Compound preparation containing lutein ester and preparation method thereof
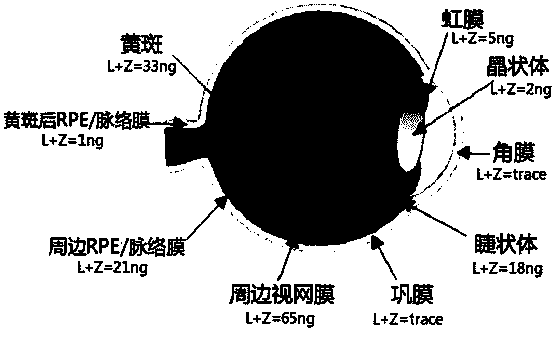
A technology of compound preparation and lutein ester, applied in the field of prevention or treatment of age-related macular degeneration and relief of visual fatigue, compound preparation containing lutein ester and its preparation, can solve the problem of low bioavailability and high processing cost , poor stability and other issues, to achieve high bioavailability, prevent damage, and protect the retina
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0060] Example 1: Lutein Ester Capsules Containing Lycopene, Lycium Barbarum Extract and Cassia Seed Powder
[0061] Prescription (based on the total content of each drug 200mg):
[0062]
[0063]
[0064] Preparation:
[0065] Feed lutein esters, lycopene, wolfberry fruit extract, zinc gluconate, vitamin C and starch in the amount of the above prescription in 10000 times, pass through 80 mesh sieves respectively to obtain fine powder, and then mix with cassia seed powder evenly , add starch slurry to make soft material, granulate, dry in an oven at 60°C; then add magnesium stearate (magnesium stearate has passed through an 80-mesh sieve in advance), mix evenly, granulate, and fill into capsules.
Embodiment 2
[0066] Example 2: Lutein Ester Capsules Containing Lycopene, Lycium Barbarum Extract and Cassia Seed Powder
[0067] Prescription (based on the total content of each drug 200mg):
[0068]
[0069] Preparation:
[0070] In this example, sucrose is used as diluent, low-substituted hydroxypropyl cellulose is used as disintegrant, talcum powder is used as glidant, and water is used as binder. Dried in an oven, all the other preparation methods were the same as in Example 1.
Embodiment 3
[0071] Example 3: Lutein Ester Capsules Containing Lycopene, Wolfberry Fruit Extract and Cassia Seed Powder
[0072] Prescription (based on the total content of each drug 200mg):
[0073]
[0074] Preparation:
[0075] In this embodiment, glucose is used as a diluent, sodium carboxymethyl starch is used as a disintegrant, sodium lauryl sulfate is used as a glidant, and ethanol is used as a binder. Dry in an oven at 50°C, and the rest of the preparation method is the same as in Example 1.
PUM
| Property | Measurement | Unit |
|---|---|---|
| Particle size | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More