Rapid detection method of contents of multiple components in Chinese patent medicinal traumatic injury treatment tablet
A detection method and technology of Dieda tablets, which are applied in the directions of measuring devices, material separation, and material analysis, can solve the problems of poor quality controllability of Dieda tablets and the inability to comprehensively and effectively measure the quality of Dieda tablets, and achieve Avoid wasting time and reagents, control quality, and the effect of easy operation of the method
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment Construction
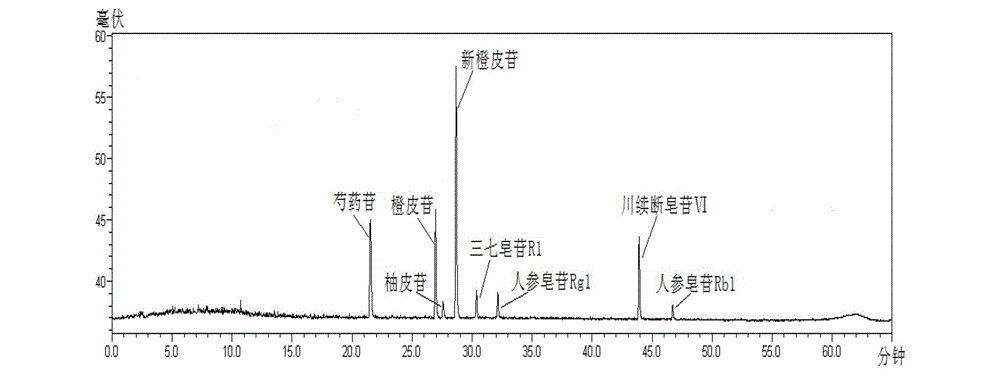
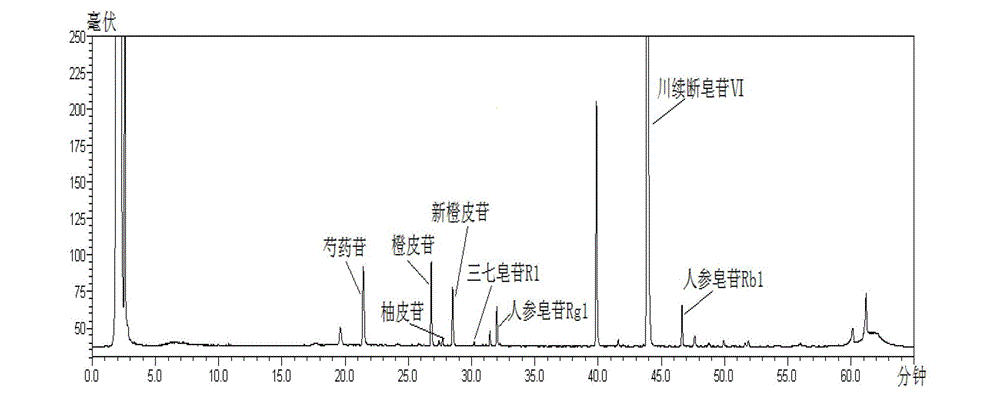
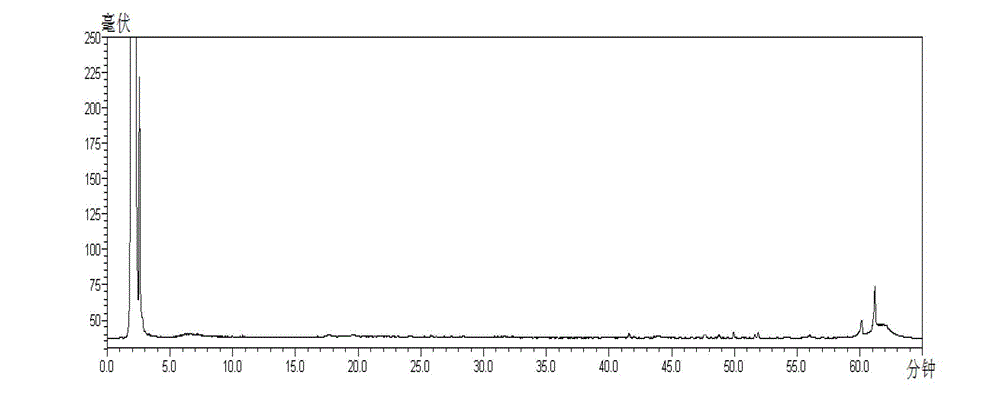
[0020] Detailed examples are given below.
[0021] The present invention adopts: instruments and reagents: Shimadzu LC-20AT high performance liquid chromatography; Alltech ELSD2000ES evaporative light scattering detector; Sartoris BP211D electronic balance; acetonitrile is a reagent for chromatography, and other reagents are analytically pure; China National Institutes for Food and Drug Control, batch number: 110736-201136), naringin reference substance (National Institute for Food and Drug Control, batch number: 110722-201111), hesperidin reference substance (National Institute for Food and Drug Control, batch number: 110721 -200613), neohesperidin reference substance (National Institute for Food and Drug Control, batch number: 111857-201102), notoginseng saponin R 1 Reference substance (National Institute for Food and Drug Control, batch number: 110745-200617), ginsenoside Rg 1 Reference substance (National Institute for Food and Drug Control, batch number: 110703-201027), ...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com