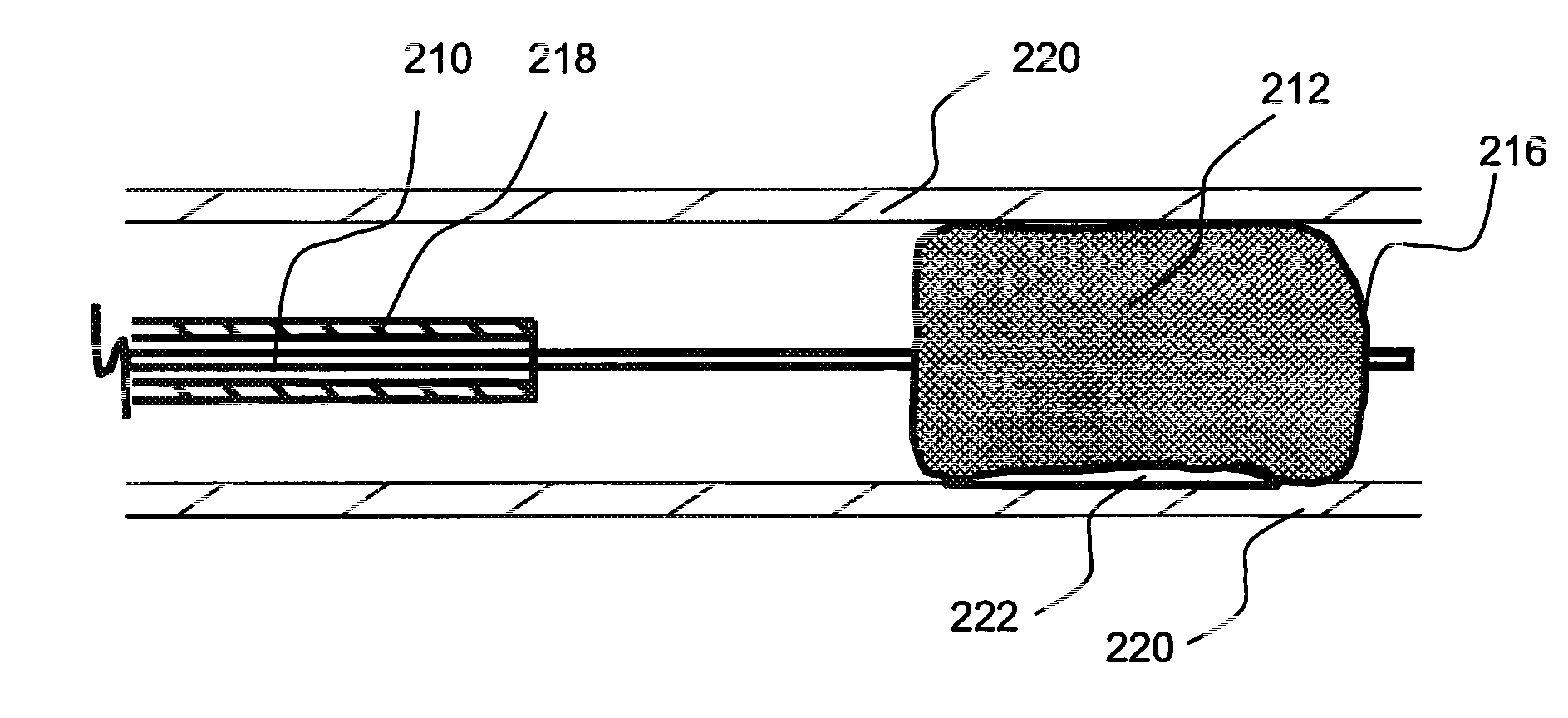
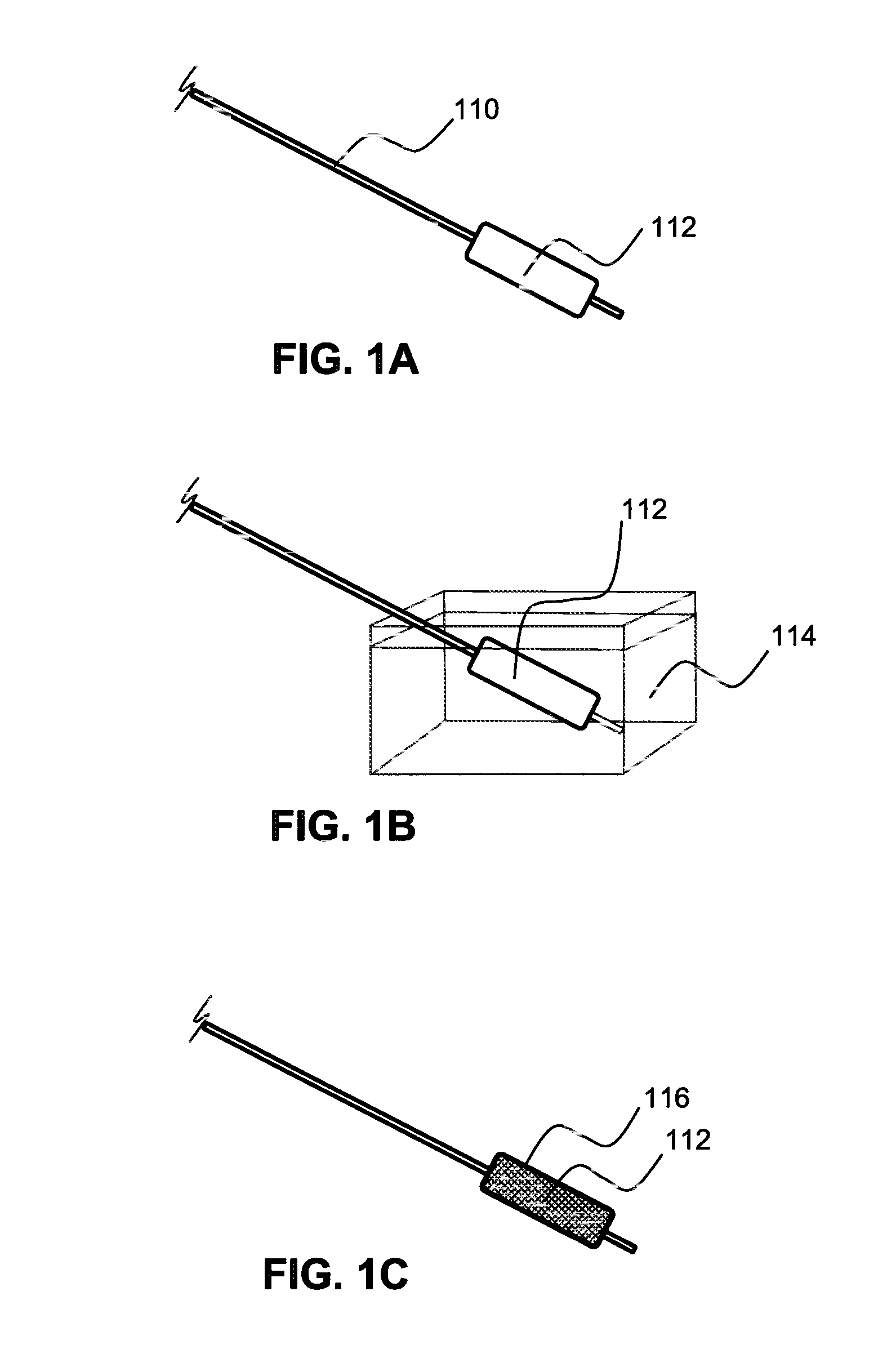
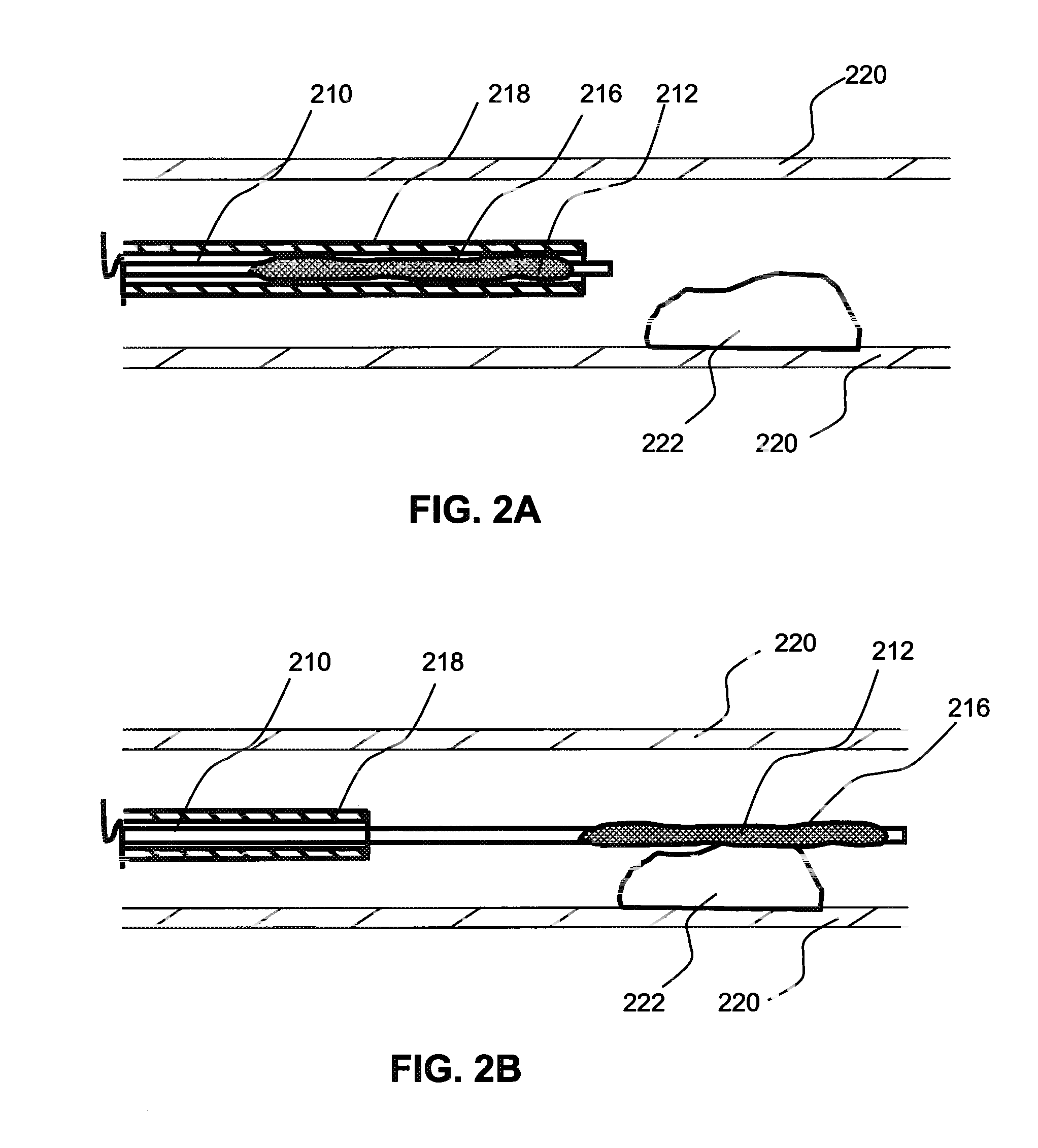
Local delivery of water-soluble or water-insoluble therapeutic agents to the surface of body lumens
a technology of water-soluble or water-insoluble therapeutic agents and local delivery, which is applied in the direction of inorganic non-active ingredients, catheters, drug compositions, etc., can solve the problems of limited efficacy of systemic drug therapy for these diseases, and the dissolution of non-durable amphiphilic polymer coatings, etc., to achieve easy manufacturability of the application process and increase the therapeutic efficiency of the procedure
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
[0059]One (1.0) grams of a 7.5% solution of 60 K Dalton HPC in ethanol is mixed with 0.15 grams of 1% solution of propylene glycol (plasticizer) in acetone, 0.075 grams paclitaxel and 0.08 grams n-butanol. The mixture is heated in a water bath to dissolve the paclitaxel; a clear solution results. When dip coated (single dip) on PET coupons at a