Polyethylene glycol modified porcine alpha-interferon, preparation method and application thereof
A polyethylene glycol and interferon technology, applied in the field of biomedicine, can solve the problems of undetectable interferon activity, short activity retention time, inconvenient drug use, etc., to achieve long retention time in the body, avoid recognition and removal, reduce The effect of enzymatic hydrolysis
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0051] The preparation method of the porcine alpha-interferon of embodiment 1 polyethylene glycol modification
[0052] 1) Preparation of pig α-interferon borate solution
[0053] Take commercially available porcine α-interferon (dissolved in PBS buffer, purchased from Beijing Luyuan Chuangsheng Biotechnology Co., Ltd.), at 4°C, dialyze with PBS buffer, concentrate, and then replace the buffer , replace porcine α-interferon into boric acid buffer (pH 9.0). Store at 4°C. The protein concentration was determined by the LOWRY method, and the protein concentration was adjusted to 10 mg / ml.
[0054] 2) Preparation of PEG hydrochloric acid solution
[0055] Take 100mg of methoxypolyethylene glycol-amide-propionic acid succinimide ester (mPEG-NHS, molecular weight 20kD, purchased from Beijing Jiankai Technology Co., Ltd.), dissolve it in 0.5ml of 0.1M hydrochloric acid solution, and set aside.
[0056] 3) PEG modification reaction
[0057] The PEG hydrochloric acid solution was ...
Embodiment 2
[0062] The preparation method of the porcine alpha-interferon of embodiment 2 polyethylene glycol modification
[0063] Operate in the same way as in Example 1, the only difference being:
[0064] The pH of the boric acid buffer in step 1) is 8.4, and the concentration of porcine α-interferon protein is adjusted to 2.5 mg / ml.
[0065] In step 2), 100mg of methoxypolyethylene glycol carboxylic acid (mPEG-COOH, molecular weight 30kD, purchased from Beijing Jiankai Technology Co., Ltd.) was taken and dissolved in 1ml of 0.05M hydrochloric acid solution.
[0066] In step 3), the PEG hydrochloric acid solution was added to 10 ml of the borate solution containing porcine α-interferon, and stirred at 500 rpm for 4 hours at 4°C. The pH value was adjusted to 4.8 by adding glacial acetic acid to terminate the reaction. Add 50 ml of sodium chloride solution.
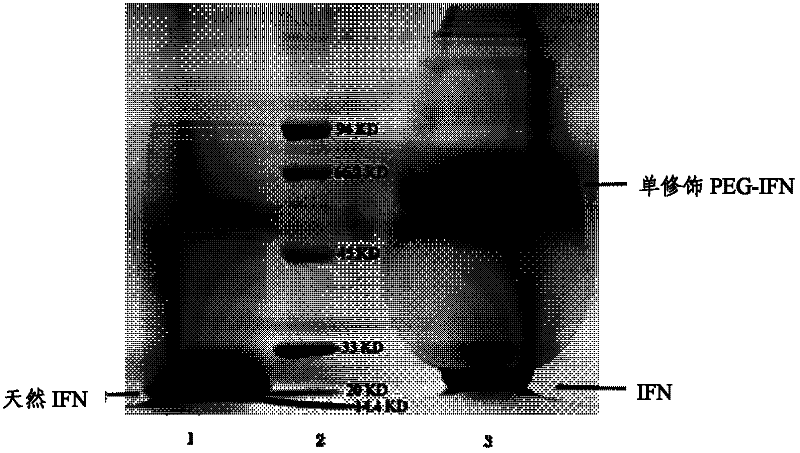
[0067] SDS-PAGE electrophoresis detection results and figure 1 same.
