Pharmaceutical composition for treatment of osteoarthritis
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
Embodiment Construction
[0027]In prior arts related to treatment of arthritis, most of them increase production of cartilage matrix, instead of suppressing the degeneration and damage of cartilage matrix. The present invention provides a pharmaceutical composition for treatment of osteoarthritis that focuses on suppression of degeneration and damage of cartilage matrix.
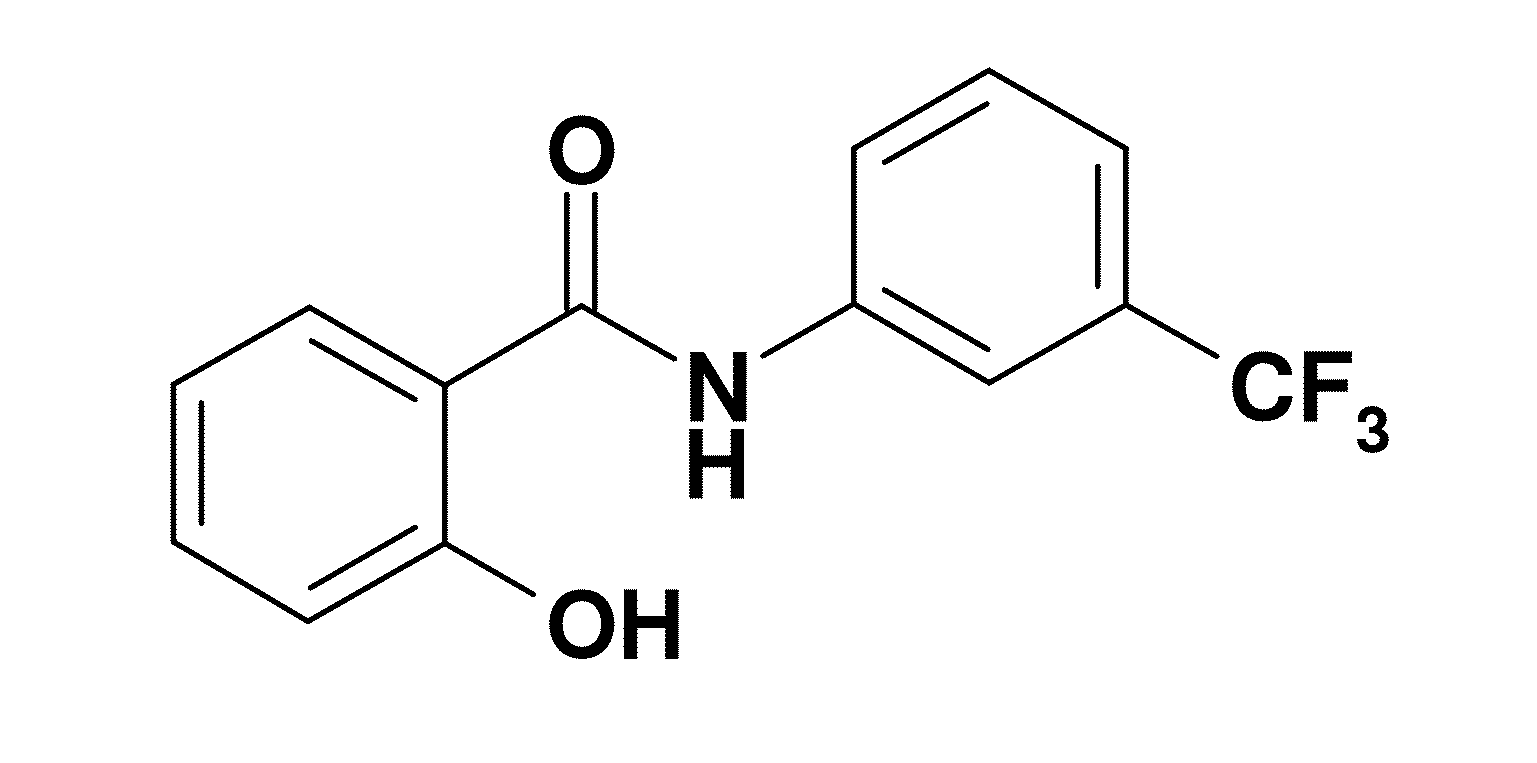
[0028]Refer to FIG. 1, a chemical structure of an embodiment according to the present invention is revealed. As shown in figure, the compound is 2-hydroxy-N-[3-(trifluoromethyl) phenyl]benzamide (hereafter referred to as HS—Cf).
[0029]Refer to FIG. 2, a method for the synthesis of HS—Cf is revealed. As shown in the figure, dissolve 0.276 g (2 mmol) salicylic acid into 10 ml dichloromethane. Then add 0.270 g (2 mmol) 1H-Benzotriazol-1-ol hydrate and 0.270 g (2 mmol) Ethylene dichloride (EDC) into the solution. Next 0.354 ml (2.2 mmol) 3-trifluoromethylaniline is further added into and reacted with the solution for 72 hours. The reacted solutio...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Composition | aaaaa | aaaaa |
| Chemical structure | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com