dip speed of about 10 inches / minute, and dried at room temperature, there results a slightly milky dry coating. About 3 cm2 of coupon surface is coated per coupon. The average coating density determined by gravimetric analysis is 6 μg / mm2 and the implied paclitaxel density is 3 μg / mm2. The dry coating is sufficiently ductile to withstand a 180 degree bend without cracking or delaminating.
[0060]A coupon coated as above is immersed in 3 ml of 37° C. water for 3 minutes with agitation, after which the coupon is removed and the turbid suspension diluted with 9 ml dimethyl sulfoxide (DMSO) to produce a clear solution. Quantitative UV analysis at 2...
example 2
[0061]0.075 grams paclitaxel is mixed with 0.9 grams of a 20% povidone-iodine solution in 2-propanol, 0.06 grams of a 10% propylene glycol solution in 2-propanol and 0.04 grams acetone. When dip coated (single dip) on a PET coupon at a dip speed of 10 inches / min, and dried at room temperature, there results a clear amber dry coating. About 2.5 μg / mm2 of paclitaxel is deposited.
[0062]The above coupon is immersed in 1.5 ml of 37° C. water for 30 seconds. All of the coating dissolves in the water, and the solution is totally transparent amber, and not turbid as in Example 1.
example 3
[0063]An identical formula to Example 2 is made, however non-iodinated PVP is employed instead of povidone-iodine of the same molecular weight (40 K Dalton). When dip coated (single dip) on a PET coupon at a dip speed of 10 inches / min, and dried at room temperature, there results a clear water white dry coating. About 2.5 μg / mm2 of paclitaxel is deposited.
[0064]This coupon is immersed in 1.5 ml of 37° C. water for 30 seconds. All of the coating polymer dissolves in the water, and the solution shows a suspension of needle crystals. This suspension becomes more turbid after 24 hours, while the above amber solution from Example 2 remains transparent. This demonstrates that the povidone-iodine changes the aqueous solubility of paclitaxel.
PUM
| Property | Measurement | Unit |
|---|---|---|
| temperature | aaaaa | aaaaa |
| temperature | aaaaa | aaaaa |
| angle | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More