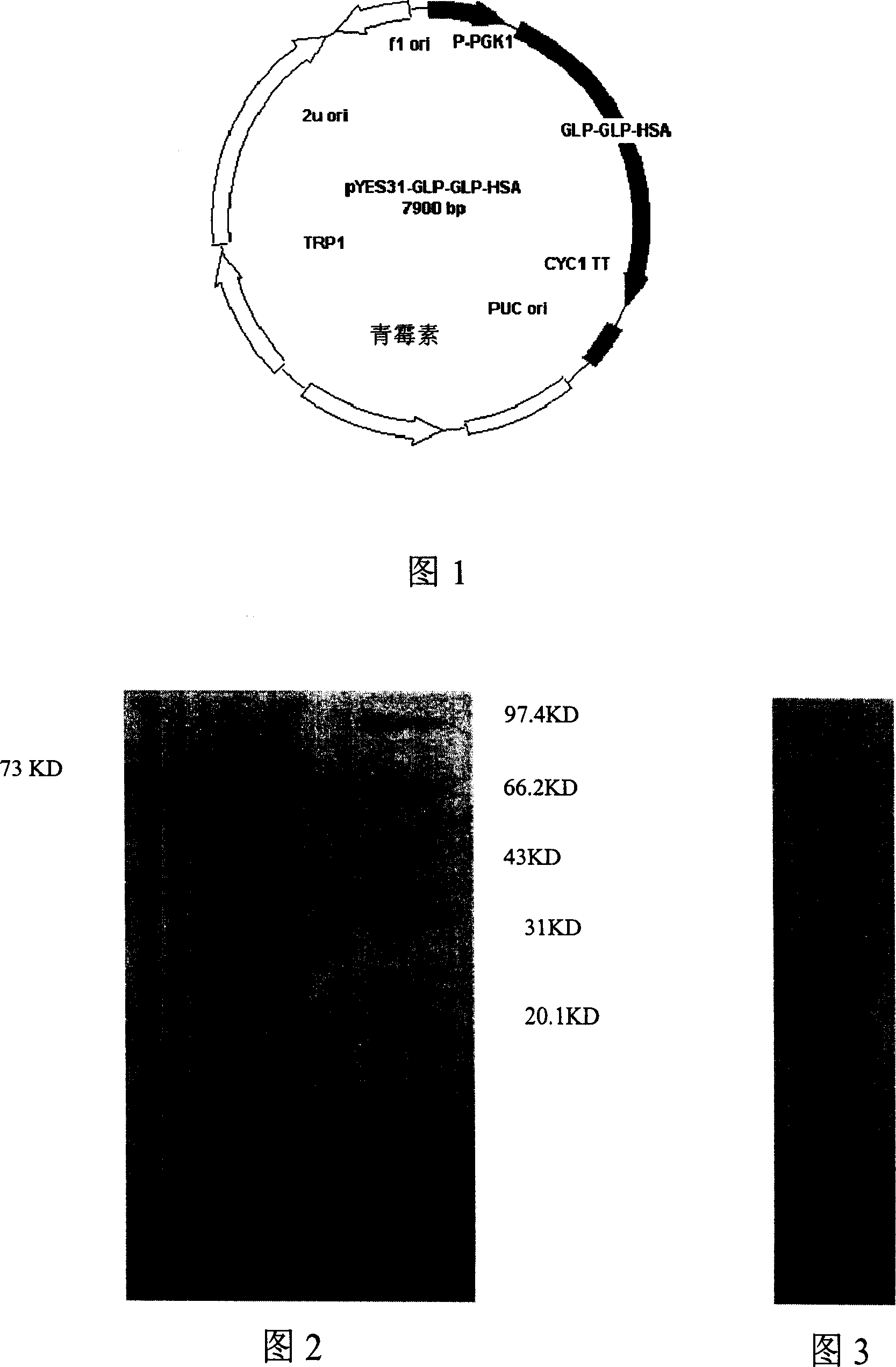
[0080] Example 1: Albumin gene cloning
[0081] Albumin gene can be obtained by RT-PCR. Firstly, human leukocytes are obtained from normal human peripheral blood, and RNA is prepared from them. cDNA was then obtained by the reverse transcription method. The albumin gene was amplified by PCR molecular cloning technique. The specific instructions are as follows:
[0082] Total RNA is first prepared. Specifically, take normal human peripheral blood human leukocytes, then add 1ml Trizol reagent to the leukocytes to lyse the cells, then extract RNA with chloroform and isopropanol, wash with ethanol, and the RNA can be reverse-transcribed by RT-PCR.
[0083] For RT-PCR, the mRNA Selective PCR Kit of Bao Bio Company was used. The specific conditions are as follows.
[0084] 2x mRNA Selective PCR Buffer I, 25 μl, MgCl 2 , 10 μl, dNTP / analog mix, 5 μl, RNase Inhibitor, 1 μl, AMV RNase XL, 1 μl, Oligo dT Primer, 1 μl, RNA, 1 μl, RNase Free dH 2 O, 6 μl. The reaction was carried...
Embodiment 2
[0091] Embodiment 2: GLP-1 gene cloning
[0092] The clone of the GLP-1 gene is obtained by artificial synthesis. The DNA synthesis of 2XGLP (repetitive sequence containing two GLPs) will be described below.
[0093] First, while keeping the amino acid sequence unchanged, the two segments of GLP were artificially synthesized separately. Then, using the PCR method, the two segments of GLP are mutually used as primers for PCR to synthesize the DNA of 2XGLP. The specific conditions are as follows: two sections of artificially synthesized GLP, 10 μl; 10×pfu Buffer, 5 μl; dNTP mixture, 2 μl; pfu DNA Polymerase, 0.5 μl; ddH 2 O, 32.5 μl. Then, put the PCR tube with the sample added into the 96 wells of the PCR instrument, and carry out the reaction according to the following procedure. At the beginning, 94°C; 5min; then, 94°C, 1min; 55°C, 1min, 72°C, 1min, a total of 10 cycles. Finally, 72°C, 5min.
[0094] A leader peptide was added before the fusion of the two GLPs. Obtaine...
Embodiment 3
[0102] Embodiment 3: fusion gene cloning
[0103] In this experiment, the HSA / GLP-1 fusion gene formed by the HSA gene and the GLP-1 gene is taken as an example to illustrate the cloning method of the fusion gene. This method is also suitable for the cloning of fusion genes of HSA gene and other GLP-1 compound genes.
[0104] The clone of fusion protein gene was obtained by Overlap PCR reaction method. details as follows.
[0105] The reaction system for Overlap PCR is: GLP DNA, 2μl; HSA DNA, 2ul; PremixTaq (Ex Taq), 50μl; ddH 2 O, 36 μl.
[0106] The PCR conditions are: start, 94°C; 5min; then, 94°C, 1min; 55°C, 1min, 72°C, 2min, a total of 5 cycles. Then, primers GLP-11 5ul and HSA2 5ul were added. Enter the next program: 94°C, 1min; 55°C, 1min, 72°C, 2min, a total of 30 cycles. Finally, 72°C, 5min.
[0107] Then, the fusion gene clones were isolated and obtained by using common molecular biology methods. After detecting the crude concentration of the recovered produ...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More