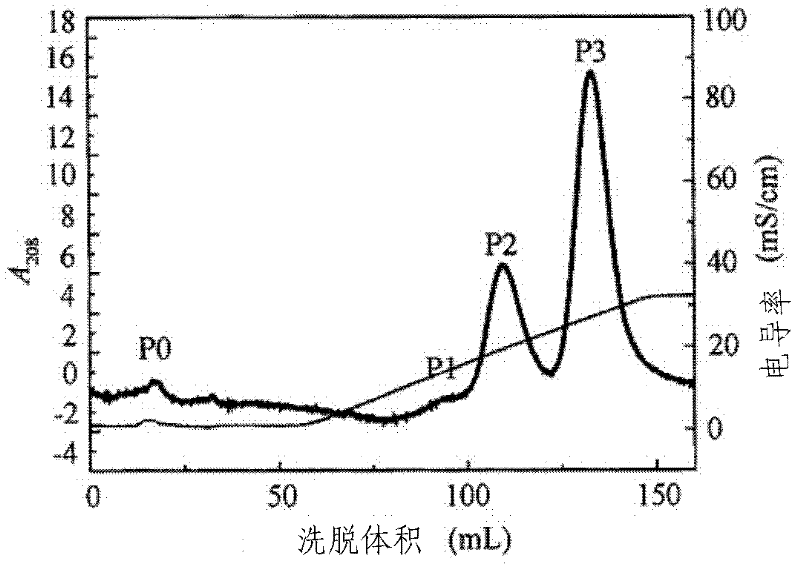
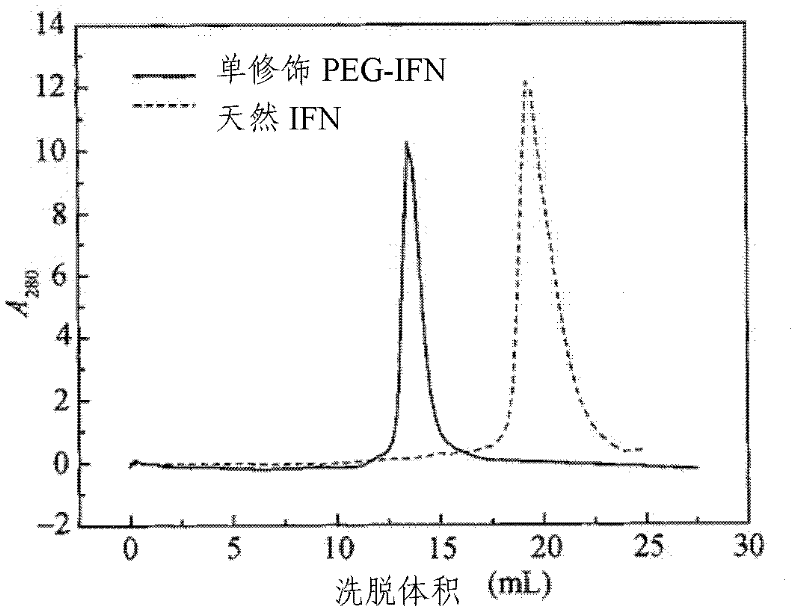
[0068] After cation exchange chromatography, a single...
Embodiment 3
[0069] The preparation method of the porcine alpha-interferon of embodiment 3 polyethylene glycol modification
[0070] Operate in the same way as in Example 1, the only difference being:
[0071] The pH of the boric acid buffer in step 1) is 8.7, and the concentration of porcine α-interferon protein is adjusted to 5 mg / ml.
[0072] Step 2) get 100mg methoxy polyethylene glycol amine (mPEG-NH 2 , molecular weight 40kD, purchased from Beijing Jiankai Technology Co., Ltd.), dissolved in 0.5ml of 0.1M hydrochloric acid solution.
[0073] In step 3), the PEG hydrochloric acid solution was added to 6 ml of borate solution containing porcine α-interferon, and stirred at 800 rpm for 4 hours at 4°C. The pH value was adjusted to 4.5 by adding glacial acetic acid to terminate the reaction. Add 50 ml of sodium chloride solution.
[0074] SDS-PAGE electrophoresis detection results and figure 1 same.
[0075] After cation exchange chromatography, a single PEG-modified porcine α-inter...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More